Chemistry - Metallic Bonding
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
Metallic bonding diagram
+ = cation. yellow dot = delocalised electron

What is a metallic bond?
An electrostatic attraction between metal cations and delocalised electrons
Physical properties of metals
High melting point → they are giant structures. We have to break many strong bonds (attraction between ions and electrons) which requires lots of energy
Conducts electricity (and heat) → the delocalised electrons are free to move throughout the whole structure
Malleable (can be hammered into shape) and ductile (can be drawn out into wires) → the layers of ions in the metal can slide past each other and in their new positions
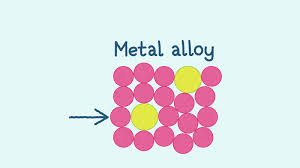
What are alloys?
A mixture of two or more elements (usually metals or carbon)
Alloy physical properties
Low melting points due to less strong bonds but are less malleable and ductile than pure metals because the different elements’ atoms are different sizes so it creates an interupted pattern. This means the layers cant slide, making it harder and stronger