Biology Exam I Test Questions
5.0(2)
Studied by 51 peopleCard Sorting
1/42
Earn XP
Last updated 12:28 AM on 2/8/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
1
New cards
Biosphere to ecosystems to communities to populations to organisms
What is the correct hierarchy of biological organization from large scale to smaller scale?
2
New cards
Changes in species toward greater complexity over time
\
Which of the following is Nor a necessary conditions for natural selection to occur within a population?
A. Variation in a trait in the population
B. The trait is heritable, at least to some extent
C. Changes in species toward greater complexity over time
D. Differential Reproductive success based on that trait
Which of the following is Nor a necessary conditions for natural selection to occur within a population?
A. Variation in a trait in the population
B. The trait is heritable, at least to some extent
C. Changes in species toward greater complexity over time
D. Differential Reproductive success based on that trait
3
New cards
Ancient pots used to hold milk are about the same age as the lactase-persistence mutations.
Which discovery supports the hypothesis that evolution of the lactase-persistence trait was driven By the use of milk in pastoralist cultures?
A. People who are lactase persistent have a mutation in the switch region of the lactase gene.
B. The lactase gene is present in both humans and domesticated animals.
C. Ancient pots used to hold milk are about the same age as the lactase-persistence mutations.
D. Scientists have discovered different mutations for lactase persistence in different cultures.
A. People who are lactase persistent have a mutation in the switch region of the lactase gene.
B. The lactase gene is present in both humans and domesticated animals.
C. Ancient pots used to hold milk are about the same age as the lactase-persistence mutations.
D. Scientists have discovered different mutations for lactase persistence in different cultures.
4
New cards
Lactose intolerant infants could not digest breast milk and therefore usually died, never having the chance to pass the mutation on to their children.
Most people are lactose intolerant as adults, but nearly everyone produces sufficient lactase as an infant. However, in some cases, genetic mutations can occur that make an infant lactose intolerant. From an evolutionary perspective --over thousands of years - why would lactose intolerance in infants be very rare in the population?
A. Lactose intolerant adults were not healthy enough to have children.
B. Lactose intolerance was such an embarrassing problem that many lactose intolerant parents chose not to have children, since the children might be lactose intolerant as well.
C. Lactose intolerant infants could not digest breast milk and therefore usually died, never having the chance to pass the mutation on to their children.
D. Infants who were lactose intolerant were intolerant of all other foods as well.
A. Lactose intolerant adults were not healthy enough to have children.
B. Lactose intolerance was such an embarrassing problem that many lactose intolerant parents chose not to have children, since the children might be lactose intolerant as well.
C. Lactose intolerant infants could not digest breast milk and therefore usually died, never having the chance to pass the mutation on to their children.
D. Infants who were lactose intolerant were intolerant of all other foods as well.
5
New cards
Heterozygous for the S (Sickling) Allele
In regions where malaria is prevalent, individuals who are _____ have a selective advantage.
6
New cards
Homozygous for the normal hemoglobin allele (two good copies)
For a woman living in the US, what genotype would be the most advantageous to have?
7
New cards
Superior fitness of heterozygotes in areas where malaria was present
The allele associated with sickle cell anemia apparently reached a high frequency in some human populations due to…
8
New cards
The melanin in darkly pigmented skin protects circulating folate from being destroyed by UV radiation, and folate is important in human reproduction
Darker skin is more prevalent in high-UV areas. In the video, Dr. Nina Jablonski proposed a hypothesis to explain the selective pressure for darker skin in these environments. On what evidence did she base this hypothesis?
9
New cards
Children born to parents with dark skin living far from the equator
Based on the risk factors discussed in the video and class, who would be most likely to develop the bone disease rickets?
10
New cards
Mouse coat color and the presence or absence of moonlight.
The graph data shows data from the light-colored soil enclosure. There is one dependent variable and more than one independent variable on the graph. What are the independent variables?
11
New cards
On light-colored soil with no moon
Under which conditions would a light brown mouse be most likely to escape predation at night?
12
New cards
No new pesticide used on the control field; new pesticide used only on the experimental field.
A farmer wants to improve crop yield in his fields by testing the effectiveness of a new pesticide available on the market. What would be the best control for this experiment?
13
New cards
100 times more H+
A solution at pH 6 contains ______ than the same amount of solution at pH 8.
14
New cards
Blood pH will decrease slightly
Carbon dioxide (CO2) is readily soluble in water, according to the equation CO2 + H20 + H2CO3. Carbonic acid (H2CO3) is a weak acid. Respiring cells release CO2 into the bloodstream. What will be the effect on the pH of blood as that blood first comes in contact with respiring cells?
15
New cards
An increase in the concentration of H2CO3 and a decrease in the concentration of HCO3-
Carbonic acid (H2CO3) serves as a buffer in human blood. Carbonic acid is a weak acid that dissociates into a bicarbonate ion (HCO3-) and a hydrogen ion (H+). Thus, H2C03 + HCO3-+ H+ A decrease in blood pH would cause the following to
happen…
happen…
16
New cards
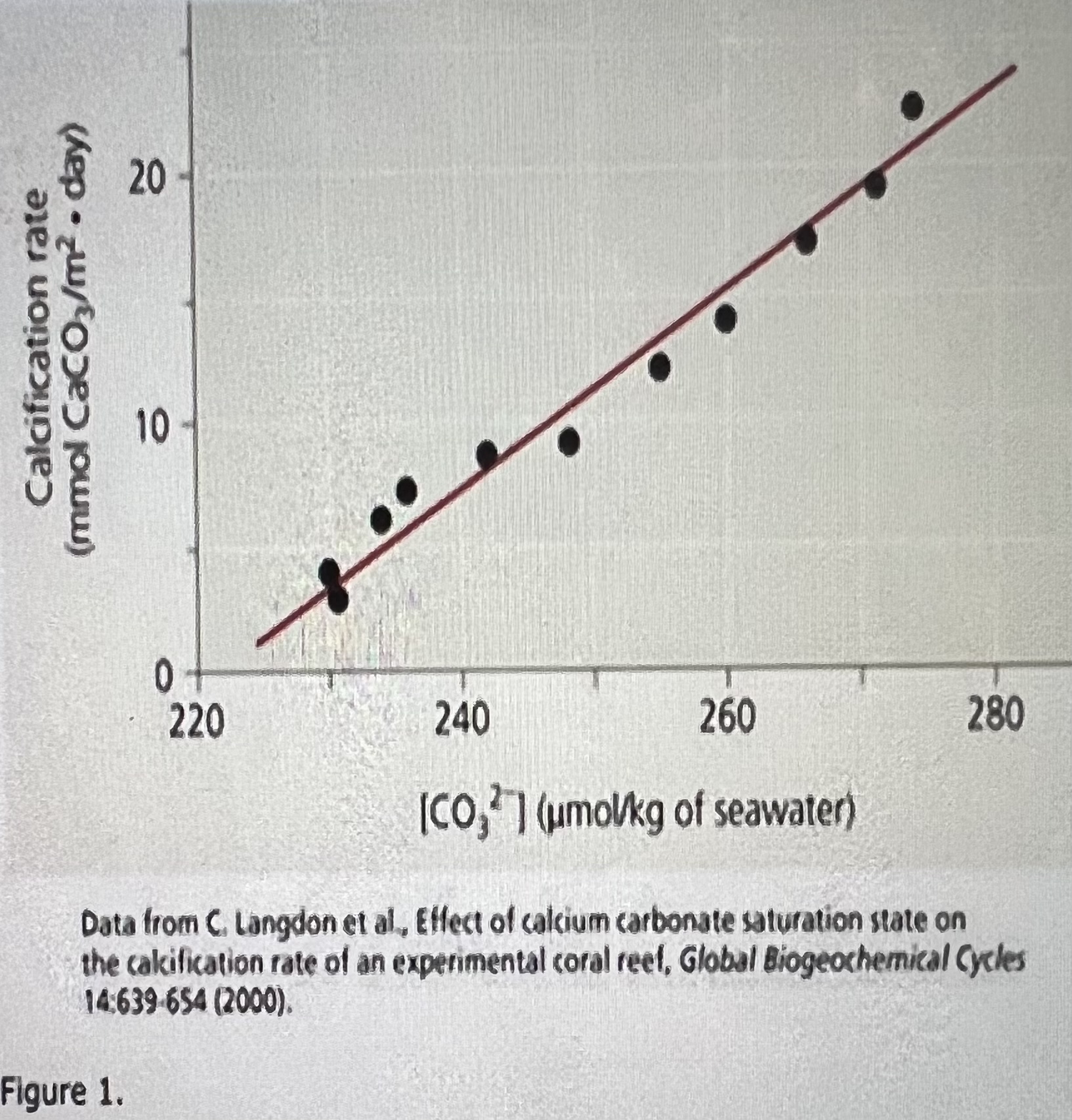
10 mmol CaCO3 per m2 per day; 3 days
How does the carbonate ion concentration of seawater affect the calcification rate of a coral reef?Scientists predict that acidification of the ocean, due to higher levels of atmospheric CO2, will lower the concentration of dissolved carbonate ions (COg 2-). Living corals use this (COg 2-) to build calcium carbonate (CaCOg) reef structures. For the next few questions, you will analyze data from a controlled experiment that examined the effect of different carbonate ion (COg 2-) concentrations in seawater on the rate of calcium carbonate (CaCO2) deposition by reef organisms into the
environment by a process called calcification.
Using the graph above, if the seawater carbonate ion (COg 2-) concentration is 250 umol/kg, what is the approximate rate of calcification, and approximately how many days would it take 1 square meter of reef to accumulate 30 mmol of calcium carbonate?
environment by a process called calcification.
Using the graph above, if the seawater carbonate ion (COg 2-) concentration is 250 umol/kg, what is the approximate rate of calcification, and approximately how many days would it take 1 square meter of reef to accumulate 30 mmol of calcium carbonate?
17
New cards
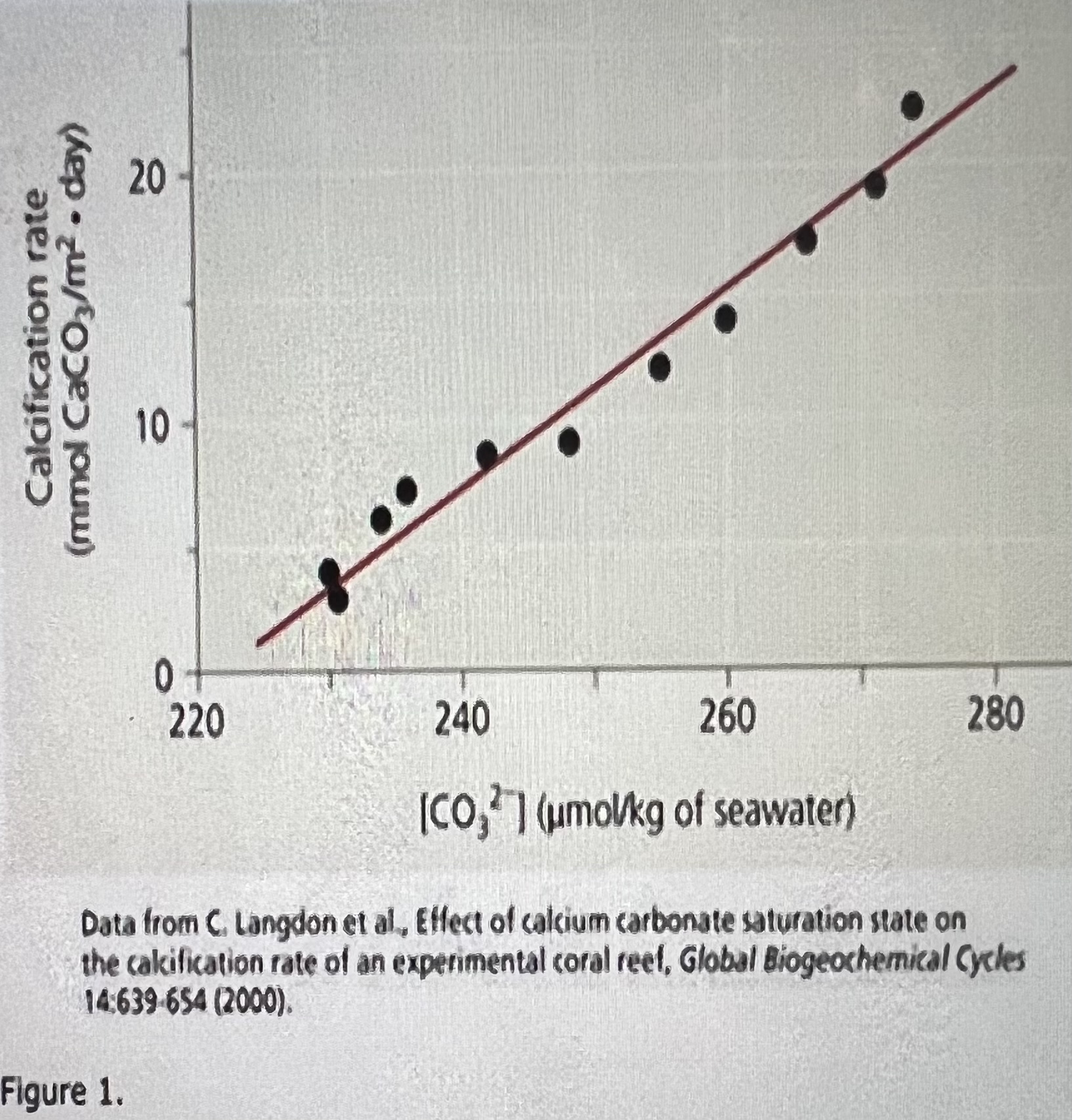
The rate of calcification decreases, and it takes longer for the reef coral to grow
Using the graph and information in the previous paragraph, describe what happens when carbonate ion concentration in seawater decreases?
18
New cards
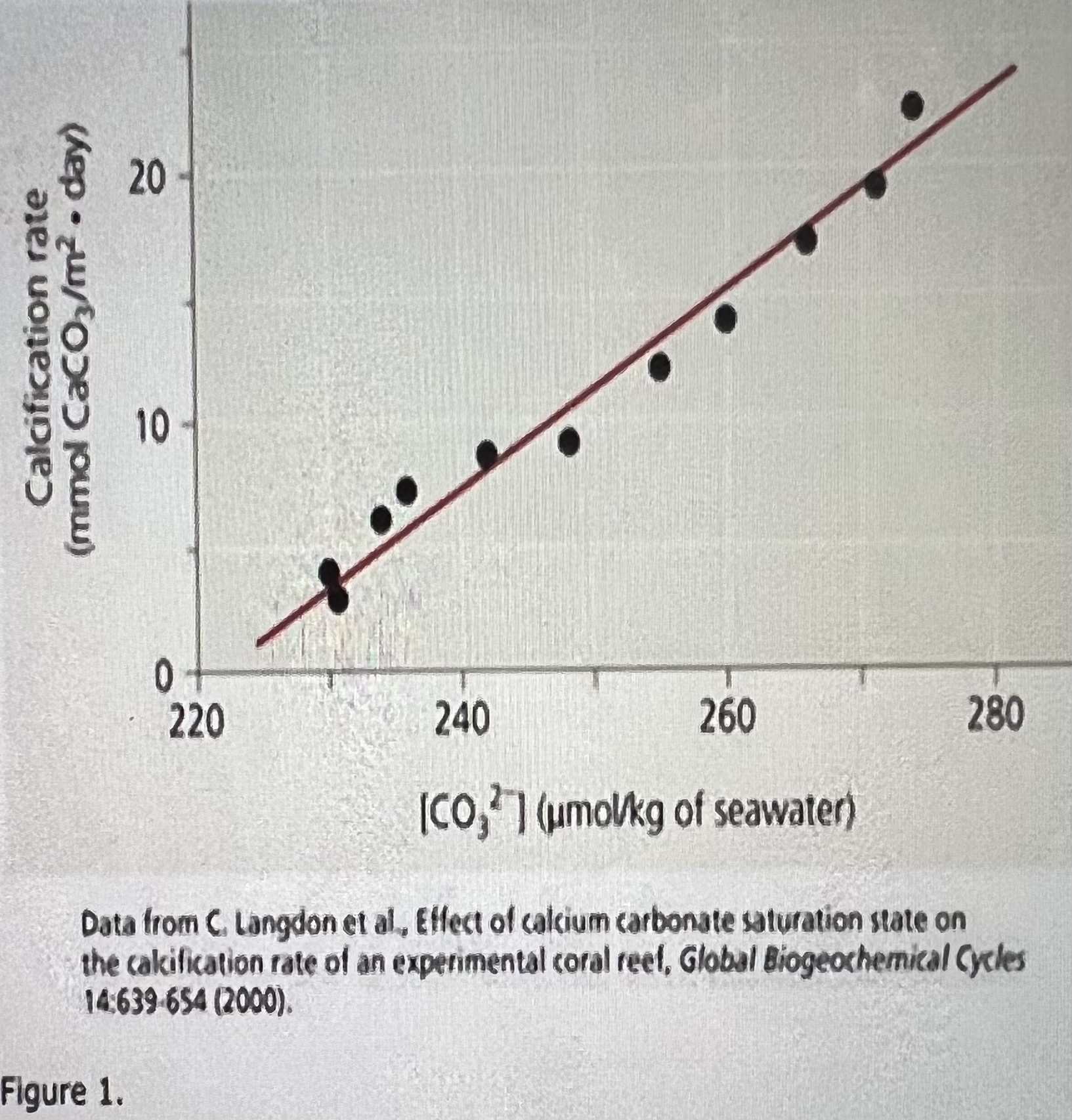
Yes; more CO2 dissolving into the oceans causes a decrease in the amount of CO3 2- in seawater, leading to a lower rate of calcification and slower reef growth.
Consider the hypothesis that increased atmospheric concentrations of COy will slow the growth of coral reefs. Do the results shown in the graph support that hypothesis? Why or why not?
19
New cards
Electron Microscopy (EM)
Which type of bioimaging technique is used to view the fine structures inside the chloroplast?
20
New cards
Large protein
Which of the following is visible ONLY with an electron microscope?
A. Eukaryotic cell
B. Prokaryotic cell
C. Mitochondrion
D. Large protein
A. Eukaryotic cell
B. Prokaryotic cell
C. Mitochondrion
D. Large protein
21
New cards
Volume increases faster the surface area
As cell size increases, the…
22
New cards
Larger cells would become limited by the ability to move materials across the plasma membrane
Over the course of evolutionary time, animal body sizes have gotten larger, yet cell sizes have not. Cells have stayed proportionally the same size to remain efficient, because…
23
New cards
Two times lower
If the radius of one cell were twice as large as the radius of another cell, how would its surface area to volume ratio change?
24
New cards
\
The need for surface area of sufficient area to support the cells metabolic needs
The need for surface area of sufficient area to support the cells metabolic needs
What is a major cause of the size limits for certain types of cells?
25
New cards
Allows them to perform several biochemical functions simultaneously
The compartmentalization of areas within eukaryotic cells…
26
New cards
An Endoplasmic reticulum
All of the following are part of a prokaryotic cell EXCEPT ____.
A. Ribosomes
B. An Endoplasmic Reticulum
C. A cell wall
D. DNA
A. Ribosomes
B. An Endoplasmic Reticulum
C. A cell wall
D. DNA
27
New cards
Block synthesis of proteins
Many antibiotics work by blocking the function of ribosomes. Therefore, these antibiotics will…
28
New cards
The lysosome
Tay-Sachs disease is a human genetic abnormality that results in cells accumulating and becoming clogged with very large, complex, undigested lipids. Which cellular organelle must be involved in this condition?
A. The lysosome
B. The Endoplasmic reticulum
C. Mitochondrion
D. Membrane bound organelles
E. The Golgi apparatus
A. The lysosome
B. The Endoplasmic reticulum
C. Mitochondrion
D. Membrane bound organelles
E. The Golgi apparatus
29
New cards
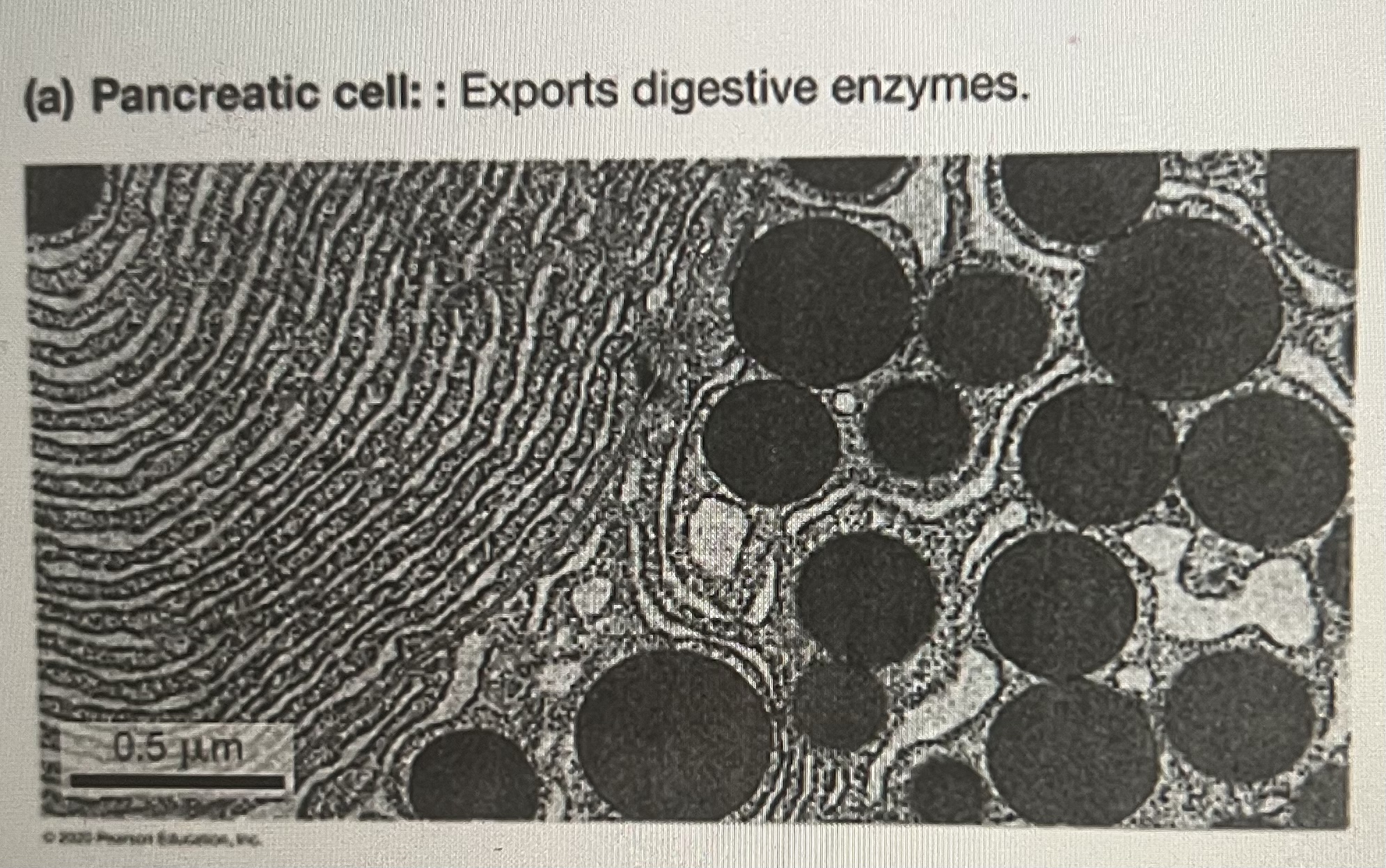
Secretory cells’ main function is to manufacture in secrete digestive enzyme proteins
Given that you can visualize an increased amount of Golgi apparatus in addition to lots of rough ER in the secretory cell from the pancreas shown in the figure below, what would you predict its function is?
30
New cards
Chloroplasts and mitochondria
A biologist ground up some plant leaf cells and then centrifuged the mixture to fractionate the organelles. Organelles and one of the heavier fractions could produce ATP in the light, whereas organelles in the lighter fraction could produce ATP in the dark. The heavier and lighter fractions are most likely to contain, respectively, ______.
31
New cards
Lysosomes
Asbestos is a material that was once used extensively in construction. One risk from working in a building that contains asbestos is the development of asbestosis caused by the inhalation of asbestos fibers. Cells will phagocytize (engulf) asbestos but I’m not able to degrade it. As a result, asbestos fibers accumulate in ______.
A. Peroxisomes
B. Ribosomes
C. Mitochondria
D. Lysosomes
A. Peroxisomes
B. Ribosomes
C. Mitochondria
D. Lysosomes
32
New cards
Abnormally shaped RBCs
Spherocytosis is a human blood disorder associated with a defective cytoskeletal proteins in the red blood cells (RBCs). What do you suspect is the consequence of such a defect?
33
New cards
Mitochondria
Which organelle is most abundant in cardiac muscle cells?
A. Mitochondria
B. Lysosomes
C. Golgi complexes
D. Plastids
A. Mitochondria
B. Lysosomes
C. Golgi complexes
D. Plastids
34
New cards
Lysosomes assist in the removal of defective organelles by digesting them.
How does a cell rid itself of defective or malfunctioning organelles?
35
New cards
Capture the energy of sunlight to meet metabolic demands
Mitochondria and chloroplasts share all of the following characteristics EXCEPT that they:
A. Are capable of ATP synthesis
B. Capture the energy of sunlight to meet metabolic demands
C. Possess their own DNA
D. Are surrounded by a double membrane
E. Contain ribosomes
A. Are capable of ATP synthesis
B. Capture the energy of sunlight to meet metabolic demands
C. Possess their own DNA
D. Are surrounded by a double membrane
E. Contain ribosomes
36
New cards
The movement of the lipids and proteins from the endoplasmic reticulum to the Golgi apparatus
Excel biologist has developed a new drug that will block the *cis* face of the Golgi apparatus. If eventually approved by the FDA, she wants to use this new drug to aid cancer patients. What specifically will this drug prevent from happening inside a cancer cell?
37
New cards
Rough ER to Golgi to Plasma membrane
A type of cell called a lymphocyte makes proteins that are exported from the cell. It is possible to track the path of this protein with the cell by labeling them with radioactive isotopes. What might be the path of a protein from the site where is polypeptides are made to the lymphocytes plasma membrane?
38
New cards
Researchers have been able to study the pathway of a secreted proteins by “tagging it with a fluorescent marker. Using this method, you would observe florescence moving from the ER to which organelle?
39
New cards
Insulin is carried in small sacs of membrane (vesicles) that moves from the endoplasmic reticulum to the Golgi apparatus and then to the plasma membrane
Insulin is a protein made in large amounts in the cells of the pancreas. These cells secrete insulin into the blood, where it controls the uptake of sugar by body cells. How is insulin transported from the ER to the surface of the cell?
40
New cards
The formation of the peptidoglycan
Penicillin is an antibiotic that kills or slows the growth of many kinds of bacteria and not affect humans. A mechanism that could explain its effectiveness could be that it interferes with…
41
New cards
Smooth endoplasmic reticulum
Cells can increase the number or size of some organelles in response to new demands. The amount of one organelle often is increased dramatically in the livers of alcoholics. Based on what you know of organelle function, this organelle is the…
42
New cards
Endoplasmic reticulum
In some diseases like cystic fibrosis, a cell membrane receptor fails to function. In the majority of cases, the problem comes from a change in the receptor so that it cannot reach the cell surface. The place in the cell where membrane proteins are synthesized and assembled builds up with the abnormal protein. This place would likely be the…
43
New cards
Preventing the sperm from swimming to the egg cell
A woman is having trouble becoming pregnant. Examination of her partners sperm indicates that microtubules are dysfunctional from the flagella in his sperm cells. A physician explains that this could interfere with fertility by…