Chemistry - Environmental Chemistry
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
How long has Earth’s current atmosphere been the same?
For the last 200M years.
What was early earth like 4.5B years ago?
Atmosphere had 95% carbon dioxide, 4% water vapour, and traces of nitrogen, ammonia and methane. Surface temperature was more than 400 C.
How has Earth’s atmosphere evolved over the past 4.5B years?
Volcanic activity released CO2, water vapour, methane, ammonia and nitrogen. Earth cooled over millions of years, condensing the water vapour, forming oceans.
The CO2 dissolved in the oceans, forming carbonate sediments, e.g. limestone. Photosynthetic organisms also evolved to photosynthesise. They removed CO2 from the atmosphere and released oxygen.
CO2 got trapped in organisms as glucose (biomass), being preserved as fossil fuels. These actions saw CO2 levels declining and O2 levels increasing.
Why has nitrogen been gradually increasing over millions of years, and has never decreased?
It is an inert substance (unreactive)
Why are ideas of the early atmosphere theoretical?
It was over 4.5B years ago, and no one was there then to conduct experiments - we have very little evidence / data.
How is data about the early atmosphere obtained?
Scientists take samples of ancient Arctic ice. Its composition is analysed, determining the composition of the air when the ice formed millions of years ago.
What is the greenhouse effect?
A natural process, where greenhouse gases (CO2, methane, water vapour) absorb radiation from the sun. This is needed to keep Earth’s temperature high enough to maintain life.
How are temperatures rising because of this?
The greenhouse gas layer is thickening, resulting in too much radiation being absorbed, causing global temperatures to rise (global warming).
How does the greenhouse effect work?
Short wavelength radiation from the sun is absorbed the planet’s surface. The surface re-radiates long wavelength radiation into the atmosphere. This is absorbed by greenhouse gases, preventing it from escaping. This traps heat at the Earth’s surface, rising the average surface temperature.
How can the greenhouse effect be enhanced?
If the concentration of greenhouse gases in the atmosphere increases.
How is human activity enhancing the greenhouse effect?
Combustion of fossil fuels - releases CO2 and water vapour. Increases carbon emissions.
Deforestation - clearing land. Trees release CO2, reducing global photosynthesis.
Mass farming - fuel use releases CO2, cattle farming releases methane.
Rice farming - flooded rice paddies release methane from decomposing organic matter.
Landfill - anaerobic fermentation releases methane.
What are the effects of global warming?
Rising average global temperatures.
Melting of polar ice caps, meaning rising sea levels.
Extreme variations in seasonal temperatures.
More frequent weather-based natural disasters, e.g. hurricanes, wildfires, droughts, flooding, etc.
Increased desertification - making more places desert-like.
What is your carbon footprint?
The sum of all greenhouse gases emitted by a person, product or industry.
What things does the carbon footprint include?
Manufacture, distribution, packaging, product transport.
Repair and/or production disposal.
Human influence, e.g. diets, recycling habits, use of public transport, energy-use-consciousness.
Government laws outlining responsibilities of industries to lower carbon emissions (e.g. UK’s ‘net zero’ goal by 2050).
How are carbon dioxide and water vapour atmospheric pollutants?
Released in complete combustion. Contributed to greenhouse effect. Results in global and warming and extreme climate changes. CH4 + O2 → CO2 + H2O (methane + oxygen → carbon dioxide + water).
How is carbon monoxide an atmospheric pollutant?
Released in incomplete combustion. It is toxic to humans - binds to haemoglobin in red blood cells, meaning they can’t carry oxygen around the body. Long-term exposure can be fatal. C2H4 + O2 → CO + H2O (ethene + oxygen → carbon monoxide + water)
How are unburnt hydrocarbons, soot, and carbon particulates atmospheric pollutants?
Incomplete combustion produces solid carbon soot - C2H4O2 + O2 → 2C + 2H2O (ethene + oxygen → soot + water). Damaging to health - becomes stuck in lungs. Damages environment - global dimming at surface. Hydrocarbons are compounds containing carbon and hydrogen.
What are hydrocarbons?
Compounds containing only carbon and hydrogen.
How is sulphur dioxide an atmospheric pollutant?
Fossil fuels burnt normally contain impurities of sulphur. They are oxidised to form sulphur dioxide. S + O2 → SO2 (sulphur + oxygen → sulphur dioxide). A respiratory irritant. Dissolves in rainwater forming sulfuric acid. Major part of acid rain. SO2 + 2H2O → H2SO4 + H2 (sulphur dioxide + water → sulphuric acid + hydrogen).
What does acid rain do?
Kills plants by disrupting the soil acidity; damages aquatic environments; corrodes buildings made of limestone, and corrodes metals, weakening building structure.
How are oxides of nitrogen atmospheric pollutants?
Reacts with oxygen at high temperatures and pressures with internal combustion engines. Forms harmful pollutants called nitrous oxides (N2 + O2 —> 2NO, or, N2 + 2O2 —> 2NO2). Harmful to health - causes respiratory problems, and can contribute to heart disease. Dissolves in water forming nitric acid - major component of acid rain (NO2 + H2O —> HNO3 + NO).
What are ceramics?
Non-metallic solids with high melting points.
List three properties of ceramics.
Excellent insulators of heat and electricity, and very brittle and stiff.
How are clay ceramics made?
When dug up, clay is soft. It is moulded into the intended shape, and is hardened at high temperature.
What is most glass made from, and how is it made?
Soda-lime - a mixture of limestone, sand, and sodium carbonate heated until it melts. It cools into glass.
How is borosilicate glass different to soda-lime glass?
Has a higher melting point than soda-lime glass. It is made in the same way, but the mixture contains sand and boron trioxide.
What is a composite?
A material made from one material embedded in another.
List four examples of composites.
Wood, fibreglass, carbon fibre, and concrete.
What is the reinforcement and matrix in wood, fibreglass, carbon fibre and concrete?
Wood - reinforcement is cellulose fibre, matrix is lignin, an organic polymer.
Fibreglass - reinforcement is fibres, matrix is polymers (plastic).
Carbon fibre - reinforcement is carbon fibres/nanotubes, matrix is polymers (plastic).
Concrete - reinforcement is aggregate (sand + gravel), matrix is cement.
What is a polymer?
Very large molecules that are formed when smaller molecules called monomers join together.
List three useful properties of polymers.
Insulator of heat and electricity
flexible
easily moulded.
List an example of a polymer and its use.
Polyester - used in clothing.
What two factors influence the property of a polymer?
The monomers it’s made from and the conditions in which it is made.
What does a thermosoftening polymer look like? What are the lines called?
Polymer chains.

Why can thermosoftening polymers be melted easily and are flexible?
There are weak forces between the polymer chains.
What does a thermosetting polymer look like? What are the lines in between called?
Cross links.
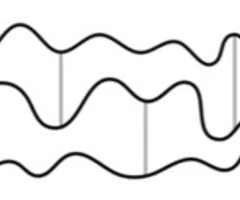
Why are thermosetting polymers rigid and unable to melt easily?
The cross links and ionic bonds provide strong forces between the chains.
Where is low-density polyethene used?
In bags and bottles.
Where is high-density polyethene used?
In water tanks and drain pipes.
What is the difference between LD and HD polyethene?
HD polyethene is made at a lower temperature and is more rigid than LD polyethene.
List five useful properties of metals.
Malleable
good conductors of heat and electricity
ductile (drawn into wires)
shiny
stiff, etc.
What is an alloy?
A mixture of two or more metals or a metal and non-metal.
Why are alloys harder than pure metals?
Different elements’ atoms mean different sizes of atoms, which makes it more difficult for the atoms to slide over each other.
List the three different types of steel and their properties.
Low-carbon steel - easily shapeable.
High-carbon steel - strong, inflexible.
Stainless steel - corrosion-resistant, hard.
What metals are used to make bronze, and what is it used for?
Copper and tin - used for statues and medals.
What metals are used to make brass, and what is it used for?
Copper and zinc - used for taps and door fittings.
What metals are used to make gold alloys, and what is it used for?
Zinc, copper and silver - used for jewellery.
What metals are used to make aluminium alloys, and what is it used for?
Unknown - used for aircraft manufacture.
What is the unit used to measure the amount of gold in gold alloys?
Carats.
What value is pure gold?
24 carats.
Define and list natural resources.
Resources produced without any human influence and instead grow/form naturally, such as water, coal, crude oil, metal ores, agricultural crops, cotton, wool and wood.
Define renewable and non-renewable resources and list examples.
Renewable resources can be reformed or replenished over a short time, such as wood, cotton or wool. Non-renewable resources are used up quicker than they are reformed, such as crude oil, which takes millions of years to reform, and coal.
Define sustainability.
Using resources to meet current needs without jeopardising the needs of people in the future.
What does a life cycle assessment do?
Assesses how sustainable a product is during it’s lifetime - from extraction of raw materials until its disposal.
What are the five stages of a life cycle assessment?
Extraction of raw materials
processing and manufacturing
distribution and transport
use of product by customer
disposal.
What makes a good LCA?
Low carbon emissions
long product lifespan
sustainability
reusability and/or recyclability
biodegradability
renewability.
What does recycling involve?
Separating, melting down and recasting into other products. Works for many materials, such as metal and glass.
List three limitations of LCAs.
Some impacts are easy to observe, whilst others are difficult to assess, e.g. indirect impacts.
Difficult to numerically assess environmental impacts. People have to make value judgments on the scale and severity of an impact.
LCAs can be manipulated to produce desired results. Done by deliberately judging impacts to be less severe or only including direct impacts of a product.