Messengers and Receptors 1, 2 and 3
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
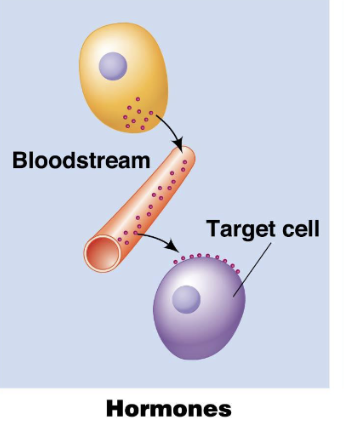
Endocrine Signals
Signaling where hormones are released into the bloodstream to reach distant target cells.
long distance signalling
use hormones as signalling molecules (insulin, cortisol)
Insulin released from the pancreas regulates blood sugar levels in distant tissues.
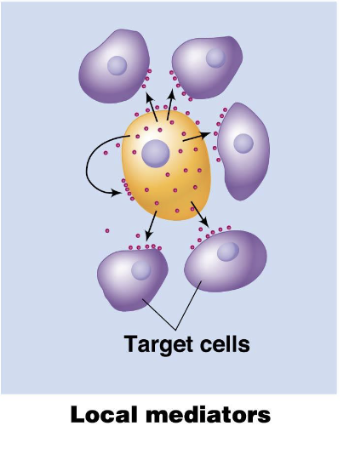
Paracrine Signals
Signaling where molecules act on nearby cells without entering the bloodstream.
short distance signalling
local mediators as signalling molecules (growth factors, cytokines)
growth factors released by a cell stimulate nearby cells to divide
if you have a scab, this signal sends a signal to grow new skin
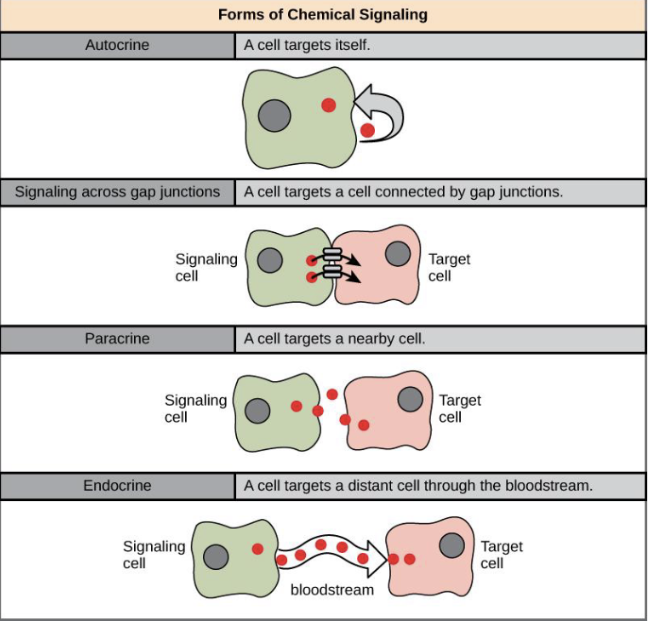
Juxtacrine Signals
Signaling where molecules directly interact with adjacent cells through cell-to-cell contact.
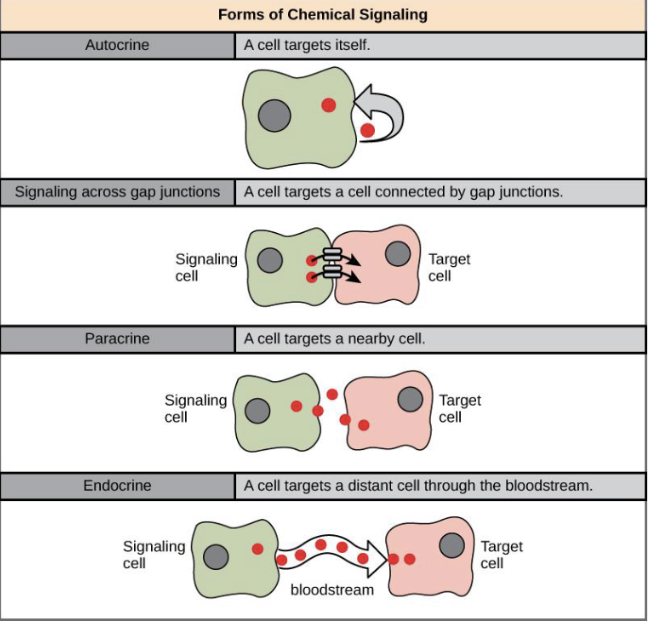
Autocrine signals
Cell targets itself, acts on same cell that produces them
how are signals perceived or detected by cells
through receptors and ligands
messenger reaches target, binds to receptors
messenger itself, that binds a target receptor is a ligand
Receptors
Proteins that bind specific ligands, triggering a cellular response.
Ligands
Molecules that bind to receptors, initiating a signal transduction pathway.
Coreceptors
Additional proteins that assist in the binding of ligands to primary receptors, enhancing the cellular response.
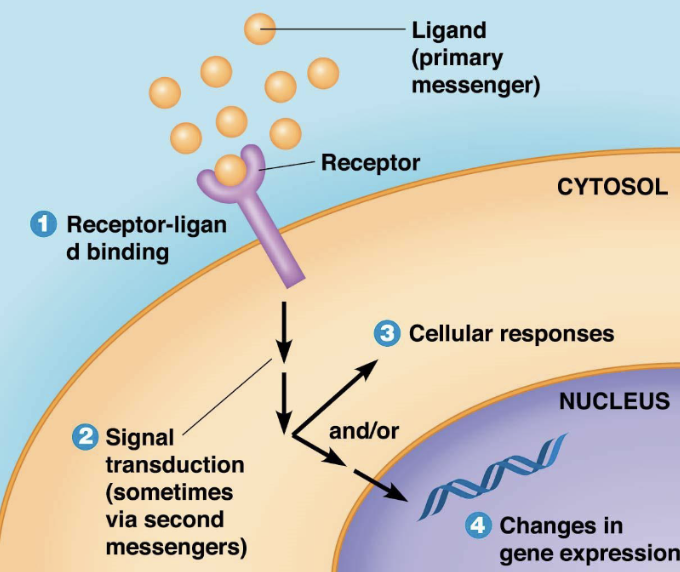
What is signal transduction?
ability for cell to respond to ligand-receptor binding by altering its behaviour or gene expression
what are the steps of signal transduction?
Reception - where a ligand binds a receptor
Transduction - molecular interactions relay signals inside cell, often involving secondary messengers such as cAMP, Ca2+
Response OR Changes in gene expression
What happens when ligand concentration increases?
more ligands bind the receptors until saturation is reached
what happens when saturation point is reached?
The receptors are fully occupied, and further increases in ligand concentration do not produce a change in response.
what is receptor affinity?
strength of binding between a receptor and its ligand: higher affinity = receptor binds even at low ligand concentrations, lower affinity = requires higher concentrations to bind.
what are the 2 ways cells can shut down signalling?
reducing ligand amount - lowering amount of signalling molecules (ligand) reduces signal since there’s less ligand available to bind to receptor
reducing receptor sensitivity or number - receptor becoming less responsive or fewer receptors available, causes the signal t become weaker even if ligand is present
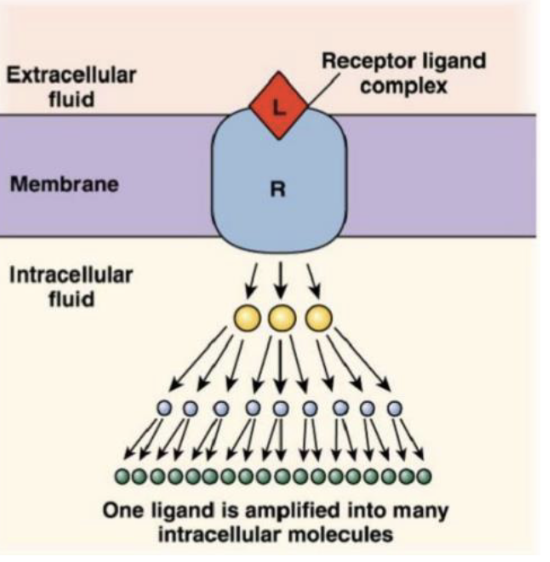
how are signals amplified by cells?
small amount of ligands can trigger a large response
at each step of signalling process, one molecule can activate many more molecules in a cascade effect, amplifying the signal.
this is called signal amplification
epinephrine binds to receptors on liver cells, triggers breakdown of glycogen into glucose
single epinephrine molecules triggers release of millions of glucose molecules, which shows how one ligand can create a massive response through amplification
Hydrophilic ligands
ligand-gated ion channels
G protein-coupled receptor (GPCR)
Receptor Kinase
Hydrophobic ligands
nuclear receptor
G Protein Coupled Receptors (GPCRs)
type of receptor that when a ligand binds to them, it changes shape and activates a G protein inside cell
once G protein is activated, it binds to target protein in cell, altering its activity
Opioid receptors, to which drugs like morphine bind and trigger effects in the body.
G protein
guanine-nucleotide binding protein
once activated, binds to target protein in cell, altering its activity
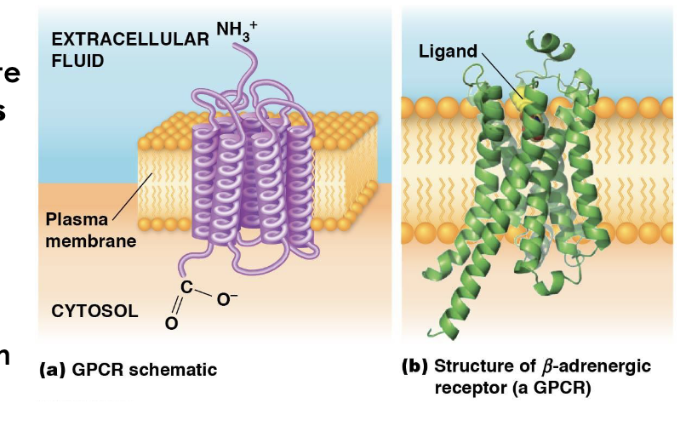
Structure and Regulation of GPCRs
have similar shape and structures, as well as functions, but different amino acid sequences
consisting of seven α-helical transmembrane regions (loops) that go through the cell membrane.
the extracellular portion of receptor has a unique site where ligand binds
heterotrimeric G proteins
consist of three subunits: Gα, Gβ, and Gγ.
stimulatory G proteins (Gs)
activate signalling pathways
inhibitory G proteins (Gi)
inhibit signalling pathways
What determines whether a G protein is "on" or "off"?
G proteins are "on" when bound to GTP and "off" when bound to GDP.
2 classes of G proteins?
small monomeric G proteins
Heterotrimeric G proteins (Ga, Gb, Gy)
what is the function of Ga subunit?
binds to GTP (active) or GDP (inactive) to regulate signalling
What happens when a ligand binds to a G protein-coupled receptor (GPCR)?
The receptor changes shape, allowing it to interact with a G protein and cause GDP release.
What does the Gα subunit do after binding GTP?
It detaches from the Gβγ subunits and may activate a signaling pathway.
What are the two possible signaling initiators in G protein activation?
Either the Gα subunit or the Gβγ subunits can initiate signal transduction, depending on the type of G protein.
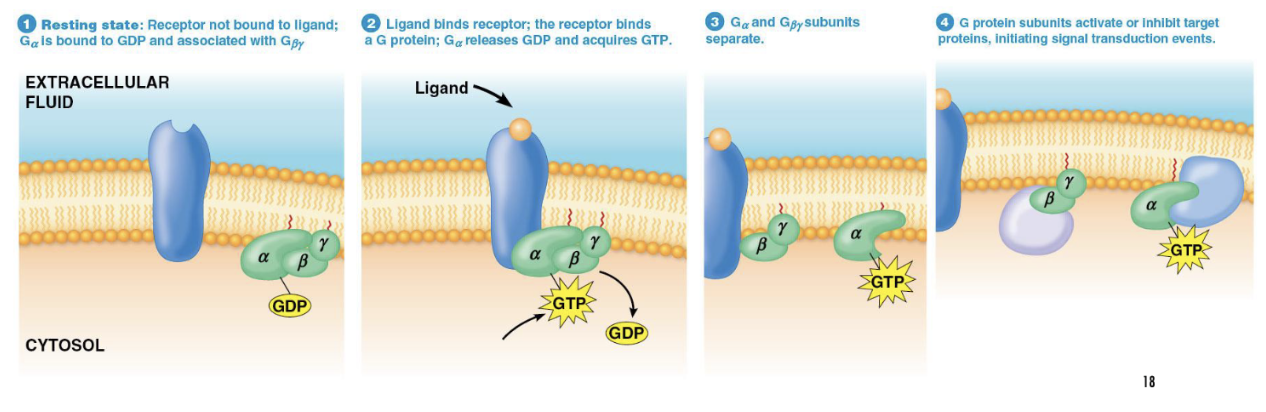
What does GTP binding cause in a G protein?
It activates the Gα subunit, causing it to separate from Gβγ and trigger a signaling pathway.
What is the role of GDP in G protein activation?
GDP keeps the G protein inactive; it must be replaced by GTP to activate the protein.
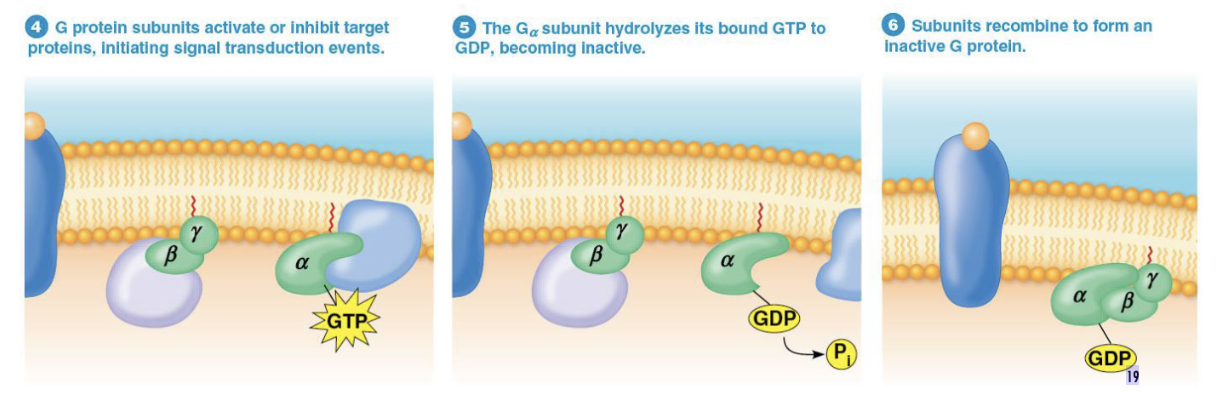
What happens when Gα subunit hydrolyzes GTP to GDP
reassoicates with Gβγ, which causes signal transduction pathway to shutdown once the messenger (ligand, signalling molecule) is no longer present
returns G protein to its inactive state
How is the process of GTP hydrolysis sped up?
the process is sped up by RGS proteins (regulator of G protein signalling proteins), such as GAP proteins that increase the hydrolysis rate, thus returning the G protein to its inactive state more quickly.
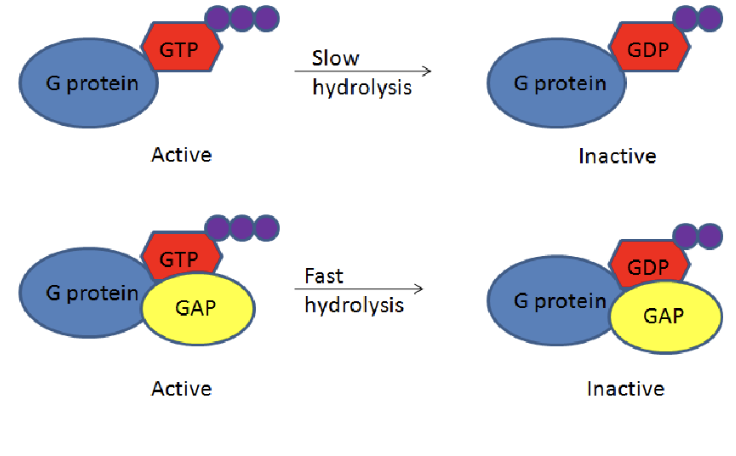
What are RGS (Regulator of G protein Signaling)?
RGS proteins are molecules that regulate the signaling activity of G proteins by accelerating the hydrolysis of GTP to GDP, thereby facilitating a quicker return to the inactive state.
simply, it aids in the quicker deactivation of G proteins
What do the α subunits of activated G proteins interact with?
Adenylyl cyclase and phospholipase C.
Which subunit of a dissociated G protein can activate G protein receptor kinases?
The βγ subunit
What is the most important function of G proteins?
The release or formation of second messengers.
what is cAMP (Cyclic AMP)?
a secondary messenger that is produced when Gsa subunit binds to the enzyme, adenylyl cyclase.
activation of G proteins is short lived, so they quickly stop producing cAMP when Gsa becomes inactive
this leads to cAMPs already made, to be broken down by an enzyme called phosphodiesterase.
How is adenylyl cyclase activated?
Adenylyl cyclase is activated when it binds to the activated Gsα subunit.
What does GTP-Gsα do?
GTP-Gsα activates adenylyl cyclase to produce cAMP.
What happens when Gsα becomes inactive?
Adenylyl cyclase stops making cAMP.
How is cAMP removed from the cell?
cAMP is broken down by the enzyme phosphodiesterase.
What is the main target of cAMP?
PKA (protein kinase A)
How does cAMP activate PKA?
by separating the regulatory and catalytic subunits of PKA.
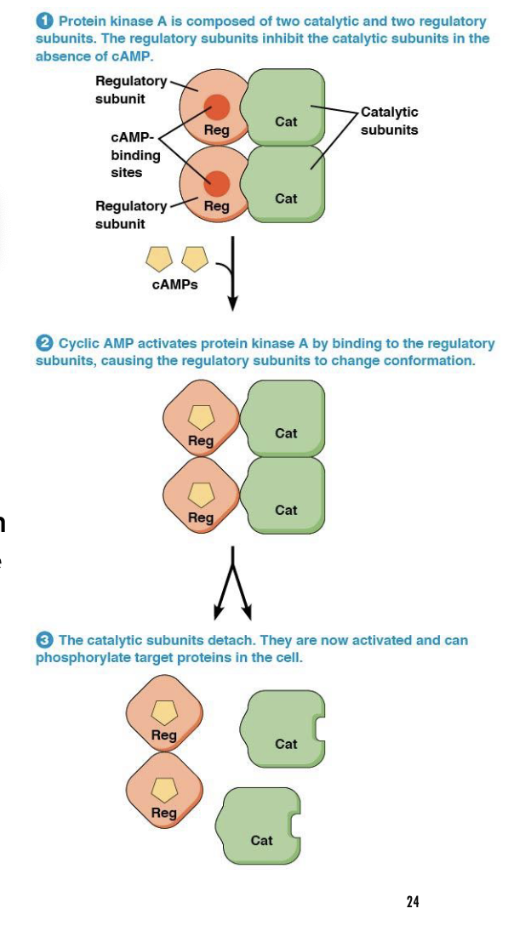
What does PKA do once activated?
PKA phosphorylates (adds phosphate groups) proteins on serine or threonine residues, using ATP as the source of phosphate.
Grab A Cold Pepsi
G protein
Adenylyl Cyclcase
cAMP
Protein Kinase A
What enzyme does the activated G protein stimulate?
Phospholipase C (PLC).
What two molecules does PLC produce from PIP₂?
IP₃ (inositol trisphosphate) and DAG (diacylglycerol).
What does IP₃ do after being produced?
It binds to an IP₃ receptor on the endoplasmic reticulum, causing calcium release into the cytosol.
What does DAG do in the cell membrane?
It helps activate Protein Kinase C (PKC).
What two molecules are needed to activate Protein Kinase C (PKC)?
Calcium (Ca²⁺) and DAG.
What does PKC do once activated?
It phosphorylates proteins on serine and threonine residues, leading to various cellular responses.
What role does Ca²⁺ play in cell signaling?
Calcium regulates many cellular functions by binding to proteins and changing their activity.
How do cells keep calcium levels low?
By using Calcium ATPases to pump calcium out of the cell or into the ER for storage.
What enzyme helps trigger calcium release from the ER?
Phospholipase C (PLC).
What protein mediates calcium-activated processes?
Calmodulin.
How does calmodulin work?
It binds four calcium ions (two per “hand”), changes shape, and activates other proteins by wrapping around them.
What happens when calmodulin binds to a protein?
It changes the protein’s activity, which can affect cellular functions, including those related to neuropsychiatric diseases.
What do protein kinase-associated receptors do?
They are receptors that also act as kinases, activating signals inside the cell.
What happens when a ligand binds to a kinase-associated receptor?
It activates the kinase, starting a phosphorylation cascade.
What are the two main types of kinases?
Tyrosine kinases and serine/threonine kinases.
What are growth factors?
Platelet-Derived Growth Factor, found in blood serum after clotting.
What kind of receptor does PDGF bind to?
A receptor tyrosine kinase.
What is a receptor tyrosine kinase (RTK)?
RTKs are receptors on the cell surface that function as kinases and are involved in signal transduction for processes like cell growth and specialization.
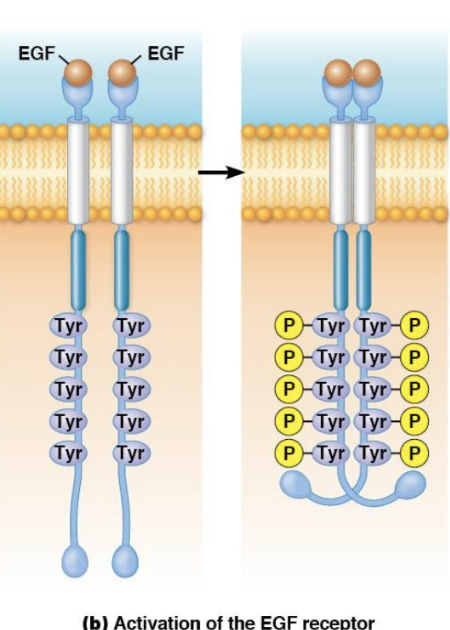
How is the receptor tyrosine kinase activated?
Ligand binding leads to dimerization or receptor clustering, resulting in autophosphorylation (self-phosphorylation).
What is autophosphorylation in receptor tyrosine kinases?
Autophosphorylation is when receptors phosphorylate themselves on tyrosine residues after activation by ligand binding.
What is the function of the SH2 domain in the RTK signaling pathway?
The SH2 domain binds to phosphotyrosine residues on the activated receptor, helping to propagate the signal inside the cell.
What is the role of Ras in receptor tyrosine kinase signaling?
is a small G-protein that, when activated by GTP, regulates cell growth and proliferation.
What is Sos in the context of Ras activation?
Sos is a guanine-nucleotide exchange factor (GEF) that helps Ras acquire GTP, activating it in the signaling cascade
What protein binds to the receptor to recruit Sos in Ras signaling?
Grb2 binds to the receptor and recruits Sos.
What does Sos do in Ras signaling?
Sos activates Ras by promoting the exchange of GDP for GTP.
What happens after Ras is activated in signaling?
Ras activates Raf, which activates MEK, leading to the activation of MAPK proteins.
What do MAPKs do once activated in Ras signaling?
MAPKs enter the nucleus to regulate gene expression and control cell growth and division.
How is Ras inactivated?
Ras is inactivated when GTPase-activating proteins (GAPs) promote the hydrolysis of GTP to GDP, making Rasinactive.
Go South Right Rounda Meat Market
GRB2
Sos
Ras
Raf
MEK
MAPK
What is a dominant negative mutation in RTK signaling?
A mutation where a defective receptor binds with normal receptors and prevents their activation.
What is a constitutively active mutation?
A mutation that keeps the receptor always active, even without ligand binding.
How do scientists study RTK signaling?
By mutating receptors or signaling proteins and observing their effects.
What type of kinases phosphorylate serine and threonine residues?
Serine/Threonine Kinases.
What major signaling pathway uses Serine/Threonine Kinases?
The TGFβ (Transforming Growth Factor Beta) pathway.
What happens when TGFβ binds to its receptor?
Type II receptors phosphorylate Type I receptors, which activate a signaling cascade.
What proteins are phosphorylated by Type I receptors in the TGFβ pathway?
R-Smads
What is the role of Co-Smad?
It forms a complex with phosphorylated R-Smads and enters the nucleus to regulate genes.
What are the three types of Smads?
R-Smads (Receptor-Regulated Smads) – Phosphorylated by Type I receptors.
Co-Smad (Smad4) – Partners with R-Smads for nuclear entry.
I-Smads (Inhibitory Smads, Smad6/Smad7) – Block signaling.
What is the role of scaffold proteins in cell signaling?
They assemble multiprotein complexes, making signaling more efficient and specific.
What is signal crosstalk?
When activated components from one signaling pathway influence another pathway, allowing for integration of multiple signals.
How do cells integrate different signaling pathways?
through scaffold proteins, signal crosstalk, and common second messengers (e.g., IP3, calcium, cAMP).
What are second messengers in signal transduction?
Small molecules like IP3, DAG, cAMP, and calcium ions that help amplify and distribute signals inside the cell.
How does phosphorylation contribute to signal integration?
Many pathways lead to the phosphorylation of key proteins, which then regulate gene expression, metabolism, and cell division.
Do all cell signaling pathways occur locally?
No, some signals, like hormones, travel long distances through the bloodstream to reach target cells.
What is the term for hormones that travel through the circulatory system (bloodstream)?
Endocrine hormones.
Where are endocrine hormones synthesized and secreted?
In endocrine tissues, such as the pituitary gland, adrenal glands, and pancreas.
How do hormones find their target tissues?
Hormones circulate in the bloodstream and bind to specific receptors on target cells.
Types of Endocrine Hormones
amino acid derivatives
peptides
proteins
steroid
What are two examples of adrenergic hormones?
Epinephrine (adrenaline) and norepinephrine.
What is the function of adrenergic hormones?
They prepare the body for fight-or-flight by increasing heart rate, breaking down glycogen, and redirecting blood to muscles.
Where are adrenergic hormones produced?
In the adrenal glands.
What are the two main types of adrenergic receptors?
α-Adrenergic receptors (vasoconstriction) and β-Adrenergic receptors (vasodilation)
What type of G proteins do α-adrenergic receptors activate?
Gq proteins.
What type of G proteins do β-adrenergic receptors activate?
Gs proteins, which stimulate the cAMP pathway to relax smooth muscles.