Chemistry all Y9 Notes & Flashcards [end of years revision]
1/106
Earn XP
Description and Tags
Separation & Purification, Atomic structure, Ionic bonding, Groups of the periodic table (groups 1, 7, 0)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
Physical changes
changes in state that can be reversed and the chemical properties of what you’re changing stay the same
Particle model of gas
random, far apart, moving fast in all directions
Particle model of liquid
random, close, flowing around each other
Particle model of solids
regular, close, vibrating about a fixed position
Filters
things that separate mixtures by letting smaller things through but trapping bigger things (insoluble substances)
Solute
a dissolved substance
Solution
a mix of solutes
Solvent
what solutes dissolve in
Saturated solution
solution containing the most amount of solute that can dissolve in that amount of solvent
Crystallisation definition
a method of separation involving evaporating the solvent to leave the solutes that forms solid crystals
What happens if crystals form slowly in crystallisation
particles have longer to form an ordered pattern and will make larger crystals
What happens if crystals form quickly in crystallisation
particles have less time to form an ordered pattern and will make smaller crystals
Crystallisation risk asessment
Wear eye protection
Use steam to heat the evaporating basin gently
Remove the bunsen burner before the solution is completely dry
Care must be taken not to overheat the solution as hot crystals may spit out
Crystallisation method
The filtrate is placed above a bunsen burner
The bunsen burner evaporates the filtrate carefully
Crystals are left to cool
Crystals are scraped out
Crystals are patted dry with cloth / filter paper
What happens if you heat crystals more after they have formed in crystallisation
they may change chemically
Filtration method
A filter tunnel is lined with filter paper
The solvent and the solute pass through
This forms the filtrate
Bits of insoluble substances that can’t fit through form the residue
Residue
bits of insoluble substances that don’t get through the filter paper and are left in it
Distillation definition
a way of separating multiple liquids by boiling them one by one
Simple distillation method
Liquid is evaporated and turns to gas (vapour)
Other materials with higher boiling points are left behind
The vapour of the first liquid is now pure. It is condensed back into a liquid
Still
the apparatus used for distillation
Ways to make a still more efficient
Plugging gaps in apparatus with bungs to stop any gas escaping
Condenser: a tube surrounded with a jacket of cold water (used instead of a delivery tube). Helps liquid condense quicker with lowered temperatures
Anti-bumping granules: makes the liquid boil more smoothly, reduces risk of liquid boiling over (put in the solution)
Fractional distillation method
Solution is put in a distillation flask and heated
The first substance’s vapour (the one with the lowest boiling point) rises up a fractioning column
A temperature gradient is created, with the hottest temperatures at the bottom
The substance with the lowest boiling point reaches the top first and passes into the condenser and is collected
The same happens with the rest of the substances, one by one
Real life examples of fractional distillation
Separate different products of crude oil
Make alcoholic drinks like whisky and vodka
Separate gasses in the air
Composition of a pure substance…
can’t be changed, is the same everywhere in the substance
Can compounds be pure?
yes, if its the only thing in the substance
Mixture
substance containing elements and/or compounds not chemically joined together. they can be separated and don’t have a fixed melting point
Melting point
the temperature at which a substance’s particles gain enough energy to overcome the forces of attraction between them and change state
Mixtures have melting points that…
aren’t fixed, instead they melt over a range of temps. this is because there different things with different melting points inside it
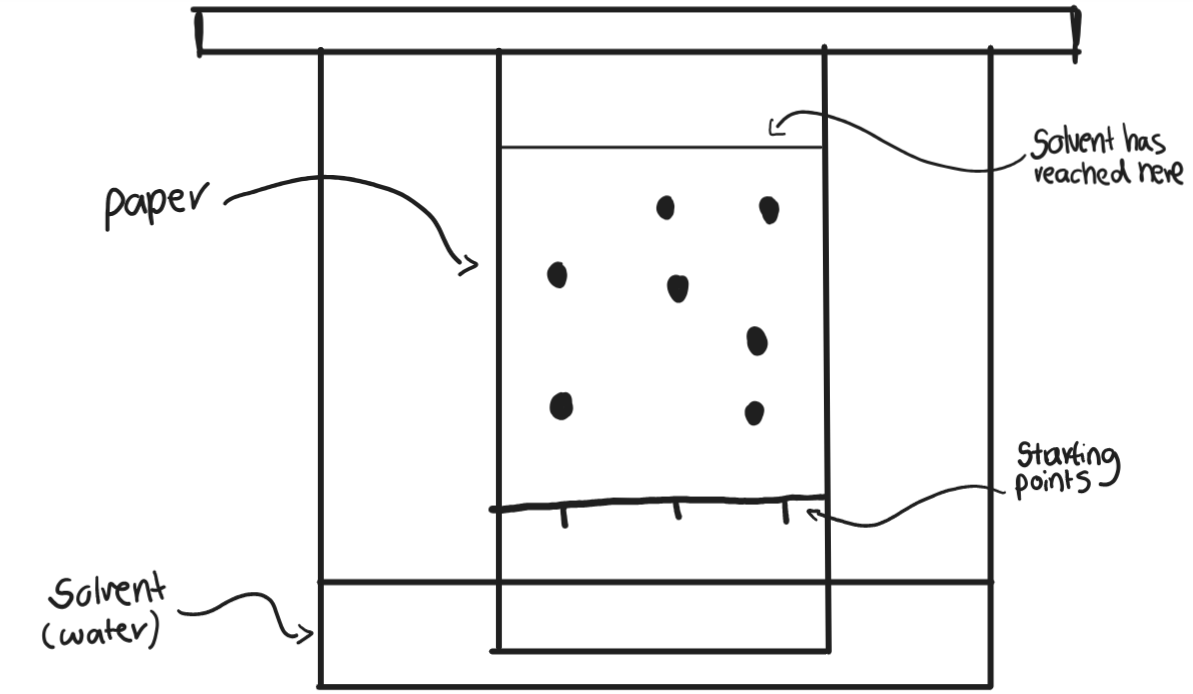
Chromatography
when a solvent moves along paper, different substances in it move at different speeds so separate
Mobile phase in chromatography
the solvent (e.g. water)
Stationary phase in chromatography
the paper
Chromatogram
the piece of paper with the separated components on it
Rf value
distance moved by spot
distance moved by solvent
measurements are made from the starting points of the solvent
Mendeleev
Russian chemist credited with the first periodic table
Mendeleev’s periodic table arrangement
he arranged the elements in increasing order of atomic masses, and elements with similar properties were near each other
Mendeleev’s period table gaps & predictions
He left gaps in his table for unknown elements, and so was able to predict the properties of them by looking at where they fitted and the properties of the elements around them
Mendeleev’s period table faults
His law couldn’t explain the existence of isotopes or where they would go on his table. As they have different atomic masses, they should have been in a separate place, but Mendeleev didn’t give them their column, which didn’t make sense with his law.
Structure of atoms
central nucleus containing protons and neutrons, and electrons arranged in shells surrounding it
What charge do protons have
positive (+1)
What charge do neutrons have
neutral (0)
What charge do electrons have
negative (-1)
Atomic number
number of protons and number of electrons (same for both)
Mass number
number of protons + number of neutrons
aka atomic number + neutrons
How to work out number of protons
atomic number
How to work out number of electrons
atomic number
How to work out number of neutrons
mass number - atomic number
How to tell which one is the mass number
it is always the biggest one
Which number determines what element an atom is
atomic number
Isotope
an atom with the same atomic number but a different mass number. they are the same element but are chemically slightly different as they have a different number of neutrons
How to write isotopes
‘element-mass number’ e.g. chorine-37
mass number in small above, atomic number in small below →
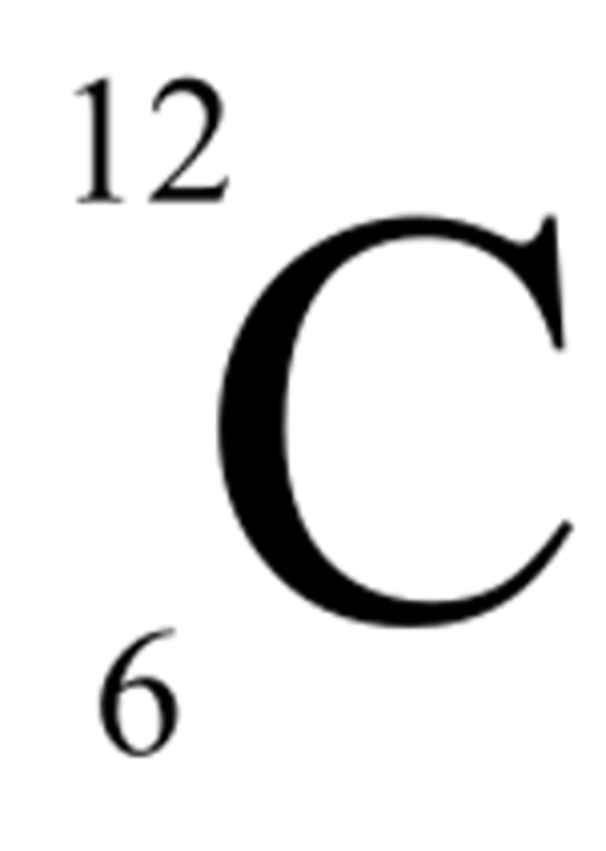
Relative atomic mass definition
weighted average of all an atom’s isotopes
Relative atomic mass equation
(% x mass number of isotope 1) + (% x mass number of isotope 2)
100
How many electrons can the first shell hold
2
How many electrons can the second shell hold
8
How many electrons can the third shell hold
8
How many electrons can the fourth shell hold
8
Writing electron configuration
number of electrons in each shell starting from the middle and going out e.g. 2, 8, 6
Atoms in the same group have…
the same number of electrons in the outer shell
Atoms in the same period have…
the same number of shells
How many electrons in their outer shell do metals have
1, 2 or 3
How many electrons in their outer shell do non-metals have
4, 5, 6 or 7
Staircase line
down from B, metals to the left & non-metals to the right
Transition metals
the middle section of un-grouped metals, the most common metals
Groups
vertical columns in the periodic table, numbered 1-7 with 0 on the end
Periods
horizontal rows in the periodic table, numbered 1-7
Bonds
the forces of attraction that hold atoms together
What happens when bonds are formed between atoms
energy is released, making the atoms more stable & less reactive
Which atoms are the most stable
the ones with a full outer shell as they contain as many electrons as possible (noble gasses)
How do atoms achieve a full outer shell
by transferring their electrons between each other
Ions
charged particles formed by transfer of electrons between atoms
What happens when metals form ions
they loose electrons and form positive ions (cations) which have more protons than electrons
What happens when non-metals form ions
they gain electrons and form negative ions (anions) which have more electrons than protons
What happens to non-metals’ names when they form ions
the end changes to -ide
Electrostatic forces
the forces of attraction between all positively and negatively charged objects, they hold oppositely charged ions together and form an ionic bond between them
Which atoms will easily form ions
ones that have a nearly full / nearly empty outer shell
What happens when group 1 elements form ions
they have 1 outer electron, so loose one to be come a positively charged ion, 1+
What happens when group 2 elements form ions
they have 2 outer electrons, so loose two to become a positively charged ion, 2+
What happens when group 6 elements form ions
they have 6 outer electrons so gain two to become a negatively charged ion, 2-
What happens when group 7 elements form ions
they have 7 outer electrons so gain one to become a negatively charged ion, 1-
Why is group one called alkali metals
they react with water to form alkalis
What are some physical properties of group 1
low melting points, soft and easily cut, very reactive, readily form compounds with non-metals
What happens when lithium reacts with water
it bubbles fiercely on the surface
makes lithium hydroxide and hydrogen gas
What happens when sodium reacts with water
it melts into a ball and fizzes about the surface
makes sodium hydroxide and hydrogen gas
What happens when potassium reacts with water
it bursts into lilac flame and flies about the surface
makes potassium hydroxide and hydrogen gas
Reactivity trend for group 1
as you go down the group, reactivity increases
Similarities in group 7
exist as diatomic molecules, are non-metals, are poor conductors of heat & electricity, all toxic & corrosive
Trend in melting/boiling point in group 7
as you go down the group, melting/boiling point increases
Trend in density in group 7
as you go down the group, density increases
What do group 7 elements form when they react with metals
salts
Why can group 7 elements be used as disinfectants/bleaches
they can kill microorganisms and remove colour from materials
Test for chlorine
put damp blue litmus paper in chlorine it will turn red and then bleach
What do group 7 elements form when reacting with hydrogen
hydrogen halides
What is produced when hydrogen halides dissolve in water
they form acids e.g. hydrochloric acid
Reactivity trend in group 7
reactivity decreases as you go down the group
Displacement reaction
reaction where a more reactive element takes the place of a less reactive element in a compound
Similarities in group 0
colourless, have low melting/boiling points, are poor conductors of heat/electricity, inert
Inert
doesn’t easily react with anything
Why are noble gasses inert
they have a full outer shell already
Melting/boiling points trend in group 0
decreases as you go down the group (but are all very low)
Uses of krypton
photography lighting because it produces a bright white light when electricity is passed through it