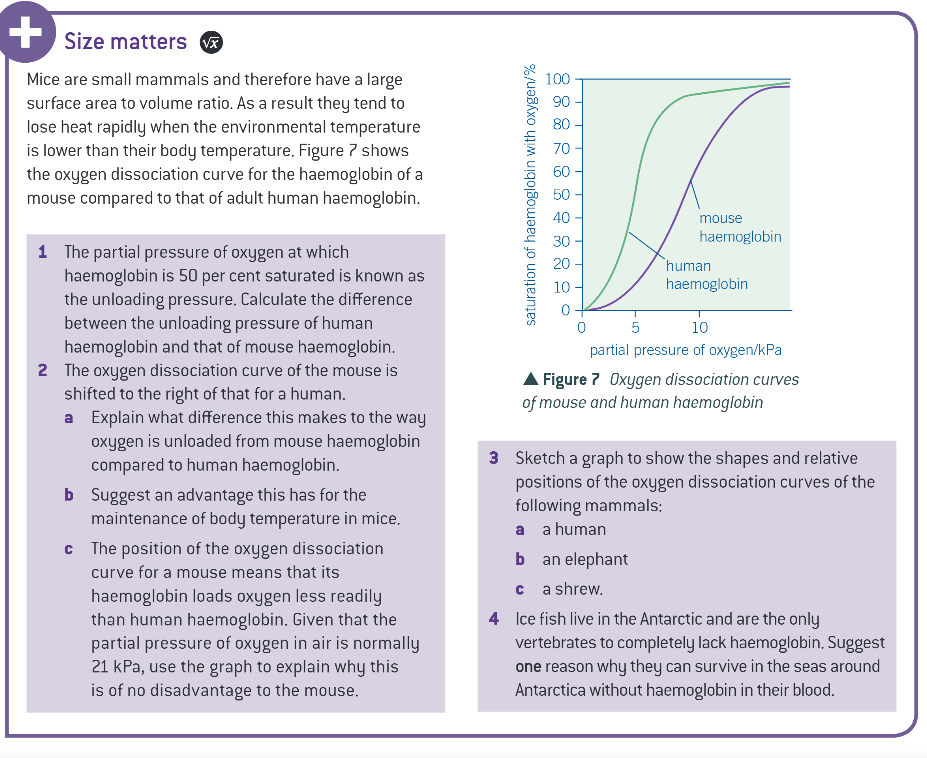
mass transport
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
what are the haemoglobins?
a group of chemically similar molecules found in a wide variety of organisms. they are protein molecules with a quaternary structured that has evolved to make it efficient at loading oxygen in one condition and unloading it in a different condition
primary, secondary, and tertiary structure of haemoglobin:
p: sequence of amino acids in the 4 polypeptide chains
s: in which each of these polypeptide chains is coiled into a helix
t: in which each polypeptide chain is folded into a precise shape - important factor to carry oxygen
quaternary structure:
in which all 4 polypeptides are linked together to form an almost spherical molecule.
each polypeptide is associated with a haem group, which contains Fe2+ (ferrous) ion
each Fe2+ ion can combine with a single O2 molecule, making a total of 4 O2 molecules that can be carried by a single haemoglobin molecule in humans
what is the process by which haemoglobin binds with oxygen?
loading/ associating (takes place in lungs)
what is the process by which haemoglobin releases its oxygen?
unloading/ dissociating (takes place in tissues)
haemoglobins with high affinity for oxygen…
takes up O2 easier but releases it less easily (opposite for low affinity)
what is the role of haemoglobin?
to transport oxygen
to be efficient at transporting oxygen, haemoglobin must:
readily associate with oxygen at the surface where gas exchange takes place
readily dissociate from oxygen at those tissues requiring it
how do these conditions take place considering they contradict each other?
it changes its affinity (chemical attraction) for oxygen under different conditions. it does this as its shape changes in the presence of certain substances, like CO2
what does haemoglobin do in the presence of CO2?
the new shape of the haemoglobin molecule binds more loosely to oxygen, making it release its O2
diagram to show affinity of haemoglobin for oxygen under different conditions:

what did scientists propose about haemoglobin when they first discovered it?
that it carried oxygen from the gas exchange surfaces to the tissues that required it for respiration, meaning that it must readily combine with oxygen
how did they find that there was different types of haemoglobin?
they investigated the ability of haemoglobin from different organisms to combine with oxygen. these exhibited different properties relating to the way they took up and released oxygen
why do different haemoglobins have different affinities for oxygen?
lies in the shape of the molecule
each species produces a haemoglobin with a slightly different amino acid sequence
therefore slightly different tertiary and quaternary structure and so different binding properties
depending on its structure haemoglobin molecules range from those that have a high affinity for oxygen to those with a low affinity for oxygen
what is the case when haemoglobin is exposed to different partial pressures of oxygen?
it doesnt bind the oxygen evenly
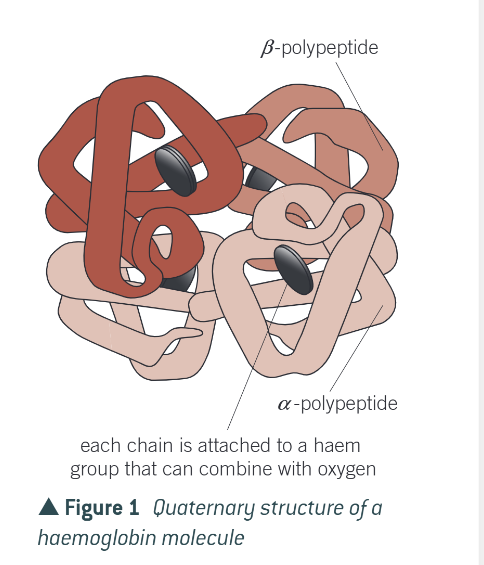
what is an oxygen dissociation curve?
the graph of the relationship between the saturation of haemoglobin with oxygen and the partial pressure of oxygen
explanation for the shape of the oxygen dissociation curve:
the shape of the haemoglobin (H) molecule makes it difficult for the first O2 molecule to bind to one of the sites of its 4 polypeptide subunits as they are closely united. therefore a low O2 concentrations, little oxygen binds to H (shallow initially)
the binding of this first O2 molecules changes the quaternary structure of the haemoglobin molecule, causing it to change shape. this change makes it easier for the other subunits to bind to an oxygen molecule (the binding of the first O2 molecule induces other subunits to bind to an O2 molecule)
therefore takes a smaller increase in the partial pressure of oxygen to bind to the second O2 molecule than it did to bind the first one. positive cooperativity (gradient of curve steepens)
situation changes after the binding of the third molecule - in theory would be easier, but in practice is harder due to probability. with the majority of the binding sites occupied, it is less likely that a single oxygen molecule will find an empty site to bind to (gradient reduces and graph flattens off)
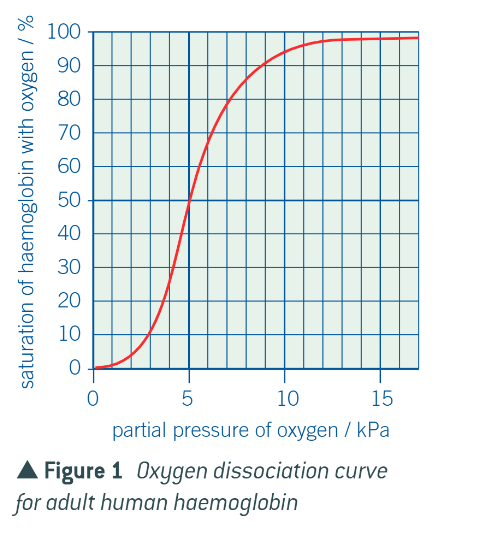
why are there different oxygen dissociation curves? (slightly)
different haemoglobins and shape can change of the haemoglobin under different conditions
what are important facts for the different oxygen dissociation curves?
the further to the left of the curve, the greater is the affinity of haemoglobin for oxygen (so it loads O2 readily but unloads it less easily)
the further to the right of the curve, the lower is the affinity of haemoglobin for oxygen (so it loads oxygen less readily but unloads it more easily)
the greater the conc of CO2…
the more readily the haemoglobin releases its oxygen
whats this known as?
the bohr effect
what does it explain?
why the behaviour of haemoglobin changes in different regions of the body
what is CO2 like at the gas exchange surface?
the conc of CO2 is low as it diffuses across the exchange surface and is secreted from the organism
the affinity of H for oxygen is increased, which (with high conc of O2 in lungs) means that oxygen is readily loaded by haemoglobin
the reduced CO2 conc has shifted the oxygen dissociation curve to the left
what is CO2 like in rapidly respiring tissues?
the conc of CO2 is high
the affinity of H for oxygen is reduced, which (with low conc of O2 in the muscles) means that oxygen is readily unloaded from the haemoglobin into the muscle cells
the increased CO2 conc has shifted the oxygen dissociation curve to the right
diagram to show this:
why does greater conc of CO2 make H release its oxygen more readily?
dissolved CO2 is acidic and the low pH causes haemoglobin to change shape
loading, transport, and unloading of oxygen:
at the gas exchange surface CO2 is constantly being removed
the pH is slightly raised due to low conc of CO2
the higher pH changes the shape of haemoglobin into one that enables it to load oxygen readily
this shape also increased the affinity of H for oxygen, so its not released while being transported in the blood to the tissues
in the tissues, CO2 is produced by respiring cells
CO2 is acidic in solution, so the pH of the blood within the tissues is lowere
the lower pH changes the shape of haemoglobin into one with a lower affinity for oxygen
haemoglobin releases its oxygen into the respiring tissues
process in short:

what is the case with H in practice?
no all haemoglobin molecules are loaded with their max 4 oxygen molecules
what is a consequence of this?
the overall saturation of haemoglobin at atmospheric pressure is around 97%
what happens when this H reaches a tissue with a low respiratory rate?
only one of these molecules will normally be released
therefore what is the case?
the blood returning to the lungs will therefore contain haemoglobin that is still 75% saturated with oxygen
what about if the tissue is very active?
3 oxygen molecules will usually be unloaded from each H molecule
diagram to show this:
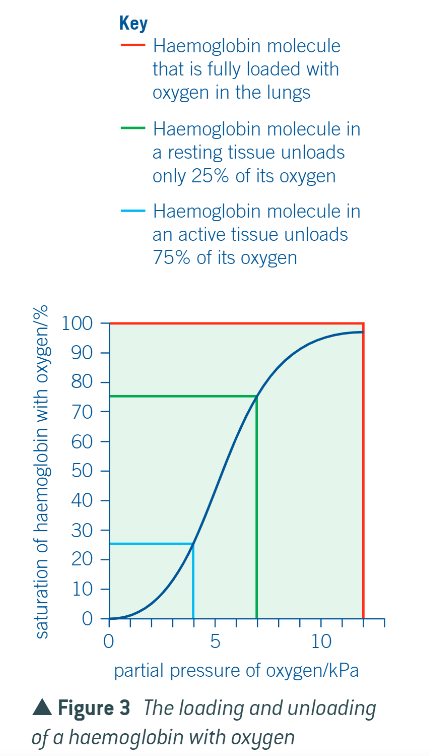
different species have different haemoglobins with different oxygen dissociation curves: how have these different types evolved?
within species as adaptations to different environments and conditions
example:
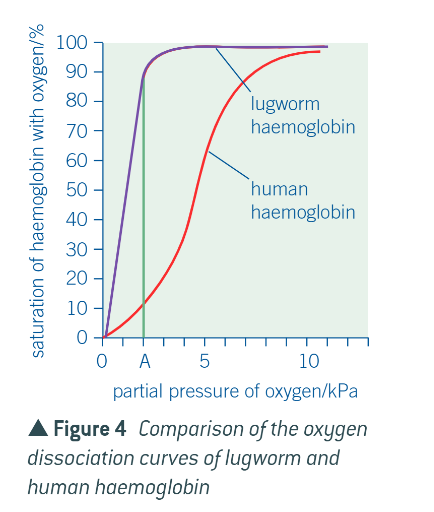
example of lugworm, an animal that lives on the seashore:

diagram to show lugworm oxygen dissociation curve in comparison to human haemoglobin
extra example: llama:

extra info: activity counts:

extra info: size matters: