CH 1 electro chem
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Oxidation numbers
OS of individual atoms = 0
total OS of alla toms in:
- neutral species = 0
ion = total charge of ion
in its compund OS of group 1 metal = +1, group 2 = +2,
F= -1
H= +1
O = -2
group 17 = -1
group 16= -2
group 15 = -3
if there is an element more electronegative that O then o = +2
OS for complex compunds
1 . identify ligand charges (ex: NH3 = 0)
2 write ox equation
electro chemistry
Exchange of chemical energy (rxn) and electrical energy (current)
oxidation
Loss of e-s (LEO)
reduction
Gain fo e-s (GER)
how to balnce redox
determine which is reduction and which is oxidation and split into two half rxns
blance main elements of rns (all atoms except o and H
Then balance O by adding H2O
then balance H by addign H+, where ever H is electons will follow
Balance charge with e-s, (look at overall charge of each side, including H+ charge) place e-s with wherevver H+ is at
blance e-s of both eqautiosn with least common multiple so they calnce out
then add the sides and cancel out common species
Acidic conditions redox
For acidc conditions follow like normal
Basic conditions redox rxns
For basc conditions, after step 5…
Add OH- on both sides to get rid of H+
convert H+ to H2O via (H+ + OH- → H2O)
cancle out waters
make sure t multi the e-s and equatiosn so they the same - then after that combien bot equations, cancleing out out common specie
or
after step 4 convert thoese directly to OH- IF there is no H+
galvanic cells
PArts of galvanic cells
how to draw galvanci cells
short hand notation of galvanic cells
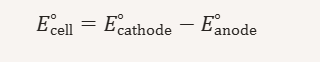
How to find E^ocell / voltage
identify which half rxn us reduction and which sis oxidation
look up at E^o values form standard reduction vlaues - tables always list reduction potential, even if rxn is ox don’t flip E^o value
use equation - anode = ox and cathode = oxidation
Don;t mutliply the half rxns
if both E^o same ?
then flip the one that will not make the final value negative (smaller one)