2.5: Transition metals
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
What is a transition metal?
A d-block element with a partially filled d-orbital, that loses e- to form positive ions with s block e- being lost first
What are characteristic properties of transition metals?
Similar atomic radii
High densities
High melting points/boiling points
Ability to form complexes
Ability to form coloured ions
Variable oxidation states
Good catalysts
What is a complex?
A central metal atom or ion (transition metal) surrounded by ligands
What is a ligand?
A molecule/ion that forms a co-ordinate bond with a transition metal by donating a lone pair of electrons
What is co-ordination number?
The number of co-ordinate bonds to the central metal atom or ion
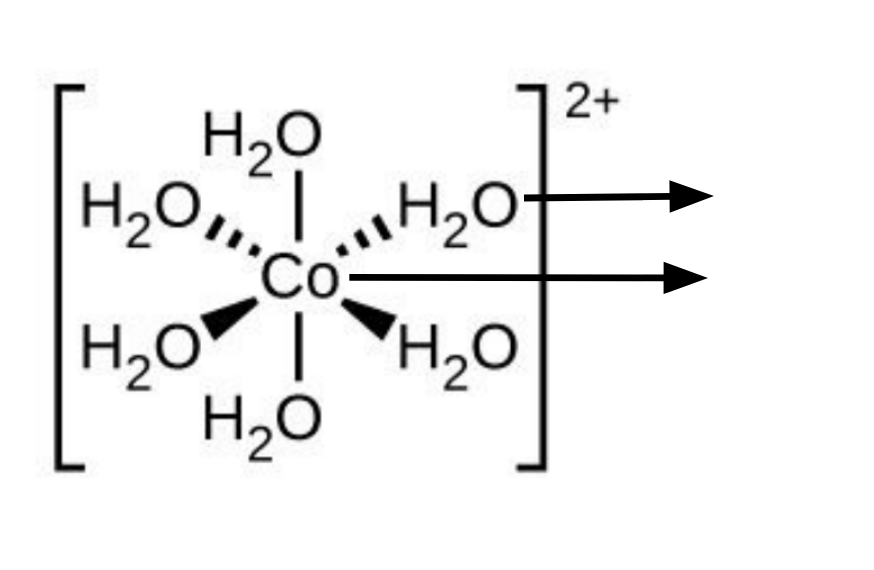
Label this diagram, give the co-ordinate number and give an alternate way that this complex can be written
Ligand, complex
Co-ordination number = 6
[Co(H2O)6]2+
![<ul><li><p>Ligand, complex </p></li><li><p>Co-ordination number = 6</p></li><li><p>[Co(H<sub>2</sub>O)<sub>6</sub>]<sup>2+</sup></p></li></ul><p></p>](https://assets.knowt.com/user-attachments/6417e220-6d5c-4288-84bd-46c9994f02c3.png)
What is a monodentate ligand, give examples, and explain how they work in ligand substitution reactions?
A ligand that forms 1 co-ordinate bond
H2O, NH3
They are similar in size and uncharged, so they can be exchanged without a change via a ligand substitution reaction without causing a change in co-ordination number
What is the ligand substitution reaction of [Co(H2O)6]2+ and NH3?
[Cu(H2O)6]2+ + 6NH3 → [Cu(NH3)6]2+ + 6H2O
What is the ligand substitution reaction of [Cu(H2O)6]2+ and NH3?
[Cu(H2O)6]2+ + 4NH3 → [Cu(H2O)2(NH3)4)2+ + 4H2O
Incomplete substitution reactions
Forms a deep blue solution
What is the ligand substitution reaction of [Cu(H2O)6]2+ and Cl-?
[Cu(H2O)6]2+ + 4Cl- → [CuCl4]2- + 6H2O
Cl- ligand is larger than NH3 and H2O, so substitution causes a change in co-ordination number
Forms a tetrahedral complex
What is a bidentate ligand, and give examples?
A ligand that is able to form 2 co-ordinate bonds to the central metal atom, as they have 2e- lone pairs
Ethanedioate and ethane-1,2-diamine
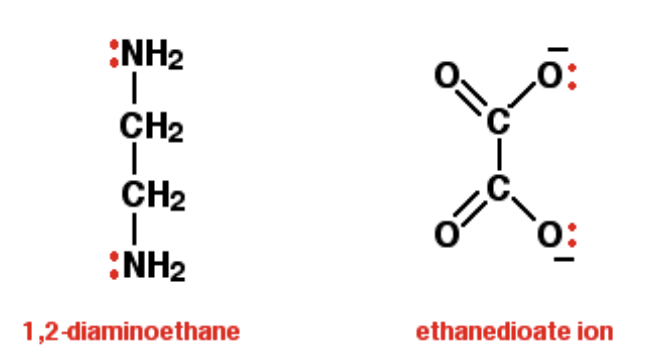
What is a multidentate ligand, and give examples?
A ligand able to form up to 6 co-ordinate bonds to the central metal ion as they have multiple e- lone pairs
EDTA4- and haem (an iron complex with a multidentate ligand)
What is the ligand substitution reaction of [Fe(H2O)6]3+ and EDTA4-?
[Fe(H2O)6]3+ + EDTA4- → [FeEDTA]- + 6H2O
What is the role of haemoglobin?
Oxygen forms a co-ordinate bond to Fe(II) in haemoglobin, allowing oxygen to be transported in the blood
Why is CO toxic?
Replaces O2 coordinately bonded to Fe(II) in haemoglobin
So oxygen cannot be transported around the body and delivered to tissues
What is the chelate effect?
In ligand substitution reactions, a positive entropy change is favourable as a more stable complex is being formed
Meaning it is better to have more moles on the right side of the equation that the left
So it is favourable so substitute monodentate ligands with bidentate/multidentate ligands
∆H for these reactions are very small as bonds being formed is similar to bonds being broken, so enthalpy = 0
What shape and bond angle is formed when transition metals form complexes with small ligands (H2O/NH3), and what type of isomerism is shown?
Octahedral
90°
Cis-trans isomerism
What shape and bond angle is formed when transition metals form complexes with bidentate ligands, and what type of isomerism is shown?
Octahedral
90°
Optical isomerism
What shape and bond angle is formed when transition metals form complexes with larger ligands, and what type of isomerism is shown?
Tetrahedral
109.5°
Optical isomerism
What shape and bond angle is formed when Pt/Ni forms complexes with ligands, and what type of isomerism is shown?
Square planar
90°
Cis-trans isomerism
What is cisplatin and why is only this molecule effective?
Cancer therapy drug
Only the cis isomer is effective as natural cells are chiral
What shape and bond angle is formed when Ag+ forms complexes with small ligands, and what is the product used for?
Linear
180°
Tollens’ reagent
How can different transition metals be identified?
Colour
What is the colour of a transition metal dependent on?
Co-ordination number of the complex
Type of ligand bonded to the ion
Oxidation state
How does colour arise in transition metals?
Some wavelengths of visible light are absorbed
When light is absorbed, d electrons move from the ground state when light is absorbed
Remaining wavelengths of light are transmitted and reflected to the human eye
Reflective wavelengths correspond to a specific colour which is observed by the human eye
How is the energy difference between the ground state and excited state of the d electrons calculated?
∆E = h ν = hc /λ
∆E = energy
h = planck’s constant (6.63×10-34)
ν = frequency (Hz)
c = speed of light (3.00×10-8m/s)
λ = wavelength (m)
What are factors that effect energy between ground and excited electrons?
Co-ordination number of the complex
Type of ligand bonded to the ion
Oxidation state
How can the concentration of coloured ions be measured?
Colorimetry
Uses absorption of visible light
Allows a calibration graph of relative absorbance (y) vs % concentration (x)
Which is then used to calculate the concentration of an unknown substance
What are the different oxidation states of vanadium, what is their ion formula, and what colour is each species?

How are vanadium species in oxidation states IV,III and II formed?
The reduction of vanadate (V) iosn by zinc in acidic solution
What is redox potential, and what is it influenced by?
How easily an ion can be reduced to a lower oxidation state
Influenced by pH and the ligand
Why is redox potential influenced by pH?
For ions to be oxidised, they react with water to produce OH-, so alkaline conditions are required
For ions to be reduced, acidic conditions are required
Why is redox potential influenced by ligands?
Some ligands are stronger than each other (NH3 is stronger than H2O)
So the stronger ligand binds tighter to the central metal ion
So redox potential becomes more negative
What is Tollens’ reagent used for, and what is the equation?
Used to differentiate between aldehydes and ketones
Tollens’ [Ag(NH3)2]+ is reduced by aldehydes to form a metallic silver mirror
RCHO + 2[Ag(NH3)2]+ + 3OH- → RCOO- + 2Ag + 4NH3 + 2H2O
What are the equations for the redox titrations of Fe2+ and C2O22- with MnO4-
MnO4- + 8H+ + 5e- → Mn2+ + 4H2O
Fe2+ → Fe3+ + e-
C2O4- → 2CO2 + 2e-
What are the molar ratios for the redox titrations of Fe2+ and C2O22- with MnO4-
MnO4- : C2O4- = 2:5
MnO4- : Fe2+ = 1:5
C2O4- : Fe2+ = 1:2
What is a catalyst?
A species that speeds up the rate of reaction by providing an alternative pathway with a lower activation energy without being used up
What is a homogenous catalyst?
A catalyst in the same phase as the reactants
What is a heterogeneous catalyst, and what are some examples?
A catalyst in a different phase to the reactants, where the reaction occurs at active sites on the surface
What are examples of heterogeneous catalysts and what are the equations?
V2O5 acts as a catalyst in the contact process
Overall equation:
2SO2 + O2 → 2SO3
Intermediates:
V2O5 + SO2 → V2O4 + SO3
V2O4 + ½O2 → V2O5
Fe acts as a catalyst in the Haber process
N2 + 3H2 → 2NH3
What is the use of catalytic support mediums, and give an example of where it is used?
Used to maximise surface area of a heterogeneous catalyst and minimise the cost
Pt is spread out in a honeycomb structure on a catalytic converter
How do solid heterogeneous catalysts work?
On the surface of a solid catalyst, reactant molecules are absorbed onto active sites
This increases the proximity of the molecules so they are closer together
So covalent bonds are weakened, so the reaction takes place more easily
So rate of reaction is increased
What is strength of adsorption based on, which transition metals have the best adsorption and why?
Dependent on the type of catalyst
Fi, Co and Ni are the best, as they increase the rate of reaction the most out of period 4 transition metals and they are cheap
What is catalytic poisoning, what is the consequence of it, and what is an example of a poision in catalysis?
Heterogeneous catalysts can be poisoned by impurities which block active sites and prevent adsorption
So bonds between molecules remain strong, so catalyst has no effect on the rate
Leads to increase in costs as the catalyst has to be replaced and cleaned regularly
Sulfur poisons Fe catalyst in the Haber process
What is an example of a homogeneous catalyst, and what are the equations?
Fe2+ catalyses S2O82- and I-
Overall
S2O82- + 2I- → I2 + 2SO42-
Intermediates
S2O82- + 2Fe2+ → 2Fe3+ + 2SO42-
2Fe3+ + 2I- → 2Fe2+ + I2
Why would S2O82- and I- be unable to react if not for a catalyst
They would both repel each other as they are both negative
What is autocatalysis?
One of the products as a catalyst for the reaction
So as product increases, the rate of reaction increases as the reaction becomes catalysed@
What is an example of autocatalysis, and what are the equations and conditions?
Mn2+ ions are used as a catalyst in the redox reaction between MnO4- and C2O42- ions in acidic solutions
Requires a small amount of heat
Overall
2MnO4- + 16H+ + 5C2O42- → 2Mn2+ + 8H2O + 10CO2
Intermediates
4Mn2+ + MnO4- + 8H+ → 5Mn3+ + 4H2O
2Mn3+ + C2O42- → 2CO2 + 2Mn2+