13. Distinction between Self and Non-Self: T Cell Receptor III
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
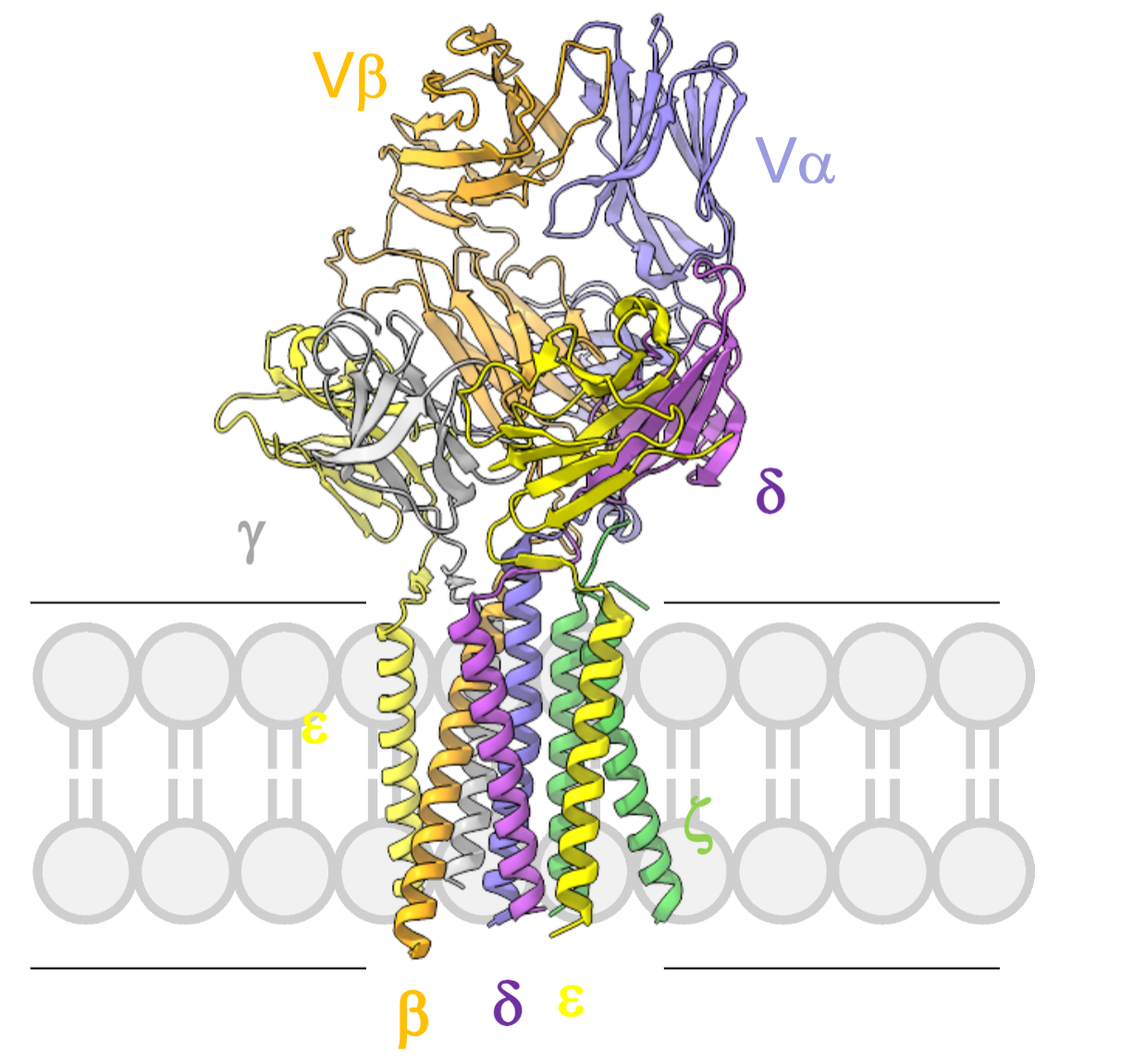
What did the cryo-EM structure of the TCR–CD3 complex reveal about the transmembrane helices?
All transmembrane helices (TCRα/β and CD3ε, γ, δ, ζ) form tight, intimate interactions, stabilising the complex.
TCR requires the full CD3 complex for surface expression and signal transduction.
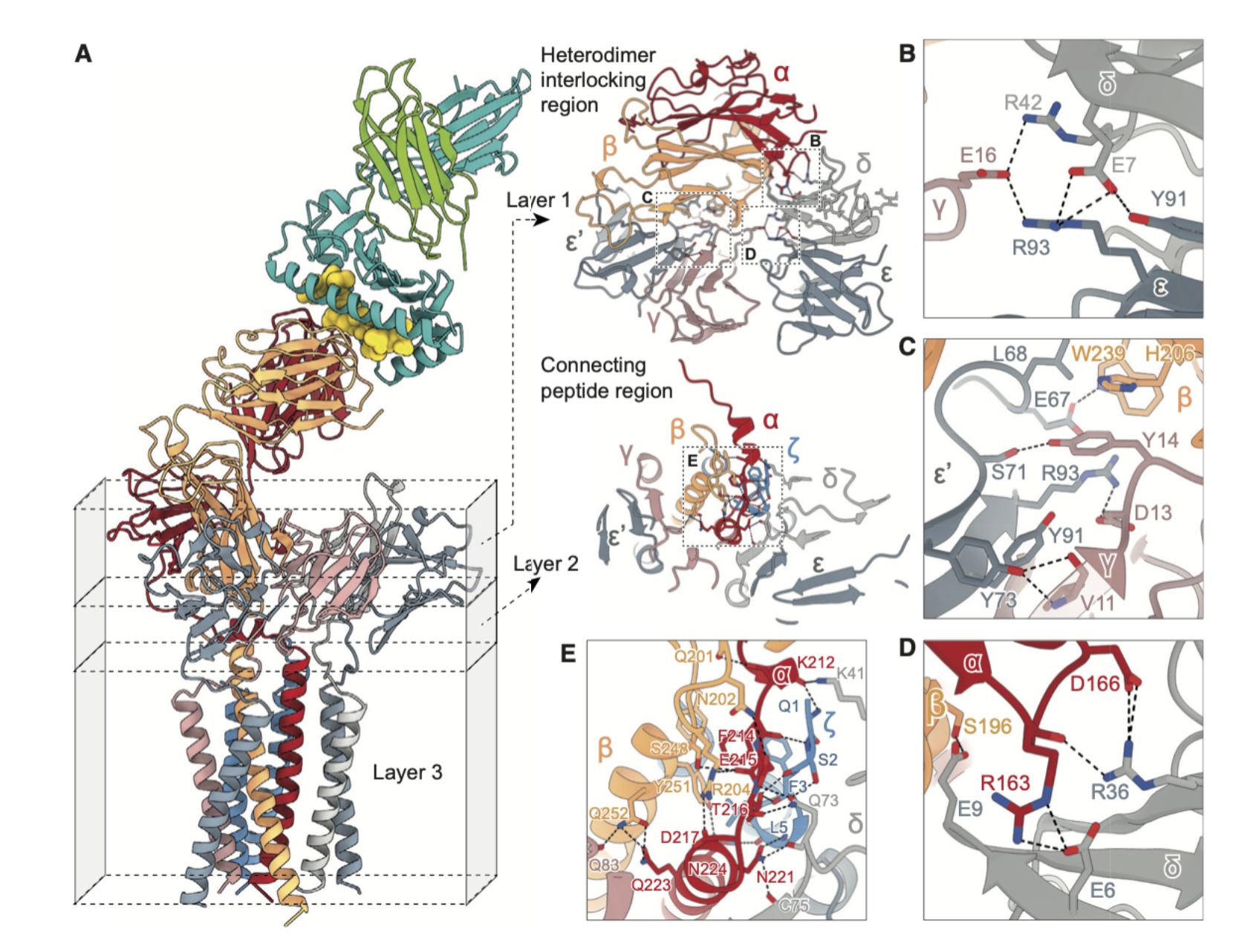
What additional structural features promote complex stability?
Covalent: Disulfide bonds (Cys–Cys linkages) between pairs of CD3 and αβ chains.
Non-covalent: Ionic interactions, H bonds promote stable inter-chain associations
This architecture is conserved across TCRs.
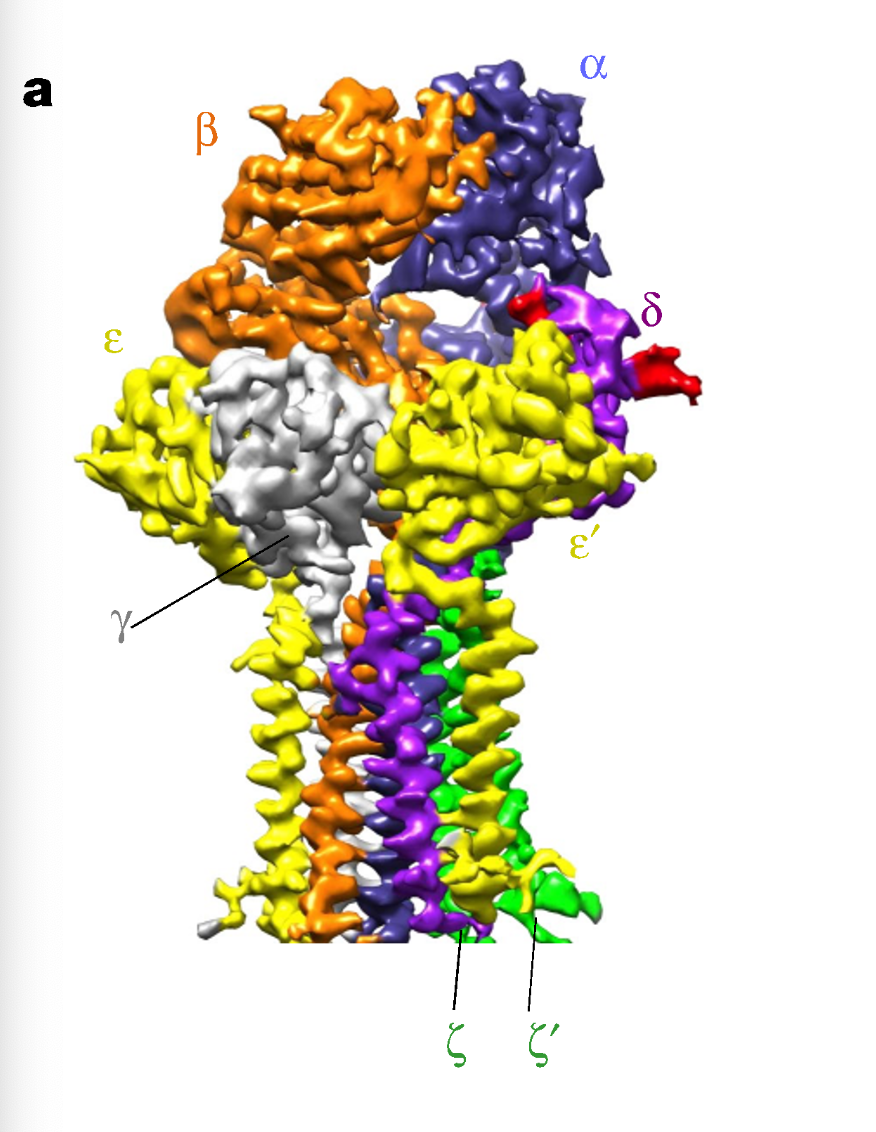
What can be observed in the cryo-EM structure of TCR–CD3 in the membrane?
The receptor assembles as a rigid, multi-subunit structure, with CD3 chains forming defined contacts, suggesting that signal transduction may involve mechanical or conformational changes initiated at the membrane.
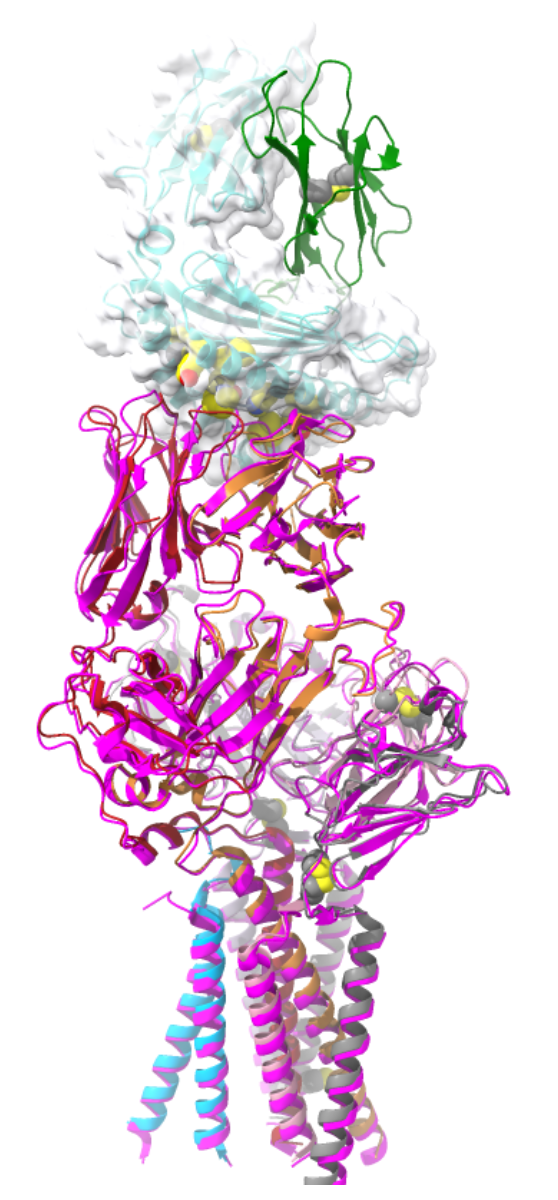
What does the superposition of pMHC-bound and unliganded TCR structures suggest?
The interaction is a rigid body docking: there are little to no large-scale conformational changes.
Allosteric changes are unlikely to transmit the activation signal.
Suggests TCR signalling is likely mechanosensitive, not driven by conformational change alone.
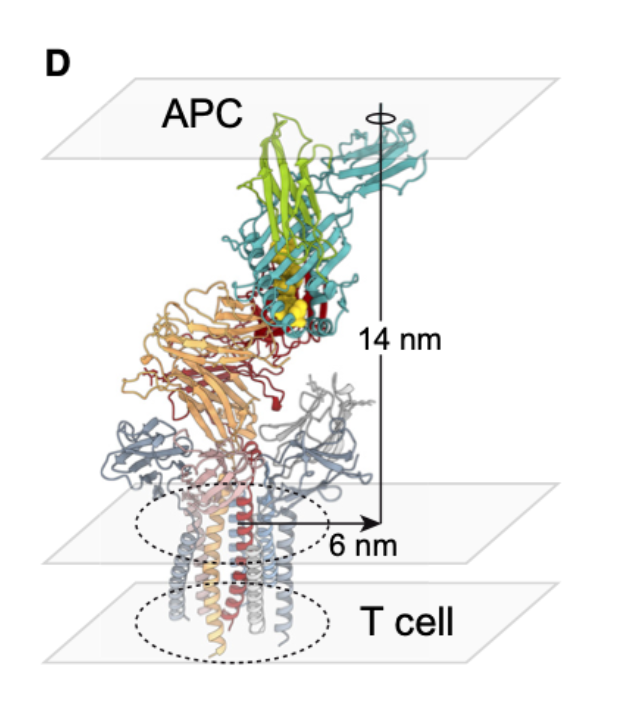
What is significant about the spacing at the TCR–APC interface, revealed by CryoEM?
~14 nm separation between membranes
TCR interaction radius ~1 nm
Suggests tight contact excludes large phosphatases like CD45 → creates a zone where signalling can proceed unopposed.
What is significant about the lateral mobility of TCR-pMHC elements within the membrane?
May relate to force generation and signal initiation through membrane mechanics.
What are the proposed models of TCR activation?
Aggregation Model
Segregation Model
Mechanosensor Model
Allosteric Model
Allostery + Mechanosensor hybrid Model
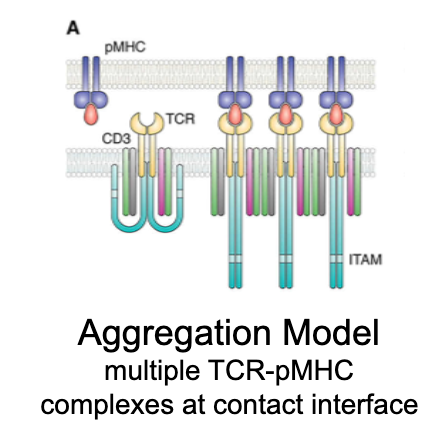
What is the aggregation model?
Activation occurs when multiple TCR–pMHC interactions cluster together, enhancing signal strength via cooperative binding.
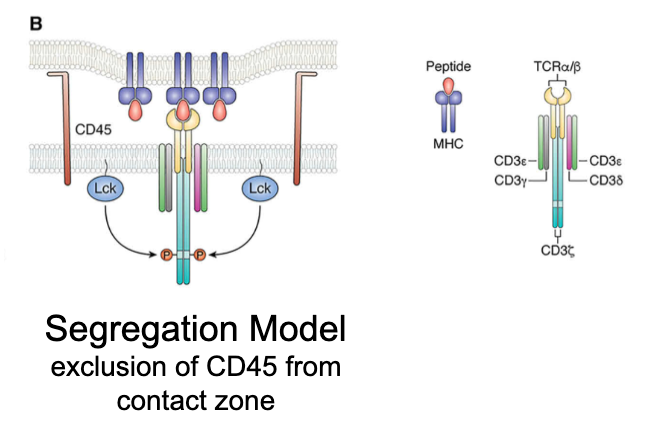
What is the segregation model?
Activation is driven by the exclusion of large molecules (e.g., CD45, a phosphatase) from the TCR–pMHC contact zone. Prevents dephosphorylation of CD3 ITAMs.
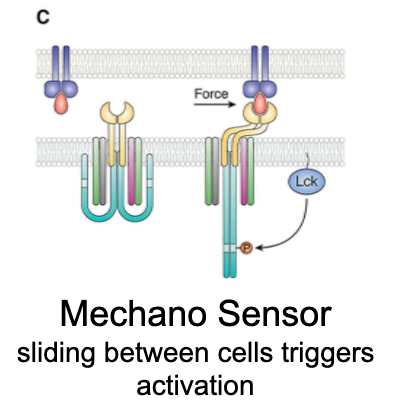
What is the mechanosensor model?
Sliding between cells triggers activation.
Mechanical force (e.g., shear from T cell–APC movement) unfolds CD3ζ chains from the membrane, thus ITAMs become exposed for phosphorylation and signalling.
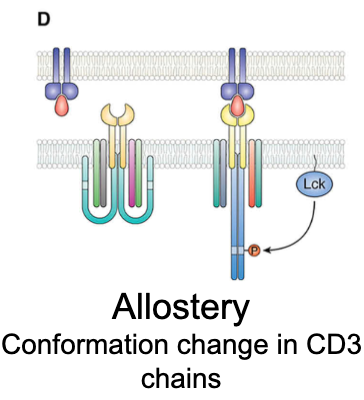
What is the allostery model of TCR activation?
Ligand binding induces conformational change in TCR/CD3 that transmits a signal allosterically. CD3 ITAMs become accessible without major structural rearrangement.
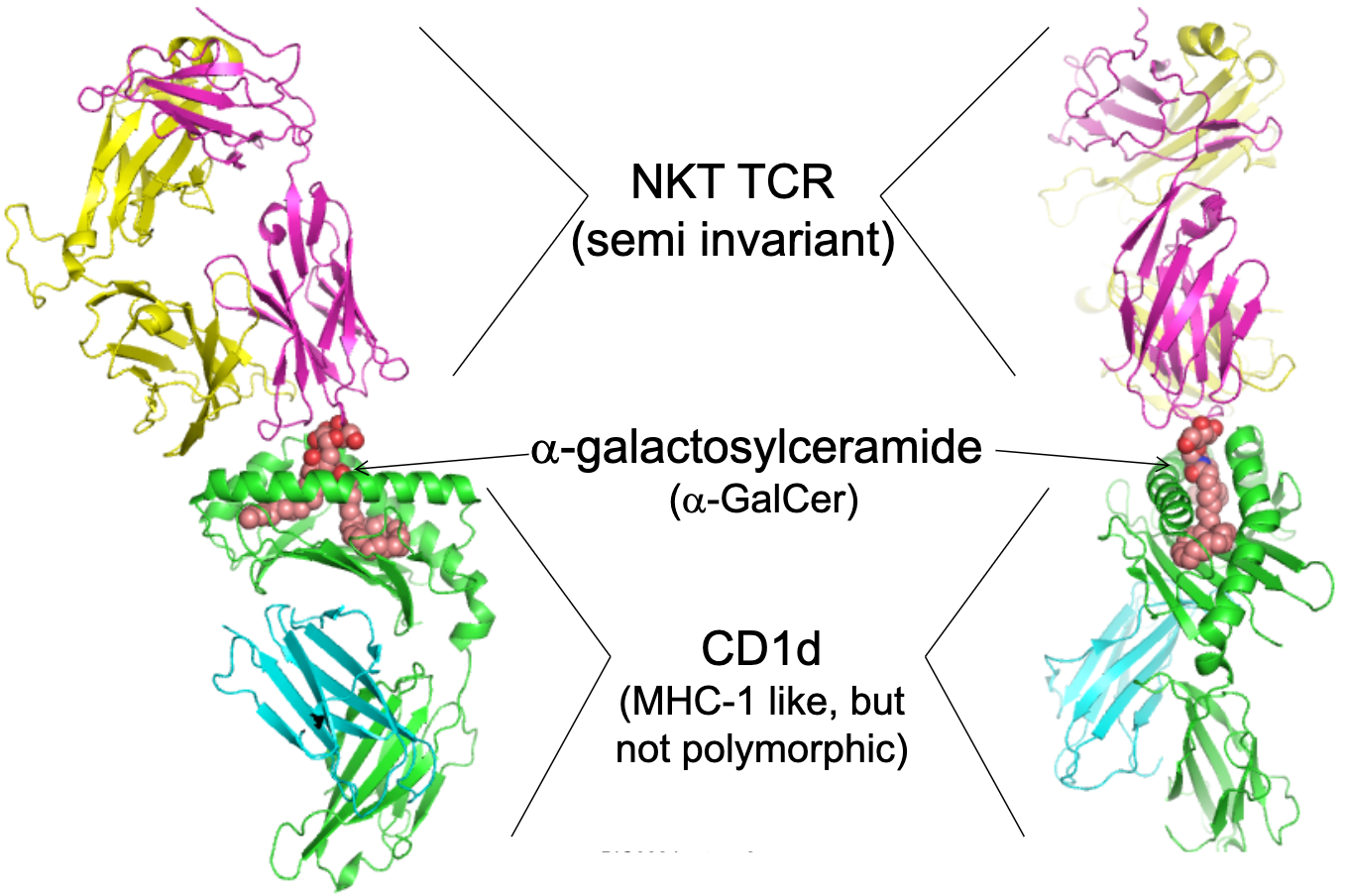
Give an example of a TCR recognising lipid antigens.
NKT cells: semi-invariant TCR.
Recognise α-galactosylceramide (α-GalCer) presented by CD1d (MHC-1-like, but not polymorphic).
TCR α-chain recognises galactosyl headgroup
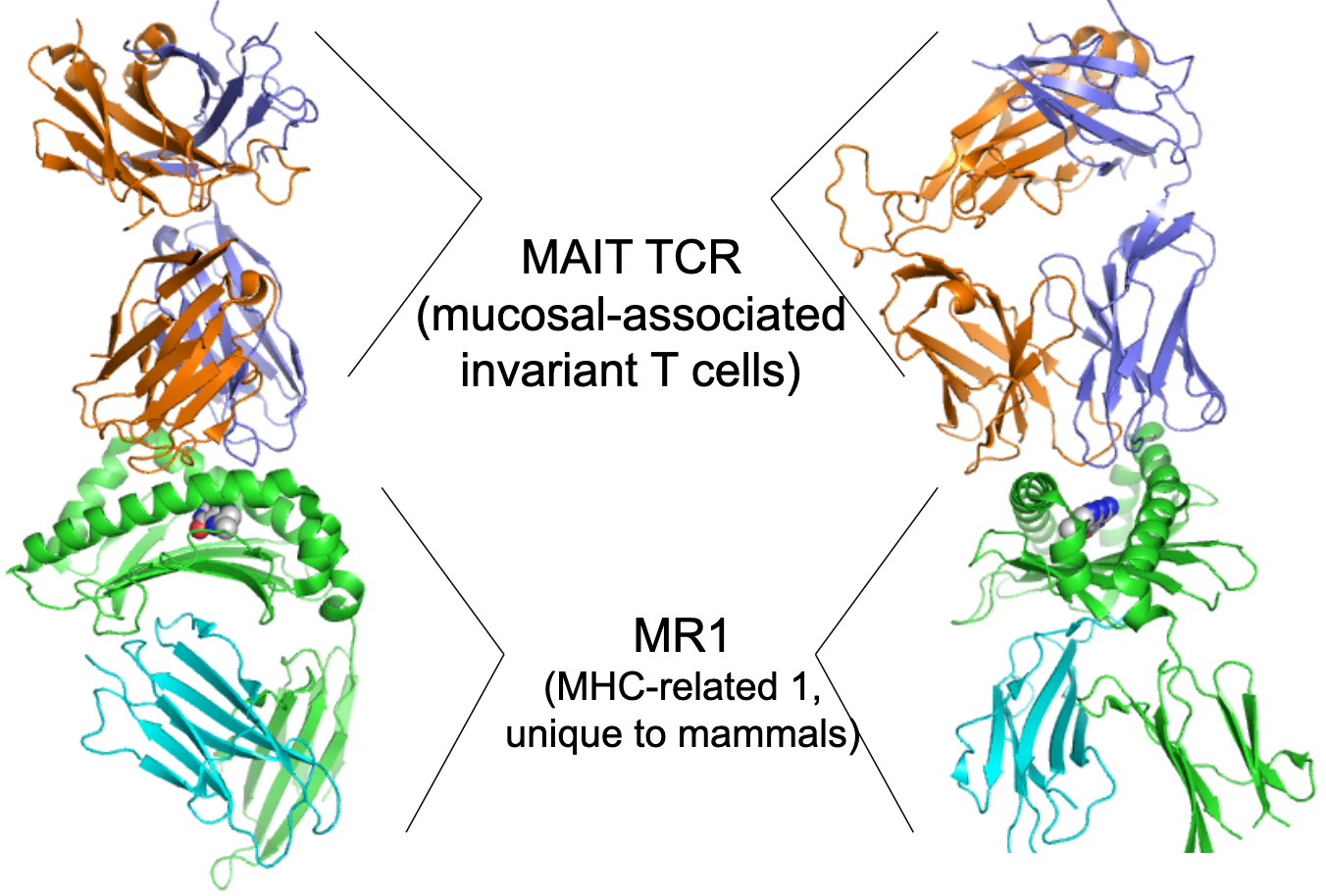
Give an example of a TCR binding small molecules.
MAIT (mucosal-invariant T cells) cells: semi-invariant TCR
Recognise riboflavin metabolites (VitB) presented by MR1 (MHC-related 1, unique to mammals), important for mucosal immunity
Why is the CD3 complex essential for TCR signalling?
Contains ITAMs (immunoreceptor tyrosine-based activation motifs)
CD3ζ alone contains 6 ITAMs, required for downstream activation of Lck, ZAP-70, and LAT signalosome
Why are there different multiple models of TCR activation?
TCR signalling is multifaceted. No single model explains all observations.
Likely involves a combination of the models.
Mechanosensing explains physical changes; allostery explains structural responses; segregation removes inhibitors; aggregation amplifies the signal.
Why are invariant TCRs important?
They allow rapid, innate-like responses to conserved microbial metabolites or lipids.