Chemical changes
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
What category of acids are weak acids?
carboxylic acids
Each time the pH increases/decreases by 1, how much does the H+ concentration change by?
-For each increase by 1 on the pH scale, the concentration of H+ ions gets 10x lower
-For each decrease by 1 on the pH scale, the concentration of H+ ions gets 10x higher
What is a neutralisation reaction?
A reaction between an acid and a base, in which H+ ions from the acid react with OH- ions from the base
What is a base?
-A substance which neutralises an acid to make salt + water
-e.g metal oxides, hydroxides and carbonates
What is the ionic equation for a neutralisation reaction?
H+ + OH- → H₂O
What is the general word equation for a reaction between an acid and a metal hydroxide?
acid + metal → salt + water
Why are strong acids more reactive than weak acids?
They have a greater concentration of H+ ions, so more of them will react. This increases the rate of reaction
What is a salt?
A substance that can be made by reacting a (reactive) metal or base with an acid
What is the general word equation for a reaction between an acid and a metal?
acid + metal →salt + hydrogen
What are acids made of?
-Freely moving ions because they conduct electricity
-H+ ions because hydrogen gas is produced at the cathode (-) when a current passes through the acid
What is an alkali?
A base that dissolves in water to form a solution with a pH greater than 7
How does pH differ between strong and weak acids (assuming they’re at the same concentration)?
Strong acids have a lower pH as it will ionise more of fully so have a higher concentration of hydrogen ions
What is the general word equation for a reaction between an acid and a metal oxide?
acid + metal oxide → salt + water
What is a strong acid?
-An caid which completely ionises in an aqueous solution
-Therefore it contains a higher concentration of H+ ions
-And have a lower pH and are better conductors (high conductivity) than weak acids
What is the general word equation for a reaction between an acid and a metal carbonate?
acid + metal carbonate → salt + water + carbon dioxide
What is pH?
a measure of the concentration of H+ ions in a solution
What are alkalis made of?
-Freely moving ions because they conduct electricity
-When an electric current is passed through an alkali, oxygen gas is produced at the anode
-Contain OH- ions
What is a weak acid?
an acid which only partially ionise in an aqueous solution
What is the effect of using reactants with increased reactivity in a reaction?
The reaction will take place more quickly, or more vigorously
How can you investigate the reactivity of a metal using an acid?
-acid + metal → salt + hydrogen
-First react the metal with an acid
-When the metal reacts, bubbles of hydrogen will be released
-The faster the bubbles are produced, the faster the reaction is, so the more reactive the metal must be
How can you investigate the reactivity of metals using temperature change?
More reactive metals will lead to larger temperature changes
-React each of the metals with either water or acid over a set period of time
-Ensure that the surface area and the mass of the metal is the same each time
-Measure the rise in temperature
-The greater the temp change, the more reactive the metal must be
Why can it be dangerous to react very reactive metals with acids or water?
Very reactive metals can react explosively and produce many bubbles
Which metals don’t react with water?
Less reactive metals, such as zinc, iron and copper
In displacement reactions, which ions are reduced?
Metal ions are reduced - they gain electrons
Will acids react more vigorously with hot or cold water?
Hot - the higher temp results in a faster rate of reaction
Would powdered metal, or a single chunk (both the same mass) react more vigorously?
powdered - larger surface area
What is a metal displacement reaction?
a reaction in which a more reactive metal displaces a less reactive metal
What is an oxidation reaction (in terms of oxygen)?
a substance reacting with oxygen
What does a metal form when it is oxidised (with oxygen)?
metal oxide
What is a reduction reaction (in terms of oxygen)?
a substance losing oxygen
What form are metals normally found it in the environment?
usually bonded to other elements, most often oxygen
referred to as ores
How can metals be extracted from their ores?
-Reduction with carbon
-Electrolysis
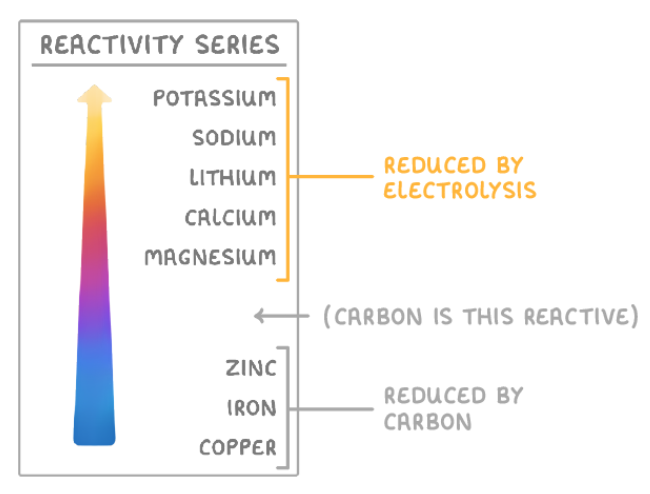
Which metals can be reduced with carbon?
Metals less reactive than carbon: zinc, iron and copper
Describe how metal ores can be reduced using carbon
-React the metal ore with carbon
-Carbon will displace the metal as it is more reactive
What is the balanced symbol equation for the reduction of FeO with carbon?
2FeO + C → 2Fe + CO₂
Why do some metals not require extraction?
Some metals are so unreactive that they exist in the earth as pure metals (not bonded to any other element)
What is the order of metals in the reactivity series?
What is oxidation (in terms of electrons)?
the loss of electrons
What is reduction (in terms of electrons)?
the gain of electrons
What is a redox reaction?
A reaction in which both oxidation and reduction take place
What is a half equation?
An equation that shows the gain or loss of electrons of a single element in a reaction
In displacement reactions, are the metal ions reduced or oxidised?
reduced - gain electrons
In displacement reactions, are the metal atoms reduced or oxidised?
oxidised - lose electrons
What is shown in ionic equations?
Only the particles that react and the products they form are shown - the ions that don’t change are ignored
What are ions not involved in equations called?
spectator ions
What does electrolysis mean?
refers to breaking something apart using electricity - a process used to separate a compound into its elements
What is an electrolyte?
a liquid or solution that conducts electricity
What is an electrode?
a solid that conducts electricity and is submerged in the electrolyte
Why is electrolysis not used to extract all metals?
it is very expensive as it requires a lot of energy
What happens to positive ions during electrolysis?
-They are attracted to the cathode (negative electrode)
-They gain electrons
-This means they have been reduced
What happens to the negative ions during electrolysis?
-They move towards the anode (positive electrode)
-They lose electrons
-This means they have been oxidised
Why can’t ionic solids be electrolysed?
Ionic solids can’t be electrolysed as the ions are in fixed positions - they can’t move and carry charge
Why can molten ionic compounds be electrolysed?
The ions can move freely when the ionic compound is molten and thus they can conduct electricity
Why should the electrodes be unreactive during electrolysis?
So they don’t react with the electrolyte and dissolve away, requiring replacement
How does electrolysis work?
-An electric current is passed through the electrolyte causing ions to move to the electrodes
-Positively charged ions will move to the cathode
-Negatively charged ions move to the anode
-Ions are discharged at the electrodes, producing elements
What ore is aluminium extracted from via electrolysis?
bauxite - the ore which contained aluminium oxide
What is used in the electrolysis of aluminium oxide and why?
cryolite - to lower the melting point
In the electrolysis of molten aluminium oxide, which 2 ions would be present at the electrolyte?
Al³+ and O²-
In the electrolysis of molten aluminium oxide, what would form at the anode?
oxygen gas
In the electrolysis of molten aluminium oxide, what would form at the cathode?
molten aluminium
In the electrolysis of molten aluminium oxide, what would be the half equation for the reaction at the anode?
2O²- → O₂ + 4e-
In the electrolysis of molten aluminium oxide, what would be the half equation for the reaction at the cathode?
Al³+ 3e- → Al
What is the overall balanced symbol equation for the separation of aluminium oxide during electrolysis?

In the electrolysis of molten aluminium oxide are the Al ions oxidised or reduced?
Reduced - they gain electrons
In the electrolysis of molten aluminium oxide are the oxygen ions oxidised or reduced?
oxidised - they lose electrons
In the electrolysis of a solution, which 2 ions are present from the water molecules themselves?
H+ ions and OH- ions
In the electrolysis of aqueous solutions, what is the rule for determining which ion will be oxidised at the anode?
It will always be the OH- unless there is a halide ion present
In the electrolysis of aqueous solutions, what is the rule for determining which ion will be reduced at the anode?
The H+ ion unless there is a less reactive element present such as copper
Why is aqueous electrolysis generally cheaper than molten electrolysis?
In aqueous electrolysis the substance is just dissolved in water, no energy is required to melt it