Reactions of ions in aqueous solutions
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What happens to metal ions in aqueous solutions?
They form metal-aqua ions
What colour solution does Fe2+ form in water
Give the formula of the metal aqua ion formed
Pale green
[Fe(H2O)6]2+
What colour solution does Cu2+ form in water
Formula of metal aqua ion produced
Pale blue
[Cu(H2O)6]2+
What colour solution does Al3+ form in water
Formula of metal aqua ion produced
Colourless
[Al(H2O)6]3+
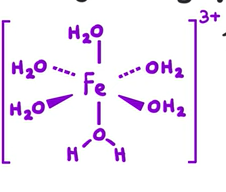
What colour solution does Fe3+ form in water
Formula of metal aqua ion produced
Yellow
[Fe(H2O)6]3+
Why are positively charged metal aqua ions soluble in water?
Attracted to the partially negative oxygen atom of H2O
Metal-aqua ions are weakly…
Acidic (Have potential to release H+)
Why is the acidity of metal aqua ions with a 3+ charge greater than the acidity of metal aqua ions with a 2+ charge
M3+ has a higher charge density (M= Metal)
Polarises the O-H bond more strongly
Weakens O-H bond
O-H bond breaks
So ion is a better H+ donor
Observation and equation of [Fe(H2O)6]2+ reacting with NaOH
[Fe(H2O)6]2+ + 2OH- → Fe(H2O)4(OH)2 + 2H2O
Green ppt
Observation and equation of [Cu(H2O)6]2+ reacting with NaOH
[Cu(H2O)6]2+ + 2OH- → Cu(H2O)4 (OH)2 + 2H2O
Blue ppt
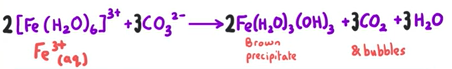
Observation and reaction of [Fe(H2O)6]3+ reacting with NaOH
[Fe(H2O)6]3+ + 3OH- → Fe(H2O)3 (OH)3 + 3H2O
Brown ppt
Observation and equation for [Al(H2O)6]3+ reacting with NaOH
[Al(H2O)6]3+ + 3OH- → Al(H2O)3(OH)3 + 3H2O
White ppt
What happens to Al(H2O)3(OH)3 in excess NaOH and why
Equation
Ppt produced dissolves in excess NaOH
Because aluminium is amphoteric→ Al(H2O)3(OH)3 acts as an acid, reacting with excess OH ions in a further deprotonation reaction to form a colourless solution
[Al(H2O)3(OH)3] + OH- →[Al(OH)4)]1- + 3H2O
Observation and equation of [Fe(H2O)6]2+ reacting with ammonia
[Fe(H2O)6]2+ + 2NH3 → Fe(H2O)4(OH)2 + 2NH4-
Green ppt
Observation and equation of [Fe(H2O)6]3+ with ammonia
[Fe(H2O)6]3+ + 3NH3 → Fe(H2O)3(OH)3 + 3NH41+
Brown ppt
Observation and equation of [Al(H2O)6]3+ reacting with ammonia

Observation and equation of [Cu(H2O)6]2+ reacting with ammonia
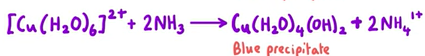
What happens when excess ammonia is added to (Cu(H2O)4(OH)2 and give equation
In excess, ammonia acts as a ligand substituting out the water ligands

Why cant M2+ complexes react with Na2CO3 and what happens instead
They are weakly acidic
So cant react in an acid-base reaction with Na2CO3 (very weak base)
Instead these reactions produce carbonate ppts
Observation and equation of [Fe(H2O)6]2+ reacting with sodium carbonate
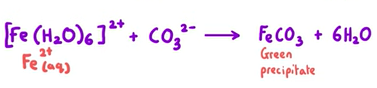
Observation and equation of [Al(H2O)6]3+ with sodium carbonate

Observation and equation of [Fe(H2O)6]3+ reacting with sodium carbonate