Oxidizing Methoxybenzyl alcohol to methoxy benzaldehyde using phase transfer catalysis
1/27
Earn XP
Description and Tags
OCHEM II LAB FINAL
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What is the oxidizing agent in this experiment?
sodium hypochlorite
What is the phase transfer catalyst?
tetrabutylammonium hydrogen sulfate
What is oxidation?
occurs when oxygen is added to or hydrogen is lost from an organic molecule
Chromate
CrO42-
used in acidic, aq condition
Turns primary OH —> carboxylic acid
Dichromate
Cr2O72-
used in acidic, aq conditions
turns primary OH —> carboxylic acid
Chromium Trioxide
CrO3
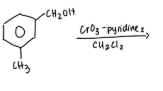
Collins Reagent
toxic complex composed of one molecule of CrO3 and two molecules of pyridine
contains no water
oxidize primary OH to aldehyde w/o further oxidation to a carboxylic acid
b/c no water used
all reagents are toxic and tertiary OH are not oxidized
What does KMnO4 do?
potassium permanganate
used in basic conditions to oxidize primary alcohols to potassium carbonate salts
strong acid is added (ex: dilute acid) —> carboxylic acid
also reacts w/ double + triple bonds so not used to oxidize an alcohol w/ those
not normally used to oxidize secondary alcohols to ketones b/c many ketones react further
Mn + 7 —>
MnO2 (Mn +4)
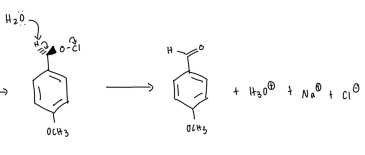
What does sodium hypochlorite do?
oxidizes aromatic + aliphatic secondary alcohols —→ ketones and primary alcohols to aldehydes
sub for collins reagent
primary aliphatic alcohols oxidize very slowly
selectively oxidizes secondary alcohols in the presence of primary alcohols
What is the main problem in this reaction?
starting alcohol is soluble in organic solvent
NaOCl is soluble in water, not in organic solvents
need to bring water soluble NaOCL and ethyl acetate soluble alcohol together to react
How can we bring NaOCl + ethyl acetate soluble alcohol together to react?
need to use phase transfer catalyst
tetrabutylammonium hydrogen sulfate
4 alkyl groups help make it soluble in ethyl acetate
TBAS is salt so water soluble
acts as a shuttle, moving back and forth between the aq. layer + organic layer
can exchange HSO4- counter ion for ClO- forming
TBAS transfers ClO- into ethyl acetate layer so it can react w/ alcohol
When using ethyl acetate what safety measures should be observed?
not using the chemical near flames or other heat sources must be observed
When using dichloromethane what safety measures should be observed?
wearing goggles and gloves must be observed as well as using the chemical in the fume hood
What does a disubstituted aromatic compound that has a 740 cm IR absorptions look like?
ortho
IR range for ortho is 735-770
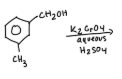
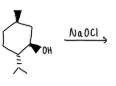
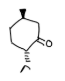
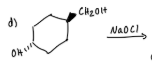
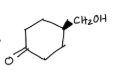
What is the purpose of this experiment?
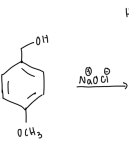
to oxidize methoxybenzyl alcohol to methoxybenzaldehyde using sodium hypochlorite and tetrabutylammonium hydrogen sulfate as the phase transfer catalyst.
helps turn a primary alcohol into an aldehyde
determine the starting alcohol as either ortho, meta, or para
What are the reactions?
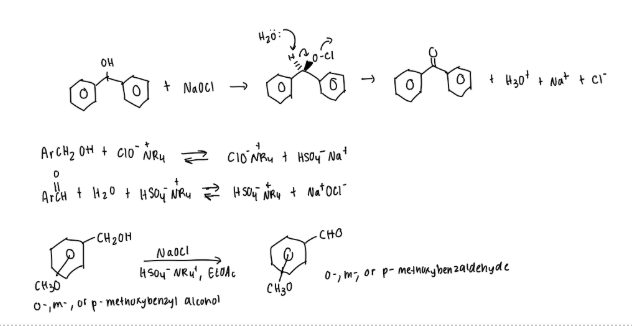
What are the steps of the experiment?
methoxy benzyl alcohol + ethyl acetate + TBAS into rbf w/ stir bar
bleach added slowly every 5 seconds while stir bar kept stirring
rxn stirred for 45 min —> two layers
solution added to sep. funnel w/ ethyl acetate, sat. NaCL, + ethyl acetate again —> both ether layers removed from the top
added to sep. funnel + washed w. 5% NaOH
extracted again with NaCl
ethyl layer into Erlenmeyer flask + dried/ MgSO4 then decanted into RBF + evaporated
The major peak in the aldehyde that is absent in the alcohol is ?
carbonyl at 1686.26 cm-1
The major peak in the alcohol that is absent in the aldehyde is ?
broad strong peak at 3330 cm-1 range
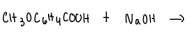
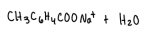
How can the crystals of methoxybenzoic acid can be isolated from the NaOH solution?
add heated HCl to neutralize solution
will reprotonate the solution which will form crystals
after solution is cooled, crystals will form and vacuum filtration can be used to isolate the crystals
How can the TLC be used to monitor the reduction of methoxybenzaldehyde to methoxybenzyl alcohol?
use NaBH4 as reducing agent
silica gel is polar and NaBH4 will increase bonds to hydrogen and decrease bonds to oxygen
methoxybenzyl alcohol will move a shorter distance due to polar nature