4.5 carboxylic acids and their derivatives
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
28 Terms
Acidity of carboxylic acids
They are weak acids and the extent of the ionization in aqueous Solutions is very small
they are acidic because of the hydrogen in the COOH group
carboxylic acids > phenols > water/alcohols

Test for relative difference in acidity
Can be shown by their reaction with sodium hydrogen carbonate solution
only carboxylic acids are strong enough to produce colorless bubbles of carbon dioxide gas
Formation of carboxylic acids by the oxidation of alcohols and aldehydes
Primary alcohols can be oxidized to aldehydes then further oxidised to carboxylic acids
acidified potassium dichromate is used as an oxidizing agent (turns from orange to green)
alkaline potassium manganate (VII) solution can also be used: turning from purple solution to a brown black sludge of manganese oxide

Volatility of aldehydes
Aldehydes and more volatile than the corresponding carboxylic acid and they should not be allowed to escape from the refluxing reaction mixture, to ensure that further oxidation proceeds to the carboxylic acid
heating under reflux: heating in a flask with a condenser placed vertically. This will prevent any aldehyde formed escaping before it has time to be oxidized to a carboxylic acid
Using excess oxidizing agents
To ensure that there is enough oxidizing agent present for the oxidation to go all the way to the carboxylic acid
when the oxidation is complete, the mixture can be distilled leaving an aqueous solution of an acid
Oxidation of secondary alcohols
Will form a ketone but any further oxidation to our carboxylic acid cannot occur this way
The reduction of carboxylic acids
Carboxylic acids are relatively stable compounds and their reduction to a primary alcohol requires the use of lithium tetrahydridoaluminate(III) ( LiAlH4)
reagent reacts violently with water and the reduction is carried out by using the solvent ethyoxethane

Sodium tetrahydridoborate
sodium tetrahydridoborate is a milder reducing agent and will reduce both aldehydes and ketones
but will not reduce a carboxylic acid

Make an aromatic carboxylic acid
Carboxylic acid group is directly bonded to the benzene ring
can be oxidized with acidified potassium dichromate or manganate to produce a carboxylic acid and water

Oxidizing a methyl side chain to a COOH
Heat the compound with alkaline potassium manganate solution
initial product is the sodium/potassium salt of benzene carboxylic acid
reaction mixture is then acidified producing the acid itself. Another product of the reduction is a brown black sludge of manganese (IV) oxide
benzene carboxylic acid is the end product of this oxidation, irrespective of the length of carbon side chain
Decarboxylation
The removal of carbon dioxide from a compound
usually uses soda lime
soda lime is largely calcium hydroxide with a little sodium/potassium hydroxide
The material absorbs both carbon dioxide and water but remains as a solid
Ca(OH)2 + CO2 — CaCO3 + H2O
Decarboxylation of carboxylic acids
As carbon dioxide is an acidic oxide it will react with a base.
In a decarboxylation reaction, organic acid or its sodium salt is heated with soda lime
a hydrocarbon is produced that contains ONE LESS carbon atom in a chain than the starting material
Decarboxylation occurs
If the calcium salt of the acid is heated in the absence of soda lime
a ketone is produced
in this reaction the chain length INCREASES
Salts of dicarboxylic acids
Decarboxylation reactions will occur if these salts are heated with soda lime. EG can form propane
substituted aromatic carboxylic acids can also be decarboxylated by heating
Making esters from carboxylic acids and their hydrolysis
Carboxylic acids can be converted to esters by heating the carboxylic acid with an alcohol in the presence of little sulfuric acid
This initially protonates the carboxylic
Esterification
Reversible reaction and the hydrolysis of esters can be carried under basic or acidic conditions
in hydrolysis, using basic conditions the ester is heated to reflux with an aqueous sodium hydroxide and the mixture is then acidified to produce carboxylic acid
The rates of hydrolysis
Depends on the Ester and the concentration of alkali used
soap making is a process of the hydrolysis of esters of glycerol
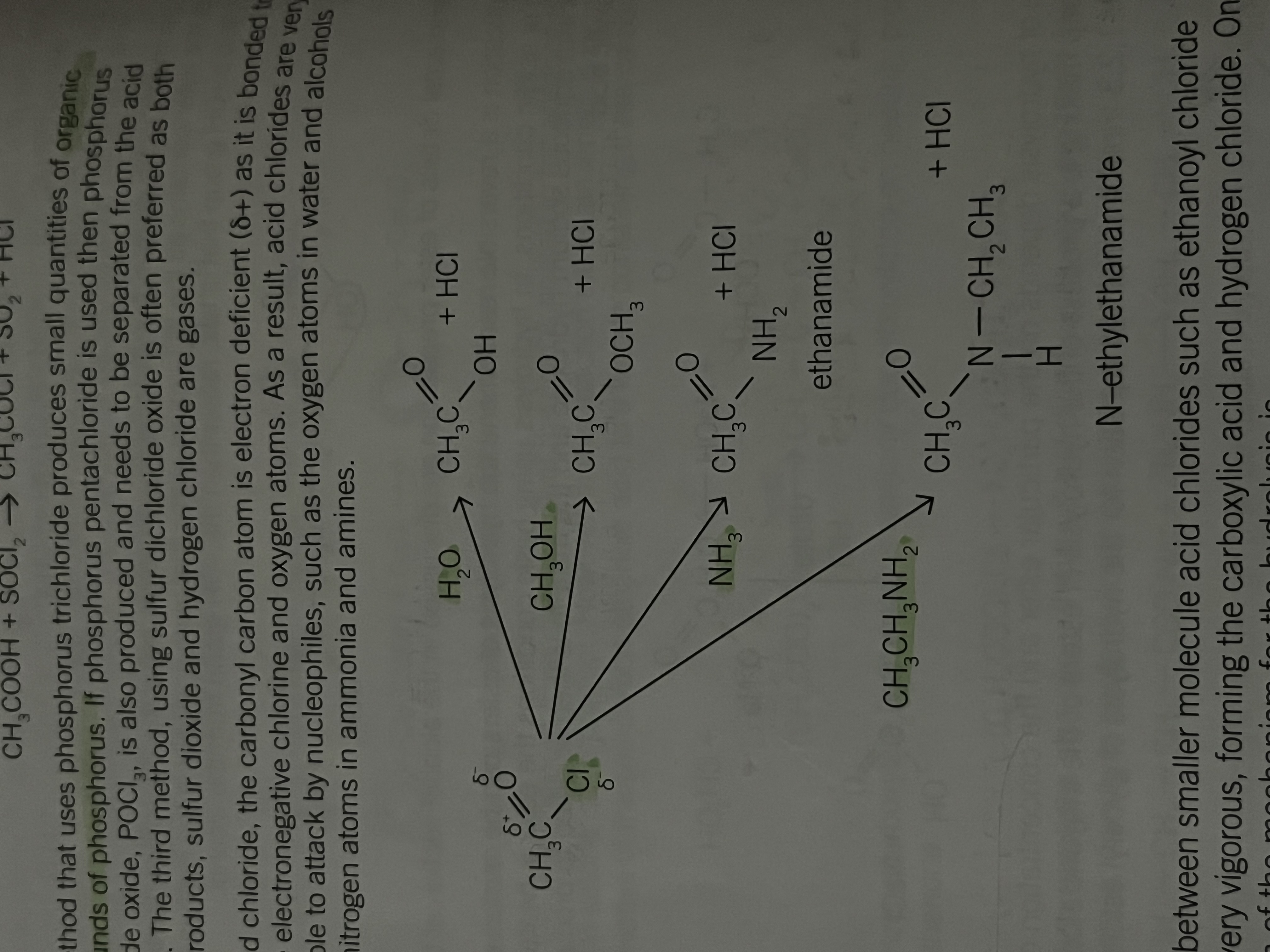
Making acid chloride from carboxylic acids and the hydrolysis of these compounds
To make an acid chloride from a carboxylic acid, the OH group of the acid needs to be substituted by a chlorine atom
acid chloride are reactive compounds and are easily hydrolysed if water is present. Method should not contain water
the reagents used are phosphorus trichloride phosphorus pentacloride and sulphur dichloride oxide SOCl2

The three methods to make acid chloride
Phosphorus trichloride produces small quantities of organic compounds of phosphorus
phosphorus pentechloride produces phosphorus trichloride oxide POCl3 and needs to be separated from the acid chloride
Salford dichloride oxide is often preferred as both the co-products sulphur dioxide and hydrogen chloride are gases
In an acid chloride
The carbonyl carbon atom is electron deficient and is bonded to more electronegative chlorine oxygen atoms
As a result acid chlorides are very susceptible to attack by nucleophiles such as the oxygen atoms in water/alcohols and nitrogen atoms in ammonia/amines
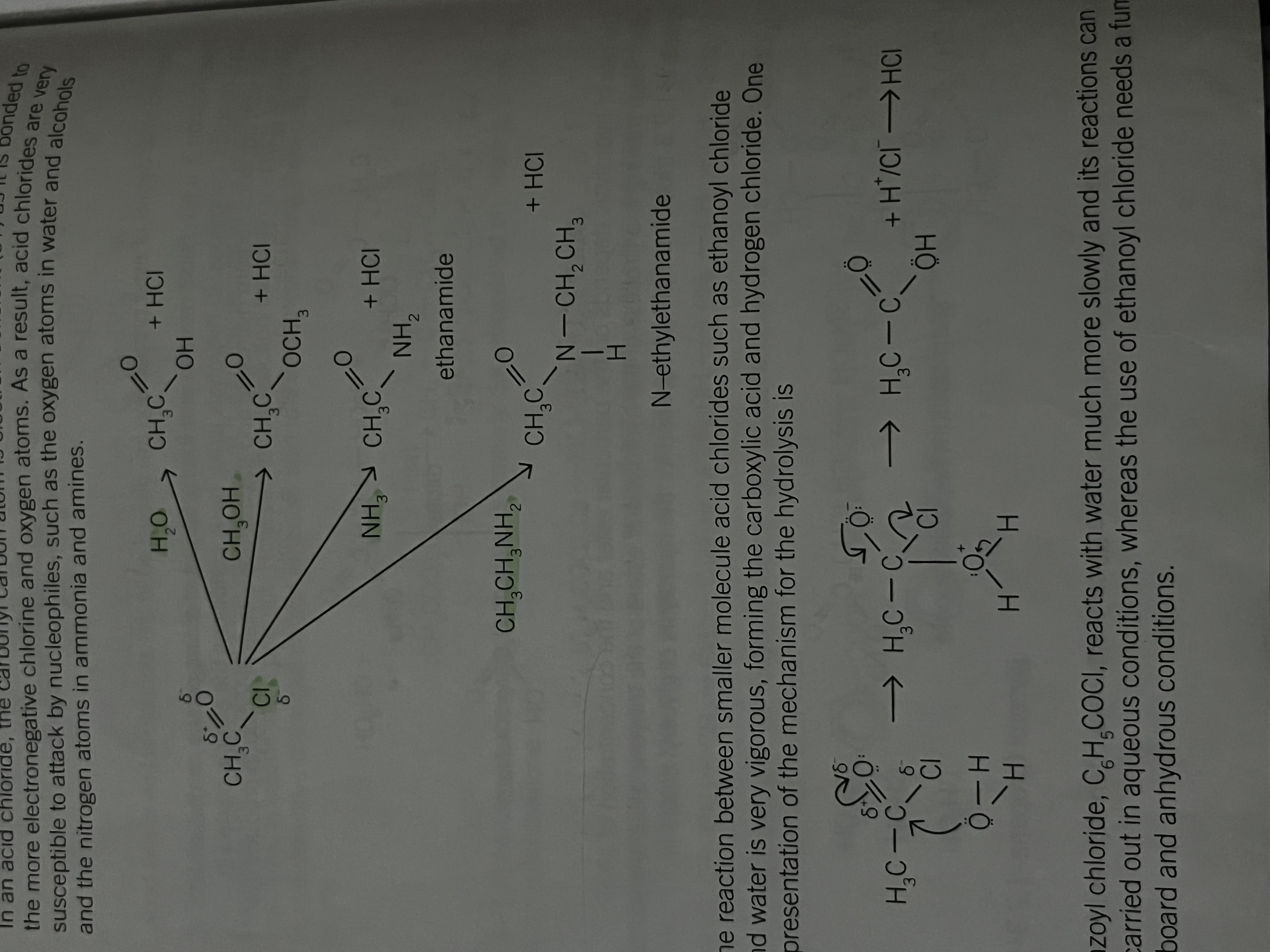
The reaction between smaller molecule acid chlorides and water
Eg. Ethanoyl chloride
is very vigorous forming carboxylic acid and hydrogen chloride
benzoyl chloride C6H5COCl reacts with water much more slowly and it's reactions can be carried out in aqueous conditions
Whereas the use of ethanoyl chloride needs a fume cupboard and anhydrous conditions
Amides and nitriles from carboxylic acid
Carboxylic acids such as ethanoic acid, reacts with ammonia to give the ammonium salt of the acid
When this salt is heated water is lost and the amideis produced
a better method is to heat the acid or it's ammonium salt with urea at 120 degrees

Formation of nitriles from halogenoalkanes
Reaction with potassium cyanide using an alcohol water mixture as a solvent
a nucleophilic substitution reaction where the Cyanide ion act as a nucleophile
chlorobenzene will not react with cyanide as the ring carbon atoms are not susceptible to nucleophilic attack

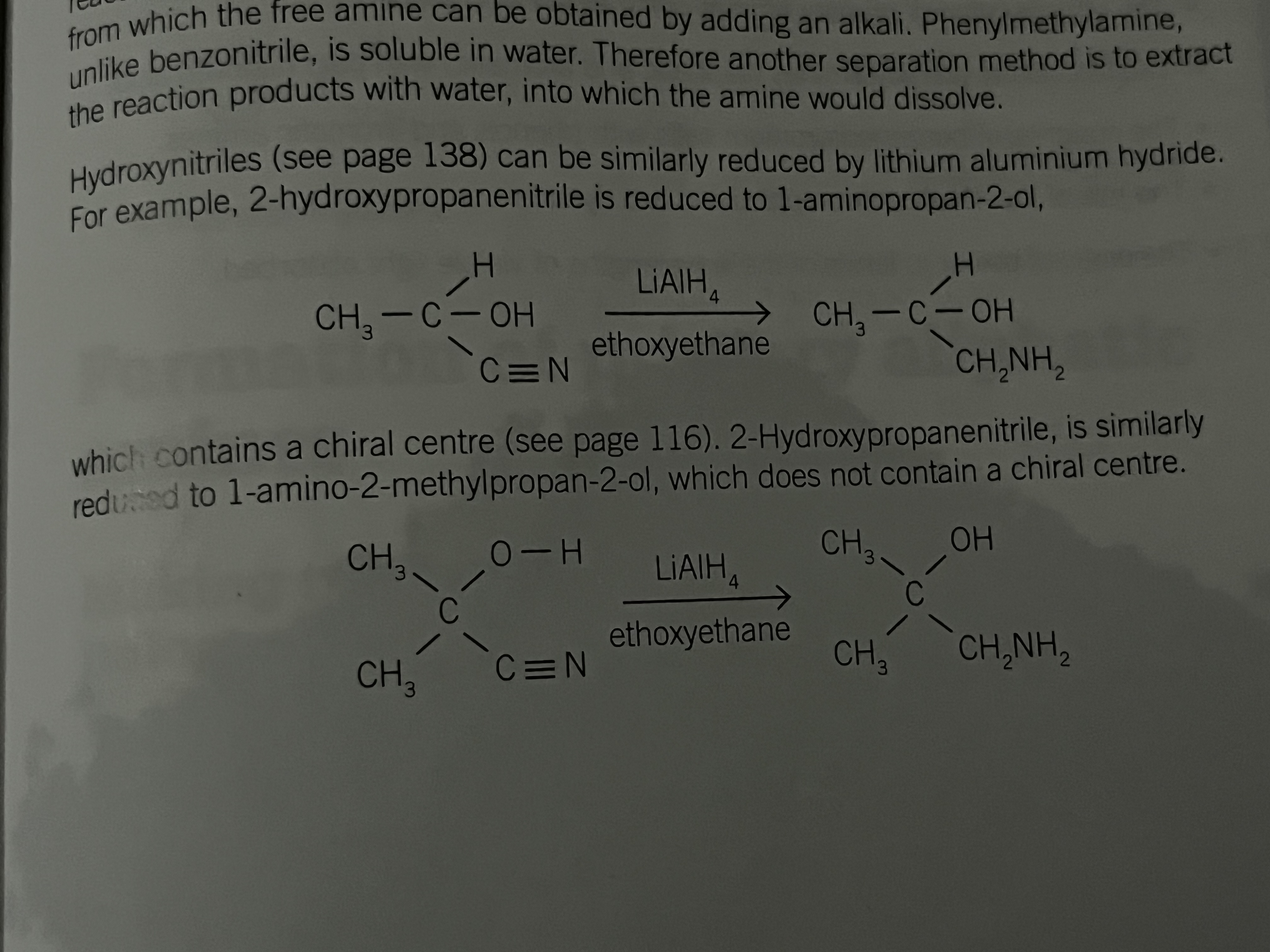
Formation of hydroxynitriles from aldehydes and ketones
An aldehyde and some simple ketones will react with hydrogen cyanide in the presence of sodium or potassium cyanide to produce a hydroxynitrile
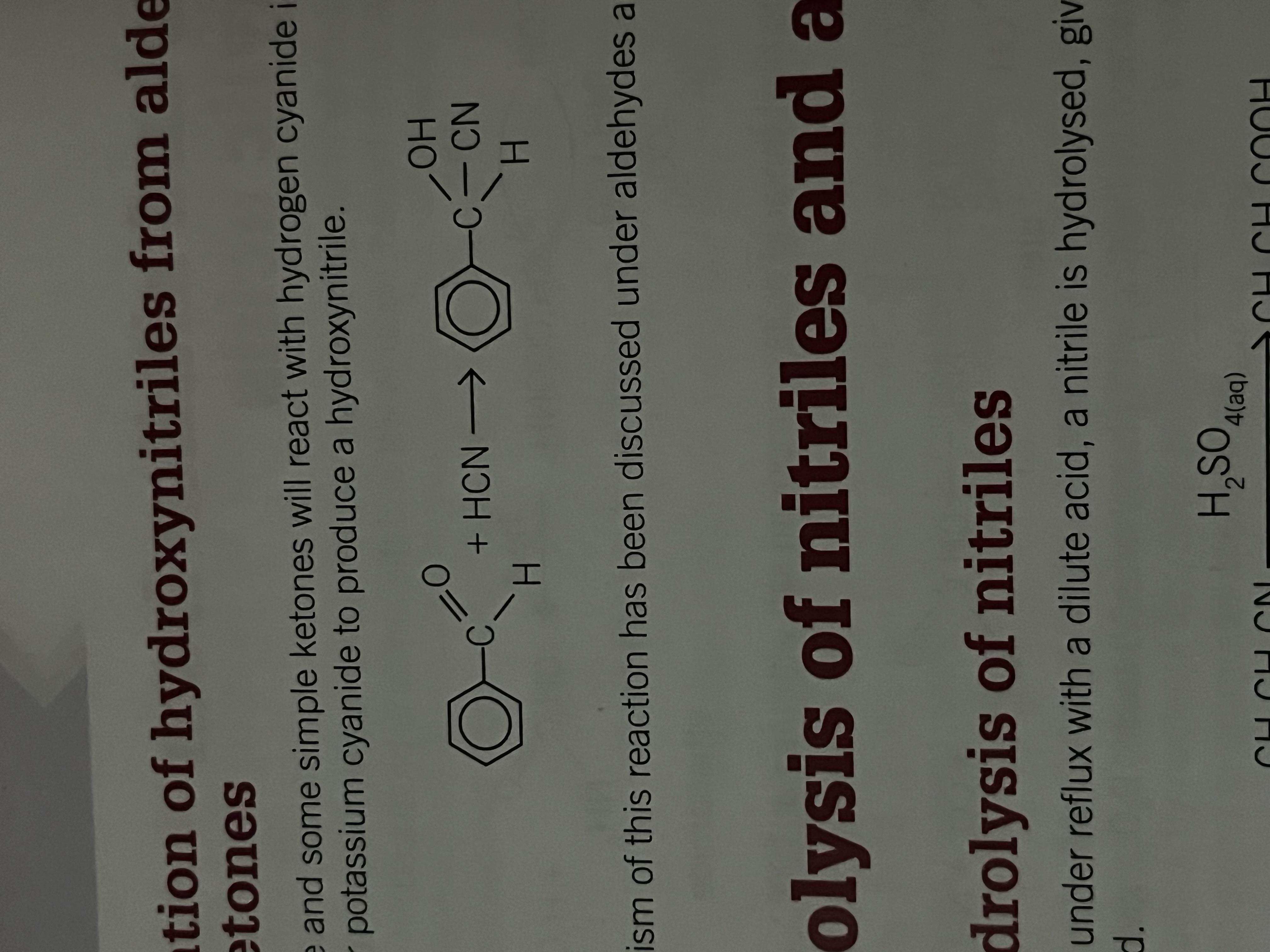
The hydrolysis of nitriles
When heated under reflux with a dilute acid, a nitrile is hydrolized giving a carboxylic acid
the nitrogen of the cyanide group becomes an ammonium group. If aqueous sulfuric acid is used, the nitrogen containing product is a solution of ammonium sulfate.
The hydrolysis can also be carried out by heating the night trial under reflux with an alkali Eg. NaOH giving the anion of the carboxylic acid.
Acidification of the mixture produces the carboxylic acid

Hydrolysis of amides
Where did amide is heated under reflux with a base (NaOH) the amide is hydrolised as the carbon to nitrogen bond is broken, producing ammonia gas

Reduction of nitriles
Warming a nitrile with a solution of lithium tetrahydridoaluminate in ethoxyethane will reduce a nitrile to primary amine. This method is preferred because the yield is higher.
Are there reducing agents include hydrogen and a nickel catalyst and sodium metal/ethanol. Can produce a secondary a mean as a by-product
formation of a nitrile produces compounds with longer carbon chains.

The boiling temperature of benzonitrile and phenylmethyl amine
Similar and separation of the products by distillation isn't possible
primary amines are bases and if hydrochloric acid is added to the reaction mixture phenylmethyl amine will react with the acid to form a water soluble salt
from which the free amiine can be obtained by adding an alkali
phenylmethylamine is soluble in water
adding another separation method is to extract the reaction products with water, into which the amine would dissolve