Western Blotting
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
what does western blotting quantify
proteins
What are the 5 steps of western blotting
Extract the total protein
Run a Bradford assay on BSA and the samples 3x. Then take the average and create a standard curve and find the concentration of the samples.
Dilute the samples accordingly. Heat them and mix them with SDS and Bmercaptoethanol. Then load the same amount of protein into each well of the SDS-PAGE gels. Do this 3x
Blotting/transfer step
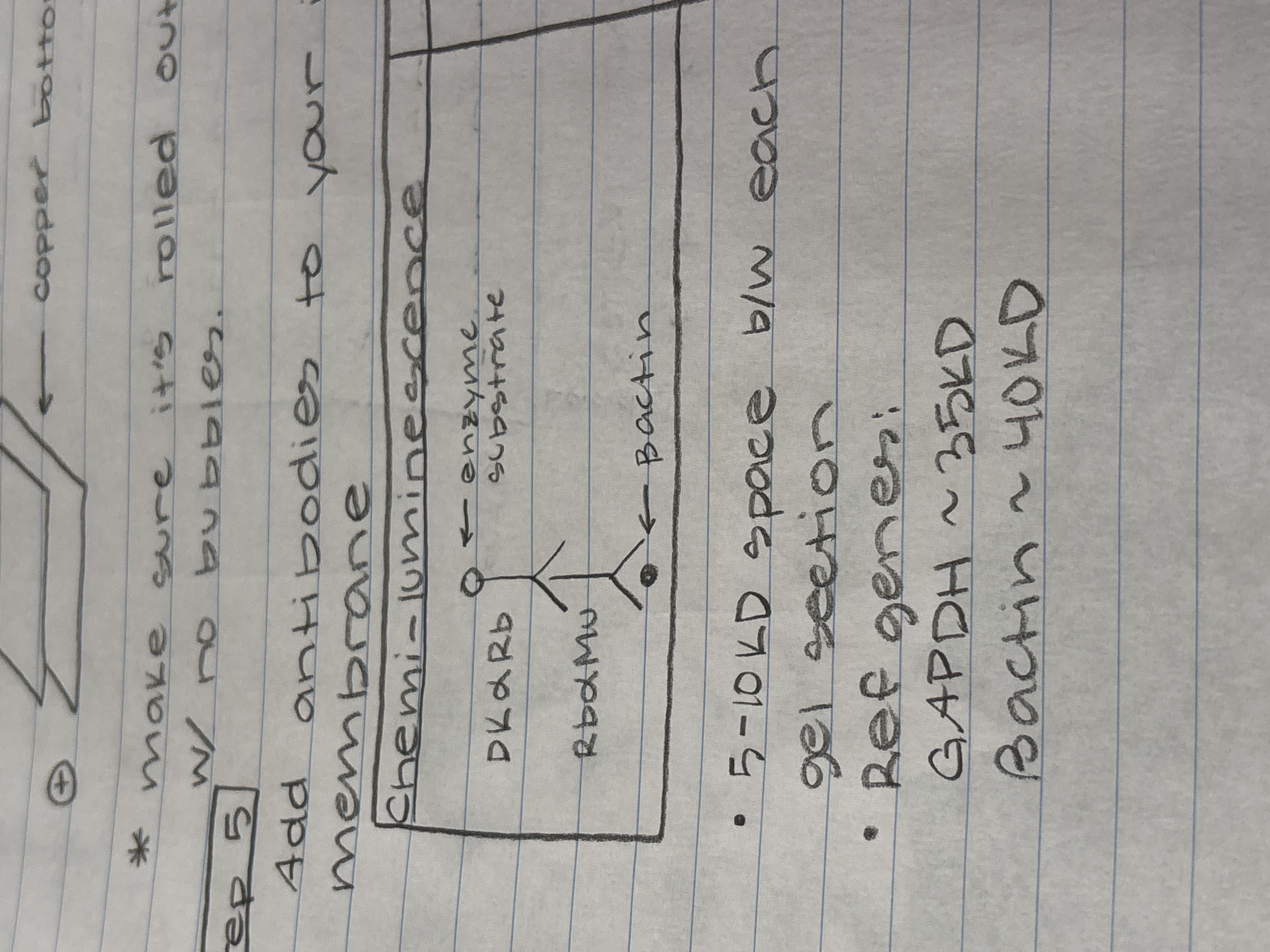
Add antibodies to your iblot membrane depending on the chosen method
Where do you extract the total protein sample from
A tissue or cell
what can you do if you extract the protein and it’s not highly expressed
concentrate it
what does BSA stand for
Bovine serum albumin
What kind of dilution do you run on the BSA for your Bradford assay
Serial dilutions
Describe the Bradford assay step for BSA
(Draw it)
Get a known [BSA] and do serial dilutions 3x
Take the ABS of each
Make a standard curve using the average
Describe the Bradford assay step for your samples
(Draw it)
Dilute your samples if needed so that they would likely fall in the linear range
Run the assay 3x
Take the ABS of each
Take the average ABS
Plug in the ABS into the linear equation to get the concentration
What went wrong if one of the ABS sample values is 1.3
You didn’t dilute the sample enough
What went wrong if one of your BSA ABS values for 0.3 is 1?
You made a mistake in your serial dilutions. Could be a miscalculation or you didn’t dilute it enough.
If the linear equation is y=.25x+.2
And your ABS sample values are
S1 = 0.72
S2 = 0.5
S3 = 0.36
What are their concentrations?
S1 = 2.88
S2 = 2
S3 = 1.44
What does SDS - PAGE gel stand for
Sodium dodecyl sulfate - polyacrylimide gel electrophoresis gel
Using the previous sample concentrations, what 2 things must you do before you’re able to load them into the gel
Dilute them all to the same concentration
Heat the and mix them with SDS and Bmercaptoethanol
calculate how you must dilute each sample so that there is 5ug of total protein in each well
Why do you heat the total proteins and mix them with SDS and Bmercaptoethanol?
It results in the unfolding and coding of the subunits of the proteins with a negative charge
How many times should you run the protein samples in the gel
3x
What can go wrong during the gel loading step
You squirt the protein too fast and it overflows
A bubble gets in the gel well and pops, contaminating other wells
The current is too high and cooks the proteins
You don’t use gloves and contaminate the proteins
What is the order of the gel sandwhich in the blotting/transfer step
(Draw it)

What can go wrong during the blotting/transfer step
It’s not rolled out smoothly
There’s bubbles in it
draw the antibodies for finding Bactin with chemi-luminescence
draw the antibodies for finding Bactin with IR. What are 3 did IR spectrums you can use? What machine do you need for it?

What is an advantage to using IR over chemi-luminescence
You can stack 2 dif protein band readings on 1 blot.
What reference genes can be used and what’s their size?
GAPDH ~ 35kDa
Bactin ~ 40kDa
How much space do you typically need between gel selection
5-10kDa
What can go wrong while you’re taking pictures of your iblot
If you’re running multiple samples that you want to compare, you don’t use the same excitation emission and timed exposure
If you’re doing chemi-luminescence: you don’t let the enzyme sit long enough
You don’t run the exposure long enough
CAPA is an inhibitor of protein X. You want to test its affects on protein X (30kDa) expression. Describe how the experimental groups were setup. Describe your experimental groups. Draw your results.
Control: protein X without CAPA, N=3. Sample extracted from mouse that didn’t have CAPA.
Reference gene: Bactin (40kDa), N=3. Sample extracted from mouse that didn’t have CAPA.
Treated group: protein X with CAPA, N=3. Sample was extracted from mouse that was given CAPA via food 3x a day for a week prior to extraction.
Must include: ladder, reference gene, control group, treated group
CAPA is an inhibitor of protein X. You want to test its high vs low dosage affects on protein B (45kDa) expression. Describe how the experimental groups were setup. Describe your experimental groups. Draw your results.
Negative Control: protein B without CAPA, N=3. Sample extracted from mouse that didn’t have CAPA.
Reference gene: GAPDH (35kDa), N=3. Sample extracted from mouse that didn’t have CAPA.
Positive Control: protein B with CAPA, N=3. Sample was extracted from mouse that was given 10ug CAPA via food 3x a day for a week prior to extraction.
Low dose group: protein B with CAPA, N=3. Sample was extracted from mouse that was given 5ug CAPA via food 3x a day for a week prior to extraction.
High dose group: protein B with CAPA, N=3. Sample was extracted from mouse that was given 20ug CAPA via food 3x a day for a week prior to extraction.
Must include: ladder, reference gene, positive control group, negative control group, high dose, low dose