Topic 8 Chemical analysis
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What is an element?
Made up of only one atom
Pure
What is a compound?
Made up of two or more different types of atoms chemically bonded together
Pure
What is a mixture?
Made up of two or more different types of elements or compounds NOT chemically bonded together
Not pure
What is a pure substance?
A single element or compound that is not mixed with any other substance
How can pure elements and compounds be identified using physical properties?
Pure elements and compounds melt and boil at specific temperatures
Whereas impure substances melt and boil over a range of temperatures
Why is melting point and boiling point data useful?
It can be used to distinguish pure substances from mixtures
What does “pure” mean in everyday language?
A substance that has had nothing added to it (unadulterated) and in its natural state - e.g pure milk
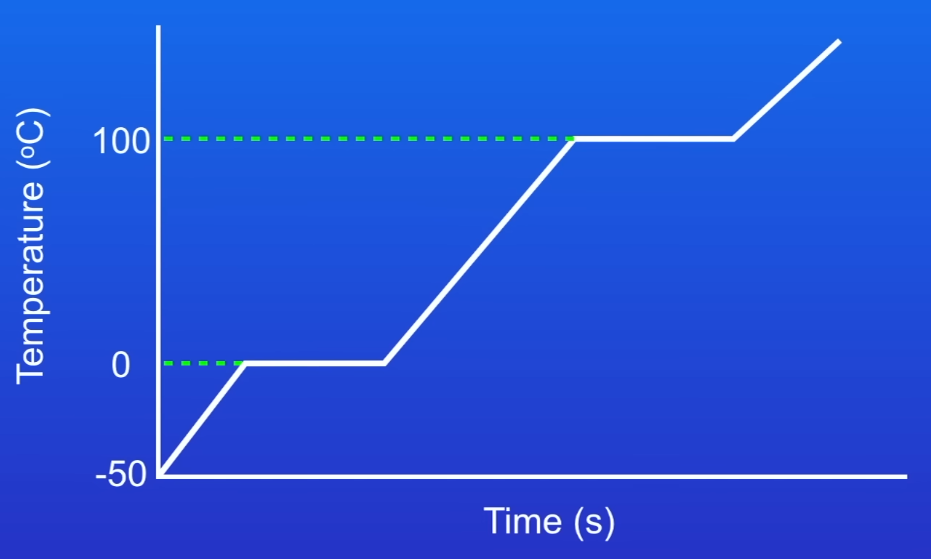
Why is this water pure?
Because both the melting and boiling points are at specific temperatures
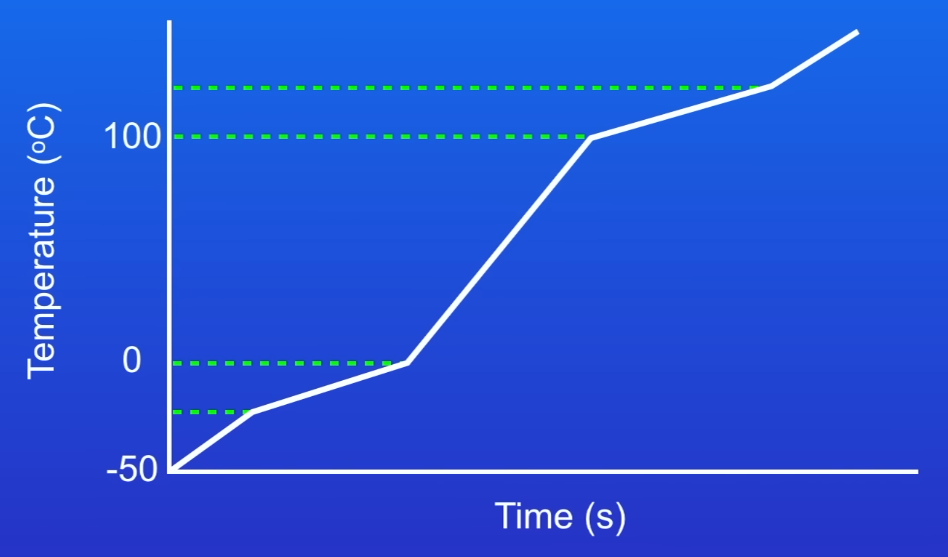
Why is this water impure?
Because the water melts and boils over a range of temperatures
What is a formulation?
A formulation is a mixture that has been designed as a useful product
In a formulation, the quantity of each component is carefully measured so that the product has the properties needed
Examples of formulations
Fuels
Cleaning prodcuts
Paints
Medicines
Alloys
Fertilisers
Foods
What is chromatography used for?
To separate mixtures based on their solubilities and provide information to help identify substances
What are the two phases involved in chromatography?
Stationary phase (does not move)
Mobile phase (moves through the stationary phase, carrying substances)
How does a pure compound appear on a chromatogram?
It produces a single spot in all solvents
How can chromatography distinguish pure from impure substances?
Impure substances produce multiple spots
Why do we draw the starting line in pencil?
If we drew the line in pen, the pen ink would move up the paper with the solvent
What determines how substances separate in chromatography?
The distribution of substances between the stationary and mobile phases
How is the Rf value calculated?
Rf = distance moved by substance / distance moved by solvent
Rf values do not have a unit
How can Rf values help identify substances?
Different compounds have different Rf values in different solvents, so comparing Rf values with known data can help identify them
Other factors to consider in paper chromatography
Several different substances could have the same Rf value. We need to repeat the experiment using a different solvent
If this substance has never been analysed before, then there will not be an Rf value on the database
Chromatography practical
1) Draw a pencil baseline 2 cm from the bottom of chromatography paper and mark spots for samples
2) Use a capillary tube to place small spots of known substances and the unknown on the line
3) Pour solvent (e.g water) into a beaker so the depth is below the baseline
4) Attach the paper to a glass rod using tape and lower the paper in the beaker so the bottom touches the solvent but the spots stay above it - the sides of the paper must not touch the walls of the beaker
5) Cover the beaker with a lid and allow the solvent to travel up the paper (do not move the beaker)
6) When the solvent is about ¾ up the paper, remove it and mark point where the water reached with a pencil (solvent front)
7) Let the paper dry, then compare the spots
8) Measure distances to calculate Rf values to find chemicals in the colours
Why must the solvent be above the baseline?
Because the samples would dissolve directly into the solvent causing the dyes to wash off the paper instead of separating
Why must the paper not touch the sides of the paper?
Because the solvent would not be able to travel evenly up the paper causing separation to become uneven and results are less reliable
How do you test for hydrogen gas and what is the result?
A test tube contains a gas which might be hydrogen
To test for hydrogen, insert a burning splint into test tube
Hydrogen burns rapidly with a pop sound
How do you test for oxygen and what is the result?
A test tube contains a gas which might be oxygen
To test for oxygen, use a glowing splint and insert into a test tube of the gas
The splint relights in oxygen
How do you test for carbon dioxide and what is the result?
To test for carbon dioxide, use limewater which is an aqueous solution of calcium hydroxide
When carbon dioxide is shaken with or bubbled through limewater the limewater turns cloudy (milky)
What is limewater?
An aqueous solution of calcium hydroxide
How do you test for chlorine and what is the result?
To test for chlorine, insert damp litmus paper into the test tube
When damp litmus paper is put into chlorine gas the litmus paper is bleached and turns white