Chem 1A Midterm 2- Energy
1/156
Earn XP
Description and Tags
Haven't done lesson 21 yet but will get there
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
157 Terms
Law of conservation of mass
Mass (atoms/matter) can’t be created or destroyed
In a balanced chemical equation, there should be (same/different) number of each element on either side
Same
When balancing a chemical equation, what part do you change to balance?
Stoichiometric coefficient
What are tips for balancing a chemical equation?
Start with balancing whatever is in the fewest formulas
Balance polyatomic ions as a single element
What is the limiting reactant?
The reactant that runs out first, stopping the reaction and limiting how much product is made
What is the opposite of the limiting reactant?
Excess
T/F: You cannot have fractional stoichiometric coefficients
False- you can, since the coefficients represent ratios and not the whole amount of each element
How do you find the limiting reactant?
Use unit conversion to find how much product can be made with either reactant, whichever makes the LEAST amount of product is the limiting reactant
What is heat capacity?
(C) how much energy is needed to raise temperature
What unit is temperature in, in heat capacity?
J/mol
What is specific heat?
(c) heat capacity per g or mol
What unit is specific heat in?
J/ g C
or
J/ mol C
(Heat capacity/specific heat) is impacted by volume
Heat capacity is impacted by volume
Specific heat is not
What is a factor in specific heat, in the case of liquids and solids?
Bonding strength/ IMFs
What are degrees of freedom?
Ways for a molecule to move when influenced by heat energy
What are the ways something can move, according to degrees of freedom?
Rotations
Vibrations
Translations
Which kind of movement increases temperature?
Translations ONLY
If something rotates or vibrates, it has a higher ____
Specific heat
More degrees of freedom means a (higher/lower) final temp
More degrees of freedom means a lower final temp
If something moves more, it will be (more/less) hot
If something moves more, it will be less hot
If something moves less, it will be (more/less) hot
If something moves less, it will be more hot
Since most of its energy can go into translational movement, which would increase the T
(More/less) bonds means more vibrations
More bonds means more vibrations
What kind of bonds limit rotations?
Linear bonds
(Weaker/stronger) IMFs make a molecule easier to move, resulting in (low/high) heat capacity and (low/high) temperature
Weaker IMFs make a molecule easier to move, resulting in low heat capacity and high temperature
What are the equations for heat input/output?
q = mcΔT or q = CΔT
What do the variables in q = mcΔT and q = CΔT stand for?
q - heat input or output
m - mass in mol
c - specific heat
C - heat capacity
ΔT - change in temperature (final - initial)
In calorimetry, what two values are always the same between the surroundings and object?
Final temperature
In calorimetry, how is q (heat input/output) related to either object?
The q of one object is the inverse of the other; or in other words
q (surroundings) = - (q object)
Because the amount of heat one object loses is the same amount as what the other gains
What is the q of an exothermic reaction?
q < 0
Which way does energy flow in an exothermic reaction?
Energy exits the system
How does an exothermic reaction feel?
Hot!
Which way do KE and PE flow in an exothermic reaction?
High → low
What is the q of an endothermic reaction?
q > 0
What is the flow of energy in an endothermic reaction?
Energy ENTERS system
How does an endothermic reaction feel?
Cold!
What is the flow of KE and PE in an endothermic reaction?
Low → high
Which reaction has an energy input?
Endothermic
Which reaction has an energy output?
Exothermic
What is sublimation?
Phase change from solid to gas
What does potential energy (at least in this class) refer to?
How close particles are to one another
What sign is attraction?
Negative
What sign is repelling?
Positive
A small radius/close together particles indicate a (weak/strong) attraction and (very/less) negative PE
A small radius/close together particles indicate a strong attraction and very negative PE
A large radius/far apart particles indicate a (weak/strong) attraction and (very/less) negative PE
A large radius/far apart particles indicate a weak attraction and less negative PE
On a phase change graph, what does an angled slope indicate?
KE change, temperature change
On a phase change graph, what does a constant slope indicate?
PE change, phase change
Explain why PE change does not change temperature
Temperature will increase until a phase change, when another particle movement overtakes translation and causes temp to be constant during phase changes
What is enthalpy?
Heat, ΔH
T/F: ΔH of opposite phase changes are the same integer
F: ΔH of opposite phase changes are opposite integers
ex) ΔH(vaporization) = -ΔH(condensation)
When looking for which system has lower PE, what can you look for?
Phase
From highest to lowest PE: gas, liquid, solid
Bond #
More bonds = more attractions = negative PE
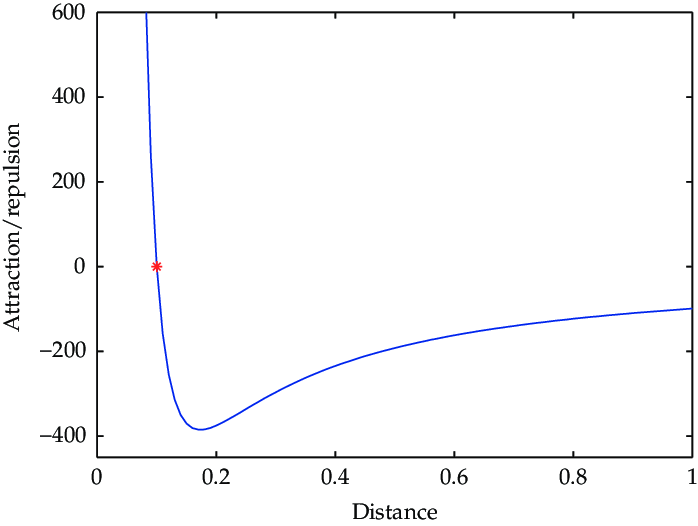
Describe what is happening in this image, and how it relates to attraction/repulsion
Read the graph from right to left
1) The distance (radius) decreases, and not much attraction is “felt” because particles are too far apart
2) The potential energy drops suddenly (indicating strong attraction!) when the particles are at optimal distance
3) As the radius decreases further, repulsion force dominates attraction force, and potential energy increases (strong repulsion)
4) Repulsion force will cause radius to increase to an optimal distance
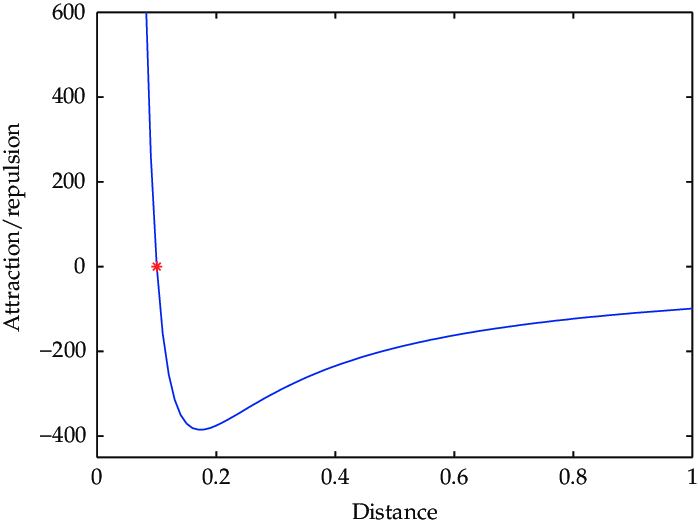
Where on the graph do particles “want” to be?
Particles want to be at the lowest PE possible, at a distance that is not too close or far
What is optimal bond length?
Just the right length of bond so that repulsion does not over take attraction
What is optimal bond energy?
The amount of energy input, in kJ/mol, required to break bond
What makes a strong bond?
Higher order (more bonds)
More polar (a more polar bond indicates very partial charges that are attracted to one another)
Shorter bond length (look at the atom size)
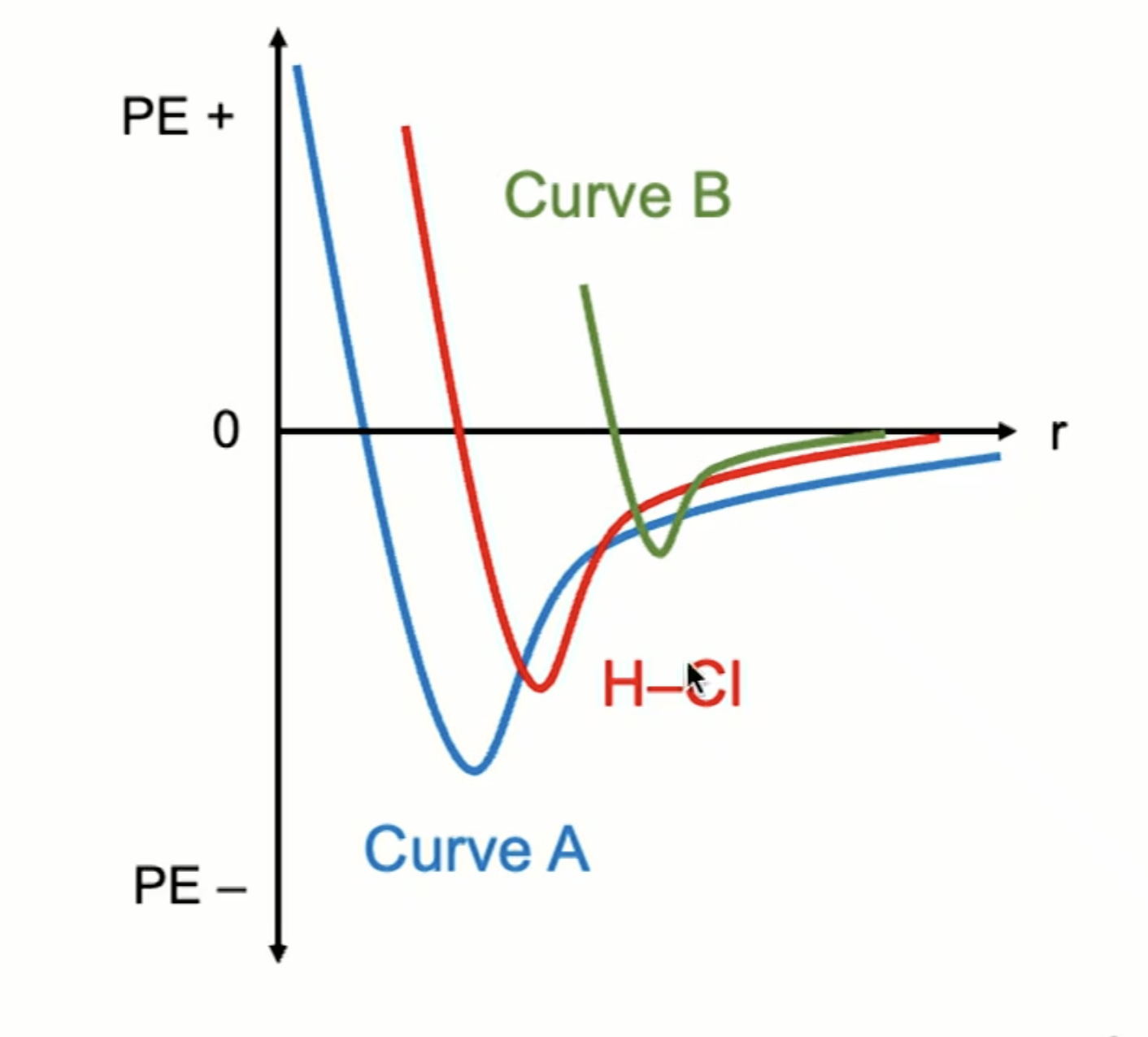
When comparing two bonds’ attraction/repulsion graph, what can you look for?
(example pictured)
Radius: a shorter bond length will be closest to the axis (smaller r = stronger attraction)
Depth of curve, which represents bond strength (deeper curve = more PE, stronger attraction)
T/F: Energy is stored in bonds, and breaking those bonds releases energy
False!! Common misconception, though :D
Bond breaking is (endothermic/exothermic)
Bond breaking is endothermic
It requires energy input to break the bonds
How can bond breakage release energy?
Forming an even stronger bond
Which characteristics does low PE correspond with?
(Negative/positive)
(Strong/weak) attraction
Low PE = negative = strong attraction
Which characteristics does high PE correspond with?
(Negative/positive)
(Strong/weak) attraction
High PE = positive = weak attraction
What sign is an exothermic ΔH(rxn)?
ΔH(rxn) < 0
or
ΔH(rxn) is negative
What sign is an endothermic ΔH(rxn)?
ΔH(rxn) > 0
or
ΔH(rxn) is positive
In an exothermic reaction, products have (higher/lower) PE and (higher/lower) bond energies
In an exothermic reaction, products have lower PE and higher bond energies
In an endothermic reaction, products have (higher/lower) PE and (higher/lower) bond energies
In an endothermic reaction, products have higher PE and lower bond energies
What does Hess’s Law say?
If a reaction can be written as a sum of several steps, the ΔH is equal to the sum of Δ of each step
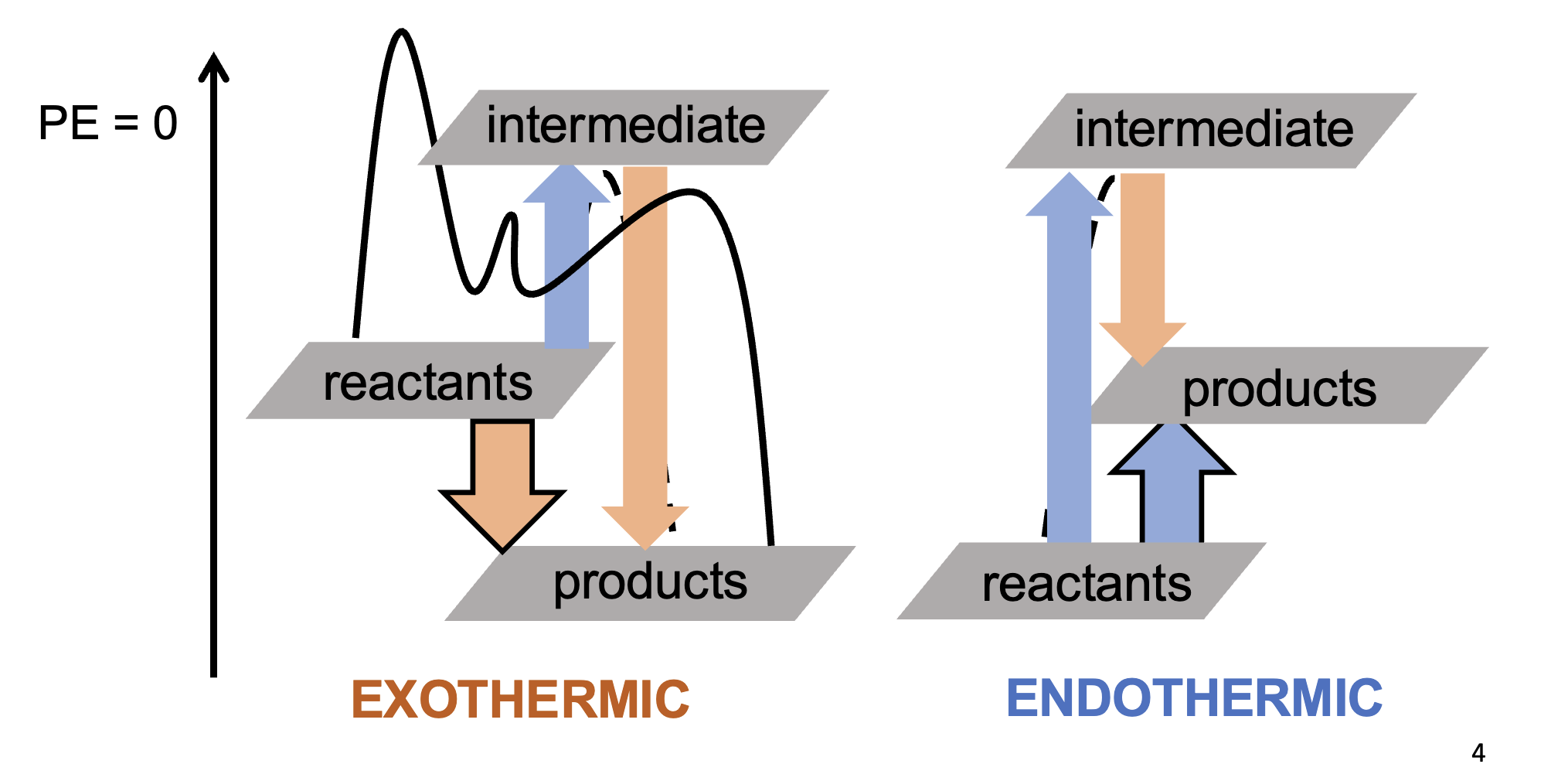
What is a pure element?
The most stable version of an element @ 1 atm and 298 K
What state of matter are most elements in at standard conditions? What about noble gases?
Most elements are solids at standard conditions
Noble gases are gases
Which elements want to be “doubled” at standard conditions?
H O N Cl Br F
What is enthalpy of formation? aka ΔHfo
Heat energy released or consumed when 1 mol of substance is formed from pure elements in the most stable form @ standard state
What unit is enthalpy of formation in?
kJ/mol^-1
What is important to note about pure elements in ΔHfo equations?
Pure elements ΔHfo = 0 since you don’t form anything with pure elements (they’re already pure, as the name suggests)
What is the ΔHrxno equation?
ΔHrxno = ∑nΔHof(product) - ∑nΔHof(reactants)
aka
[number of mols (enthalpy of formation of products)] - [number of mols (enthalpy of formation of reactants)]
Given the three ways below to model PE or KE changes, which model corresponds to which energy change?
q = mcΔT
ΔHfus/con/vap
ΔHrxn
KE: q = mcΔT
PE: ΔHfus/con/vap and ΔHrxn
Is enthalpy alone enough to predict the spontaneity of changes?
No!
What is spontaneity?
The “natural” direction of change
Is spontaneity always +ΔH?
No- can be +ΔH, -ΔH, or ΔH = 0
What is entropy (S)?
Dispersion/spread of matter and energy
How is entropy measured?
The number of ways you can arrange a system
Many ways to arrange a system = (higher/lower) entropy
Higher
Fewer ways to arrange a system = (higher/lower) entropy
Lower
What is a perfect crystal, and what are its conditions?
Perfect crystals occur at T = 0 K, there is no entropy
What is the ΔS of everything other than a perfect crystal?
S > 0
When comparing entropy, what can you look at?
Phase of matter
# of particles
Which is hotter
# of molecules
Spread
Arrange phases of matter by entropy, high to low
Gas > liquid > solid
If both objects are gases, what can you look at to determine higher entropy?
Which has more particles
More particles = higher entropy
If both objects are solids, what can you look at to determine higher entropy?
Which is hotter
Do more or less molecules signal higher entropy?
More
Does less or more spread out particles signal higher entropy?
More spread
What is the equation for ΔSorxn?
ΔSorxn = ∑nSoproducts - ∑nSoreactants
What does a negative ΔS indicate?
Nonspontaneous
What does a positive ΔS indicate?
Spontaneity
What is the relationship between ΔSsurr and ΔHsys?
ΔSsurr and ΔHsys are always the opposite sign of each other
Are ΔSsurr and ΔSsys always opposite signs from one another?
No! They can be, but it’s not a rule
What is the equation for ΔS using ΔH and T?
ΔS = ΔH/T
When should you use ΔS = ΔH/T?
Constant T, like during a phase change
What is the equation for ΔStotal?
ΔSsurr + ΔSsys = ΔStotal
What sign is ΔStotal?
Should always be ΔStotal > 0
What is the second law of thermodynamics?
Changes occur so universal entropy increases, aka so that ΔStotal is positive
If ΔStotal is positive, it is (nonspontaneous/spontaneous)
If ΔStotal is positive, it is spontaneous