Galvanic cells
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
how is standard cell potential (E cell) measured
measured at a specified temperature with no current flowing and all components in their standard states
standard electrode potential (E°half-cell)
potential of a given half-reaction when all components are in their standard states. {At SATP and 1mol/L concentration for aqueous solutions}
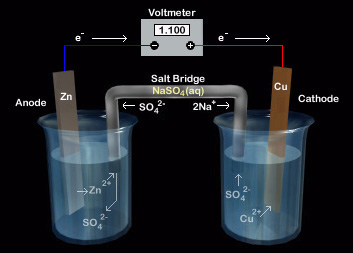
notation for a voltaic cell
Anode | Anode Solution || Cathode Solution | Cathode
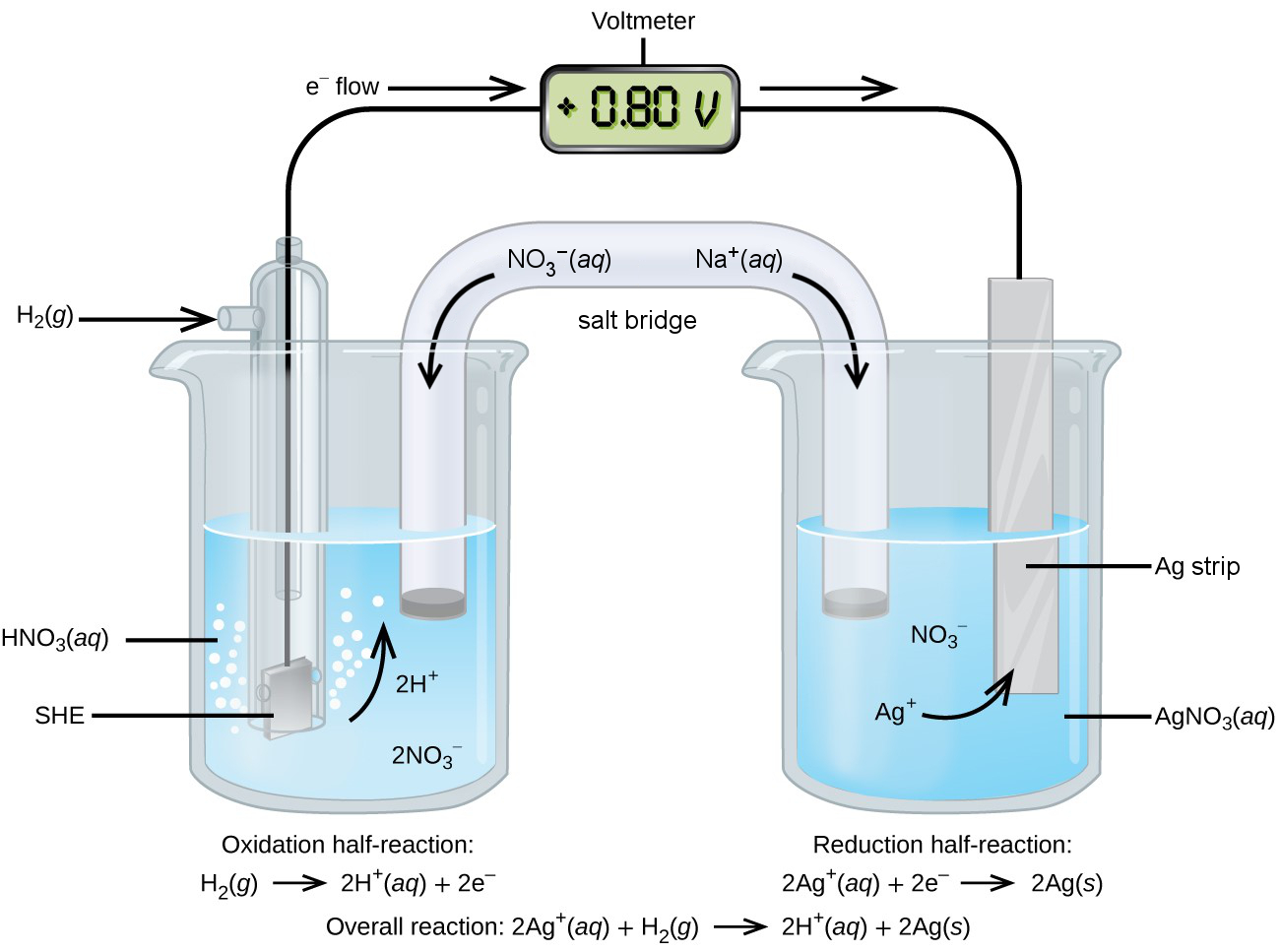
inactive electrode
to provide a surface for electrons to move without the metal itself getting involved in the chemistry
active electrode
active component in half-cell; a reactant/product in overall reaction
anode
(an, ox) NEGATIVE (-) electrode in voltaic (galvanic) cell. Polarized current enters circuit
cathode
(red cat) POSITIVE (+) electrode in galvanic cell. Current leaves a polarized electrical device
salt bridge
contains non-reacting cations & anions, often K+ & NO3- that don’t interfere w reaction
purpose of salt bridge
maintain electrical neutrality; keep charges in electrolytes constant → allowing the redox reaction to continue
why is a salt bridge needed
e- must keep moving for electricity to flow. Without salt bridge, electrolyte gets more positively/negatively charged, thus slows oxid/reduc cuz charged ions start repelling each other. Salt bridge helps ion freely between between half-cells
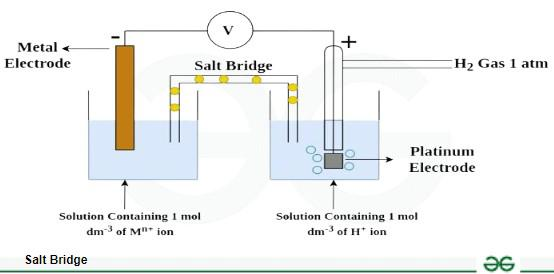
why does salt bridge deplete
the ions within them migrate into the half-cells to neutralize accumulating charges, eventually diffusing out. But the salt bridge is made not just of salt, but of an absorbent material such as filter paper. This will soak up the electrolytes it is dipped into, so ions from the solutions will diffuse into it