The Building Blocks of Molecules and Macromolecules
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Life is made up of matter.
Mater is made of elements, which is ___.
ex.
Matter is made up of elements, which is substances that can not be broken down into other substances.
ex. gold, carbon, etc.
Atom – ___
Protons are ____
Neutrons are ____
Electrons are ____
The + (protons) and – (electrons) charges ___
Atom – smallest unit of an element
Protons are positively charged atomic particles with a mass of 1 and +1 charge (located in nucleus).
Neutrons are atomic particles with no charge and a mass of 1 (located in nucleus).
Electrons are negatively charged atomic particles with negligible mass and -1 charge (located around the nucleus).
The + (protons) and – (electrons) charges balance each other in a neutral atom (0 charge).
Periodic Table of Elements
Atomic number:
Changing this number would change the identity of the element.
Atomic mass =
Number of neutrons =
Atomic number: identifies the element and represents the number of protons.
Changing this number would change the identity of the element.
Atomic mass = # of protons (atomic number) + # of neutrons
Number of neutrons = Atomic mass – Atomic number
Isotopes are ____
Some isotopes, like ____, can be used to date fossils.
Ions result when ___
gaining electron(s) results in ___
losing electron(s) results in ___
Isotopes are different forms of the same element that have the same number of protons, but different numbers of neutrons.
Some isotopes, like those of carbon and potassium, can be used to date fossils.
Ions result when the number of electrons varies.
Gaining electron(s) results in a negative charge.
Losing electron(s) results in a positive charge.
Chemical Bond Types
Chemical bonds hold atoms together to form molecules.
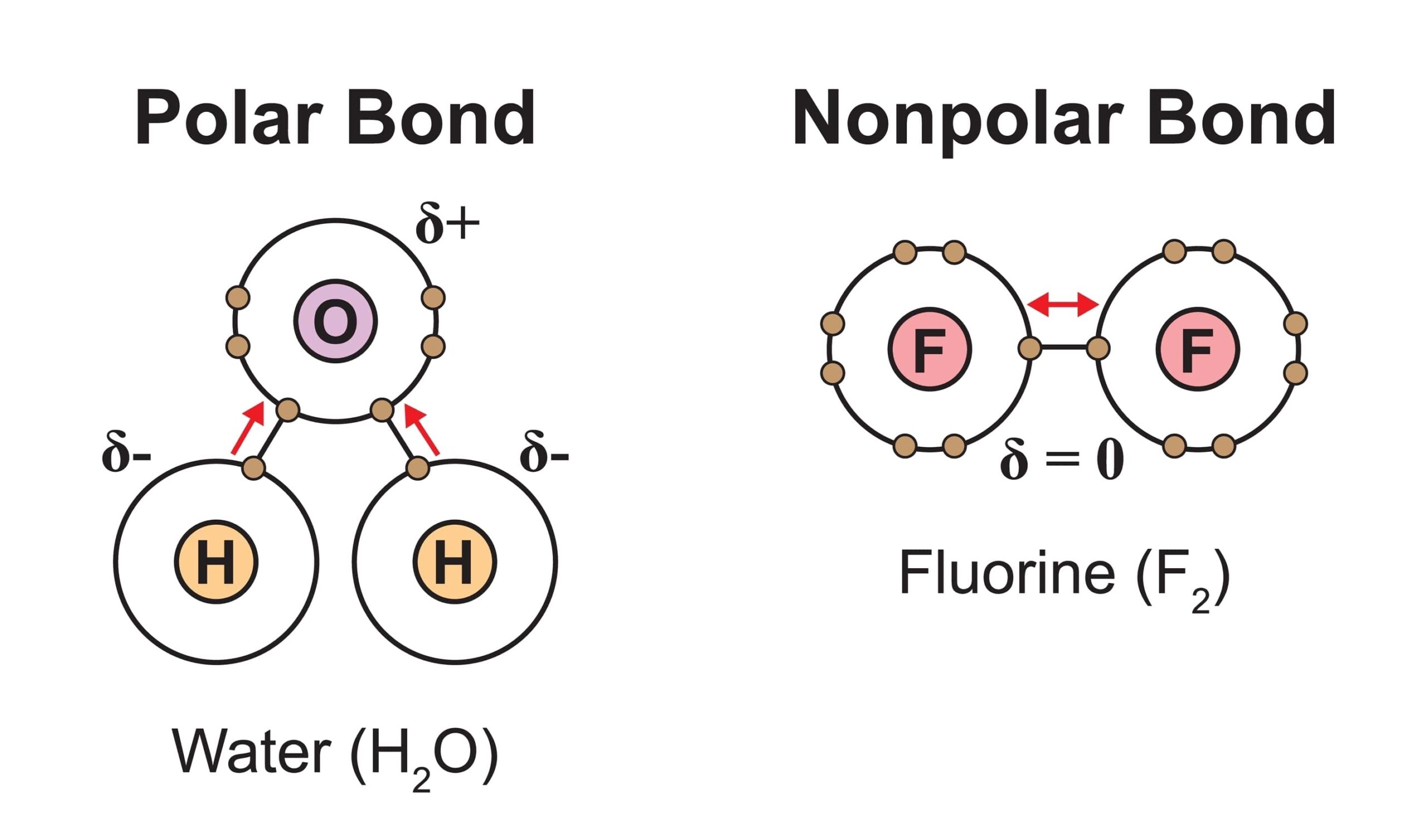
Covalent bonds are the ____.
They form when two atoms ___.
Nonpolar covalent bonds –
Polar covalent bonds –
ex.
Do covalent bonds dissociate (come apart) in water?
Covalent bonds form when two atoms share an electron
They the strongest bond.
Nonpolar covalent bonds – the electrons are shared equally.
Polar covalent bonds – the electrons are shared unequally (the electron spends more time closer to one atom than the other)
Ex. a water molecule is held together by polar covalent bonds
They don’t come apart in water.
Chemical Bond Types
Chemical bonds hold atoms together to form molecules.
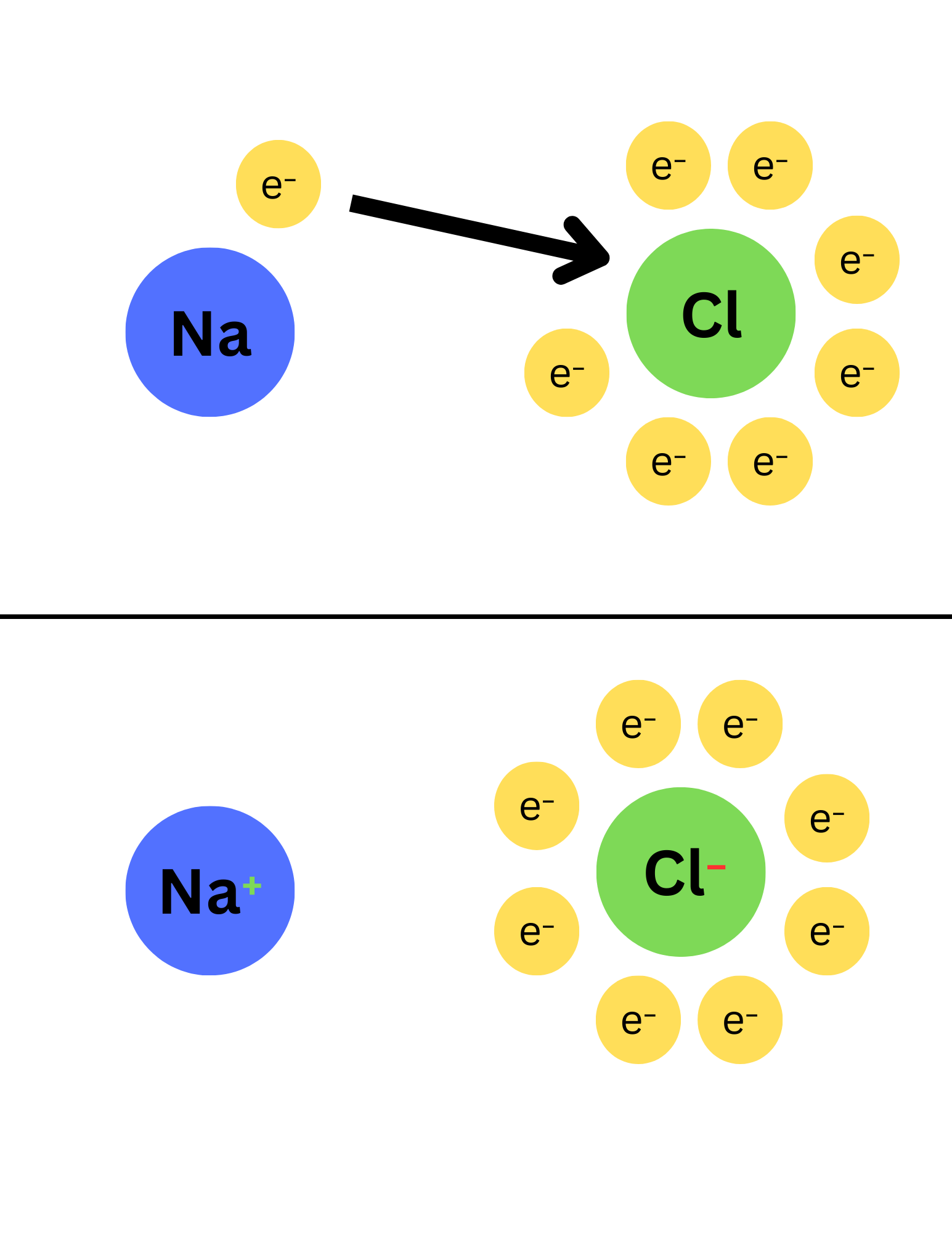
Ionic bonds – ____
Ionic bonds are ____
Do they dissociate in water?
Ex.
Ionic bonds – form when an electron is transferred from one atom to another → Form ions of opposite charges → These opposite charges attract each other, which holds the ions together.
Fairly strong bond.
They do dissociate in water.
Ex. Table Salt: Na donates an electron to Cl → forming ions (Na+/Cl-) → ionic bond will hold them together to form NaCl (table salt).
Chemical Bond Types
Chemical bonds hold atoms together to form molecules.
Hydrogen bonds –
Is the ___ bond of them all.
Ex.
Hydrogen bonds – form between the slightly-positive hydrogen atoms from a polar molecule and the slightly-negative atoms from another polar molecule.
Weakest bond of them all.
Ex. Water molecules are held together with hydrogen bonds.
What are other words for “water loving” and “water fearing”?
What type of molecules are each?
Polar molecules are hydrophilic – “water loving”
Nonpolar molecules are hydrophobic – “water fearing”
Cohesion – the force that causes__
Surface tension is the capacity of a substance to __
Adhesion – the force that causes __
ex.
Cohesive and adhesive forces allow a plant to draw water in through its roots.
Cohesion – the force that causes similar molecules (ex. water molecules) to stick together due to hydrogen bonding
Surface tension is the capacity of a substance to hold together when under stress.
Adhesion – the force that causes molecules of different substances to stick together
ex. Water molecules will also “stick” to other polar molecules by forming hydrogen bonds.
Cohesive and adhesive forces allow a plant to draw water in through its roots.
Carbohydrates ___
Monosaccharides –
Disaccharides –
Polysaccharides ___
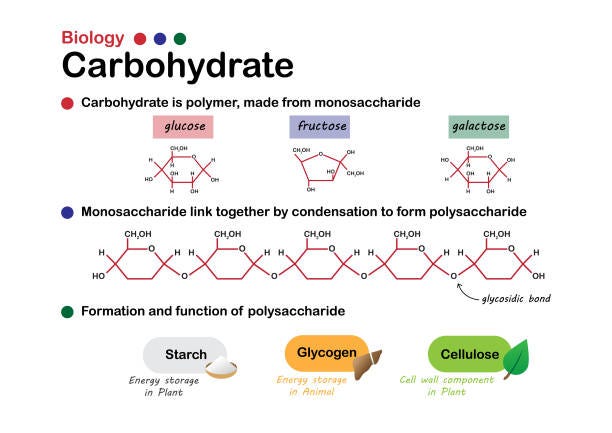
Carbohydrates are polymerrs made up of monomers called monosaccharides (simple sugars).
Monosaccharides – simple sugars (ex. glucose & fructose_
Disaccharides – 2 Monosaccharides bonded together (ex. sucrose and lactose)
Polysaccharides are complex carbohydrates; long chains of monosaccharides that are used to store energy (plants-starch and animals-glycogen) and to provide structure (cellulose).
Proteins ____
Functions:
The chemical bond between amino acids are ____.
Protein structure:
Primary Structure – ___
Secondary Structure – ____
Tertiary Structure – _____
Quaternary Structure – _____
Ex. _____
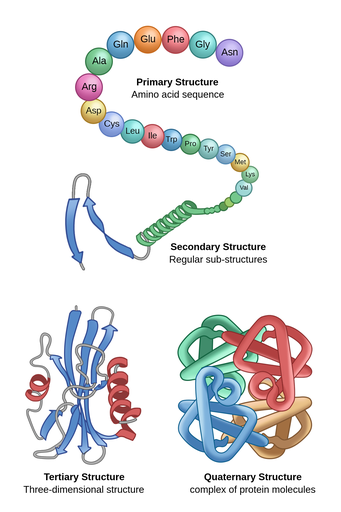
Proteins are polymers made of monomers called amino acids.
Functions: Structural, Regulatory, Contractile, Protective
The chemical bond between amino acids is peptide bonds.
Protein structure:
Primary Structure – the sequence of a chain of the amino acids
Secondary Structure – Hydrogen bonds folds the chain into “pleated sheets”
Tertiary Structure – Further folding to a 3-dimensional structure
Quaternary Structure – Sometimes more than one chain are put together, leading to a complex protein molecule
Ex. Hemoglobin (has four polypeptide chains).
Lipids are ___
How would the molecules be described?
Examples:
Triglycerides – ____
Phospholipids – ___
Steroids – ___
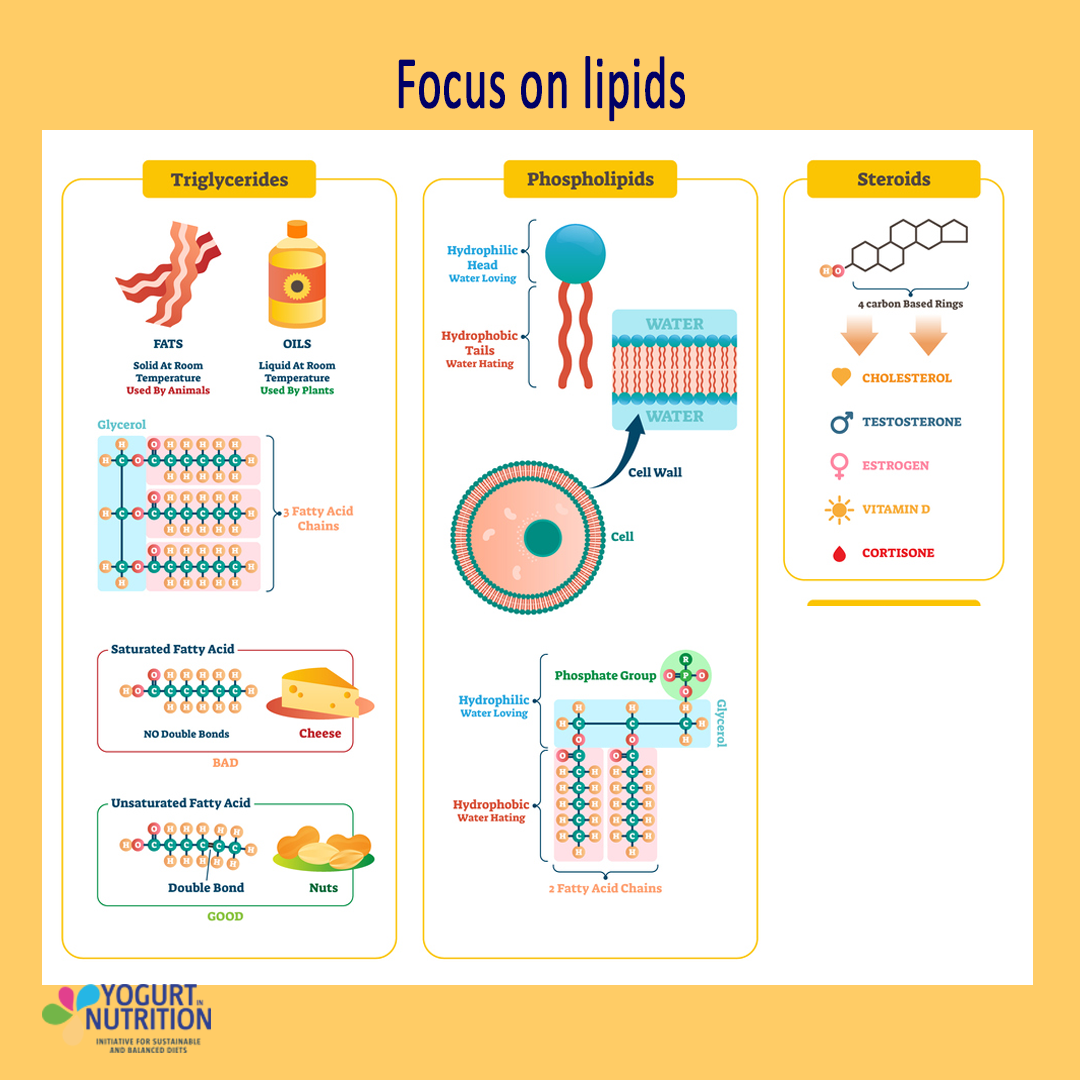
Lipids are macromolecules made up of fatty acids
Nonpolar, hydrophobic molecules
Examples:
Triglycerides – consists of 3 fatty acid chains and a glycerol.
Phospholipids – consists of a polar phosphate group (head) and two nonpolar fatty acid chains (tails).
Steroids – not made of fatty acid, but is hydrophobic, so is grouped as lipids.
Nucleic acids are ____
Nucleic acids carry ___
Can ___ itself.
Nucleotides are ___
Made up of?
Example:
DNA
Structure?
The sugar is ____.
Nitrogenous bases pairs are:
RNA
Structure?
The sugar is ____.
Nitrogenous bases pairs are:
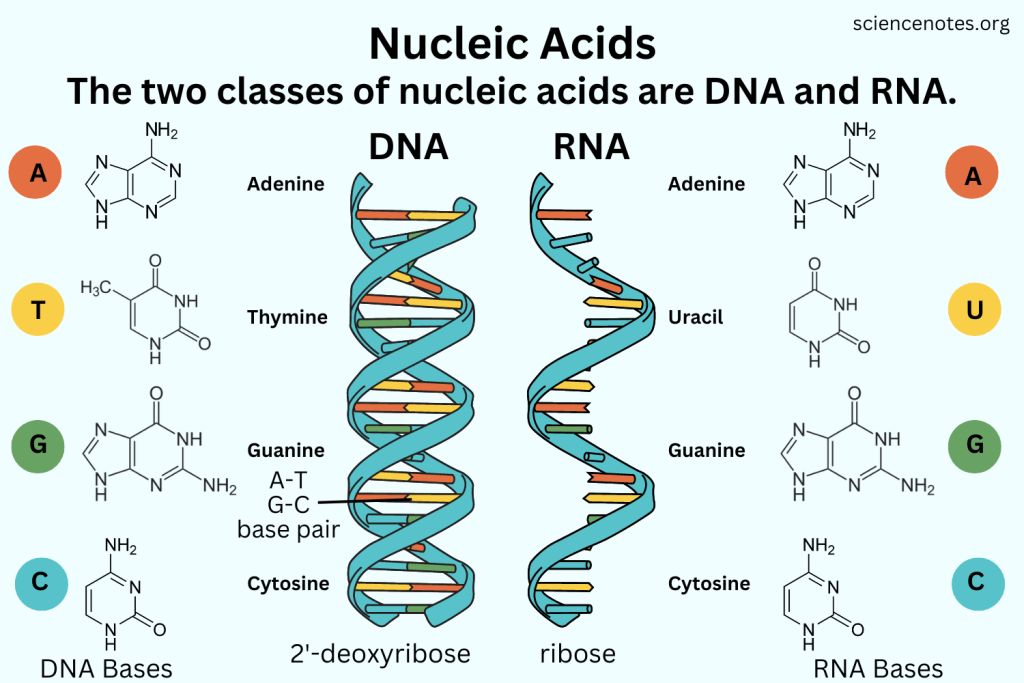
Nucleic acids (polymers) are made up of nucleotides (monomers)
Nucleic acids carry the a cells’ instructions.
Can replicate itself.
Nucleotides are made up of:
Phosphate group
5-Carbon sugar
Nitrogenous base
Example:
DNA
A double-helix molecule that has antiparallel strands
The sugar is deoxyribose.
Nitrogenous bases include:
Adenine (purine) - Thymine (pyrimidine) (2 hydrogen bonds)
Cytosine (pyrimidine) - Guanine (purine) (3 hydrogen bonds)
RNA
A single (stranded) helix molecule
The sugar is ribose
Nitrogenous bases include:
Adenine - Uracil
Cytosine - Guanine