Aldehydes and Ketones
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
aldehydes
RCHO (al)
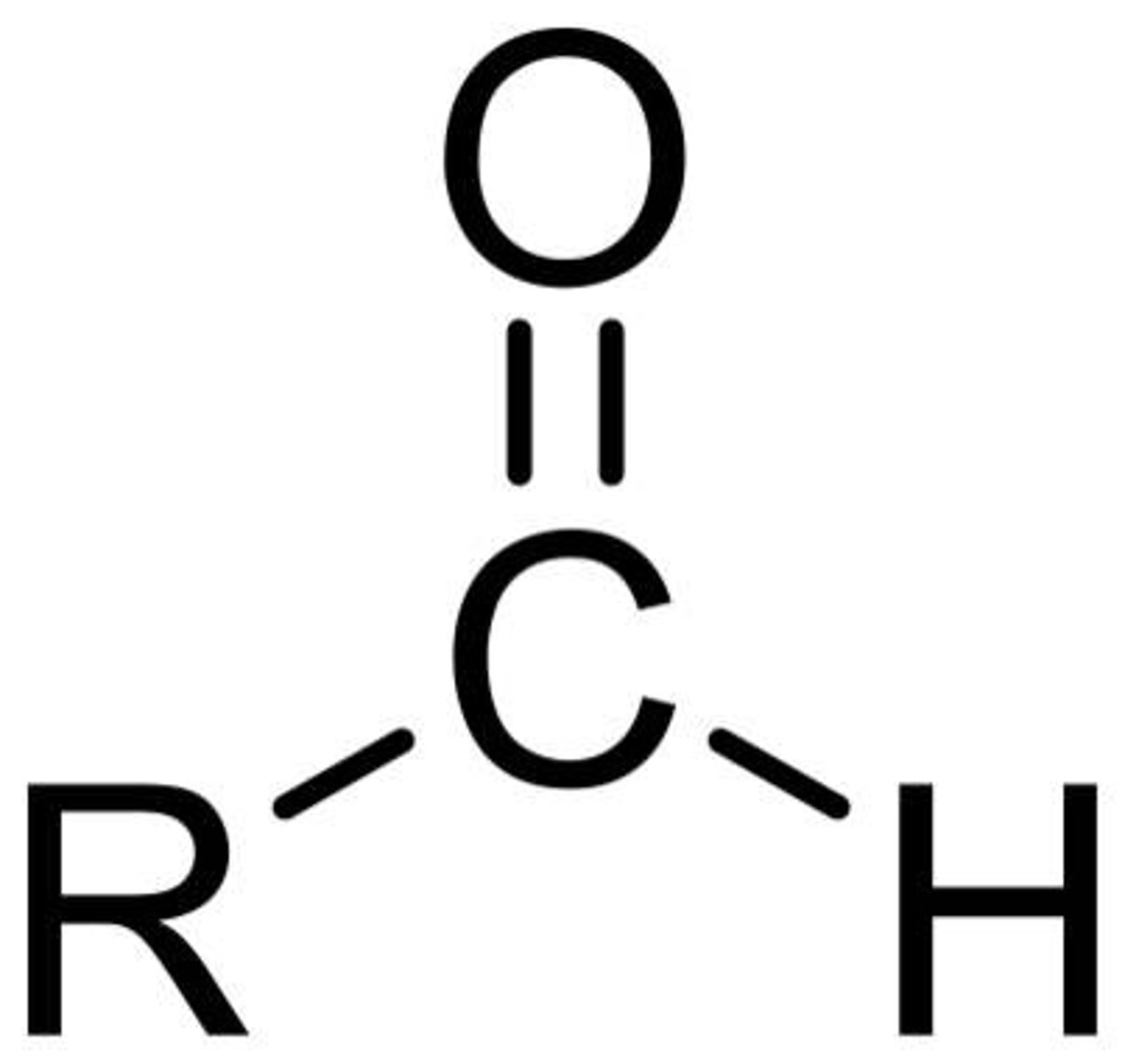
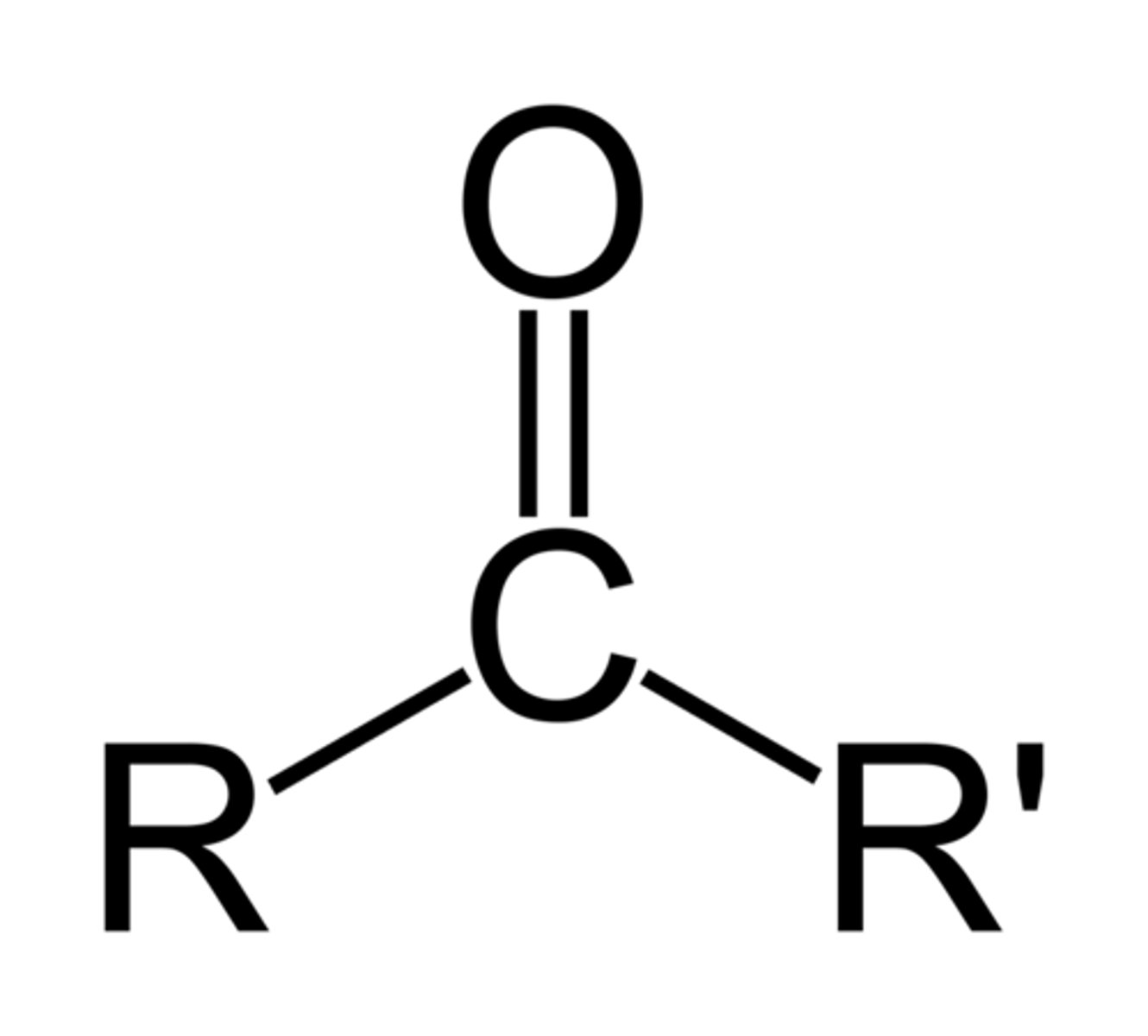
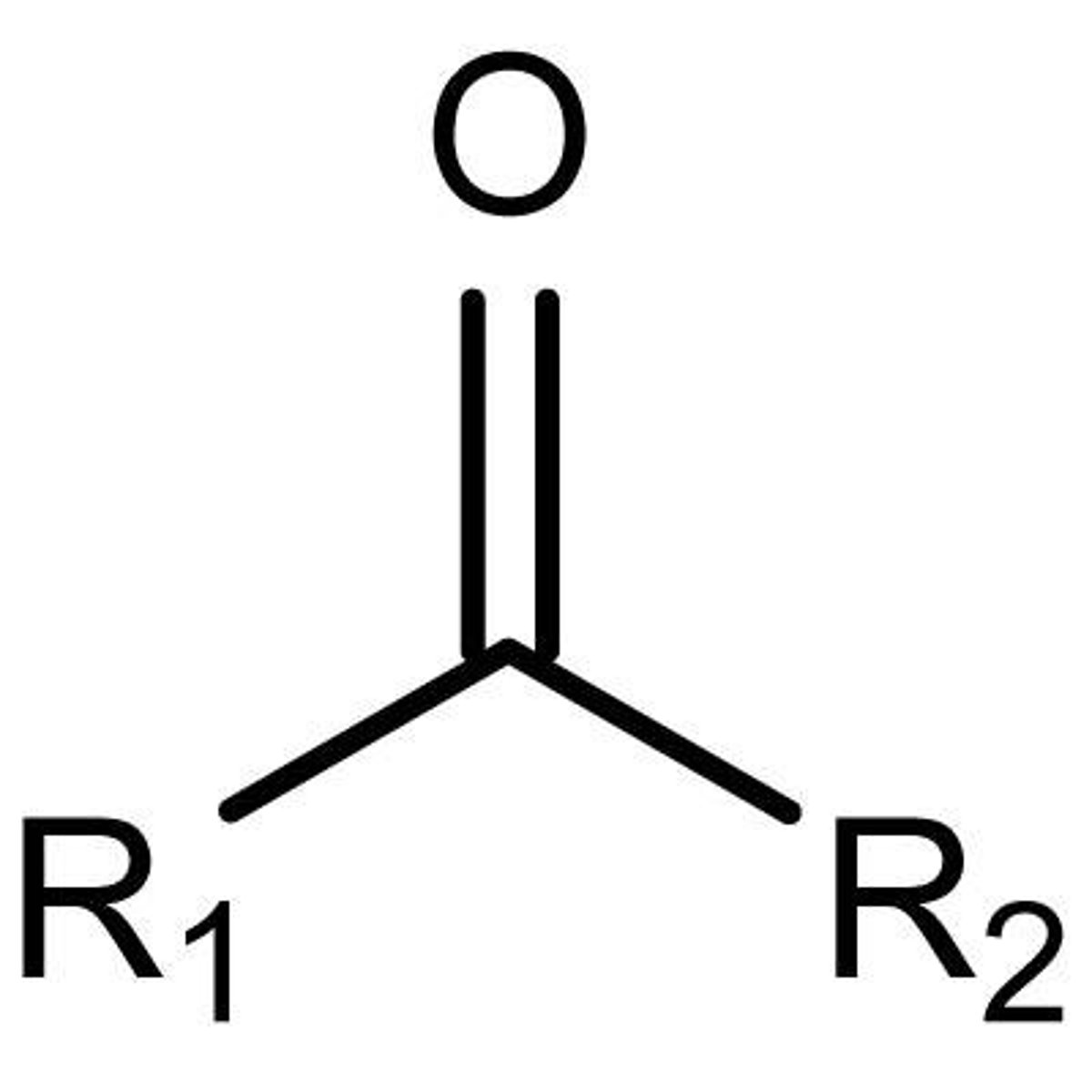
ketones
R-CO-R (one)
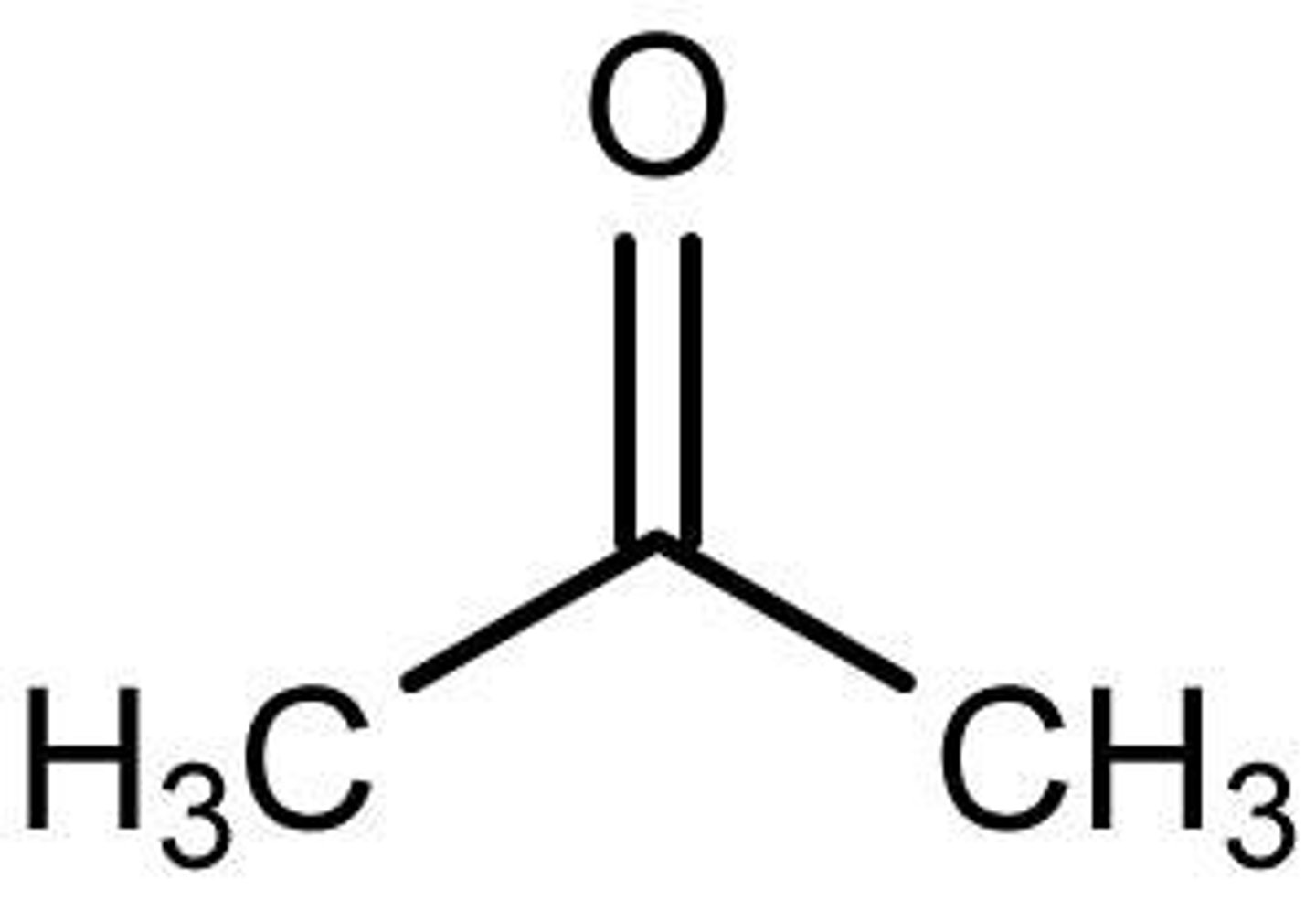
Acetone
CH3COCH3
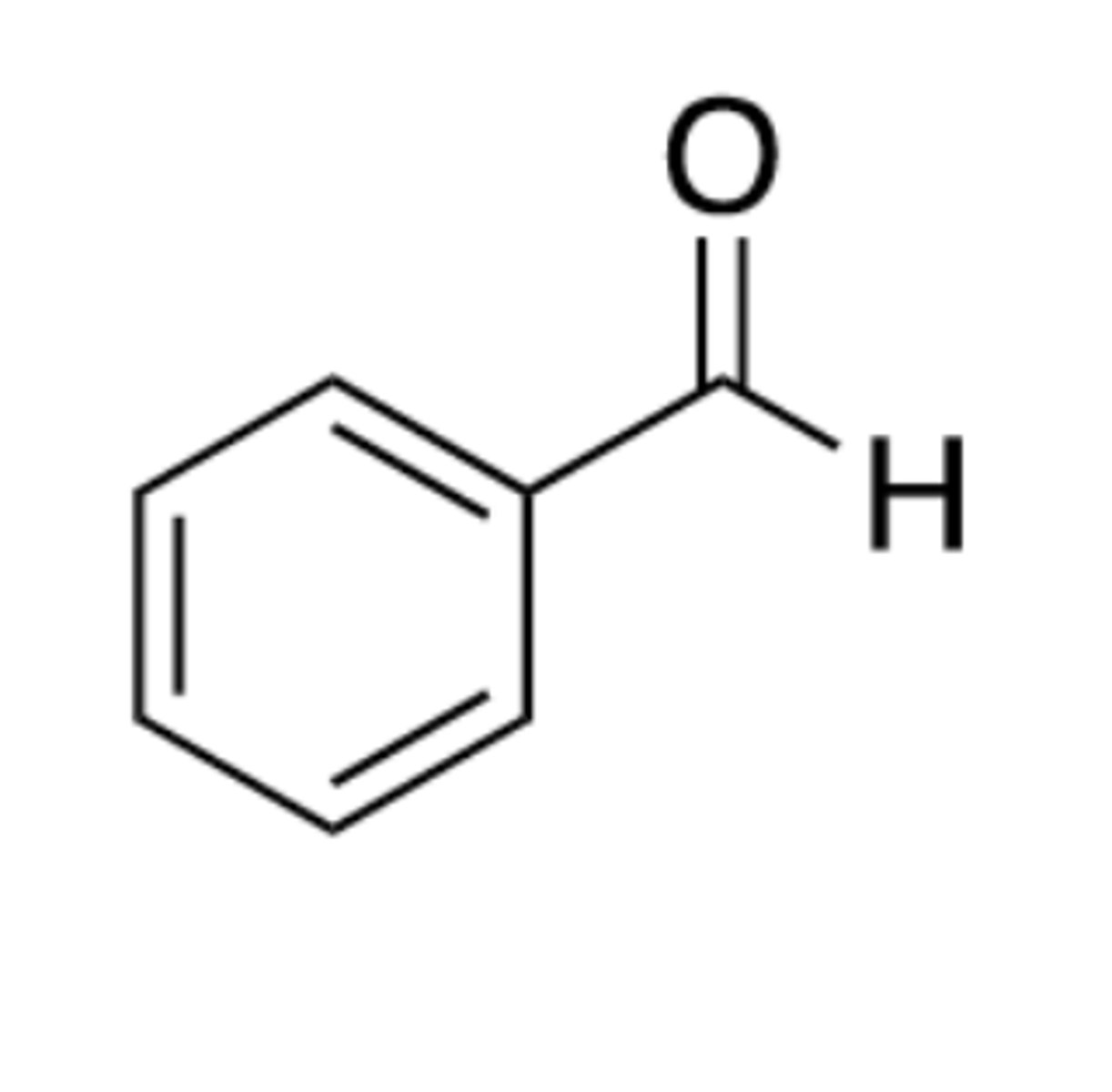
benzylaldehyde
Ph-CHO
which one is more reactive, aldehydes or ketones?
aldehyde
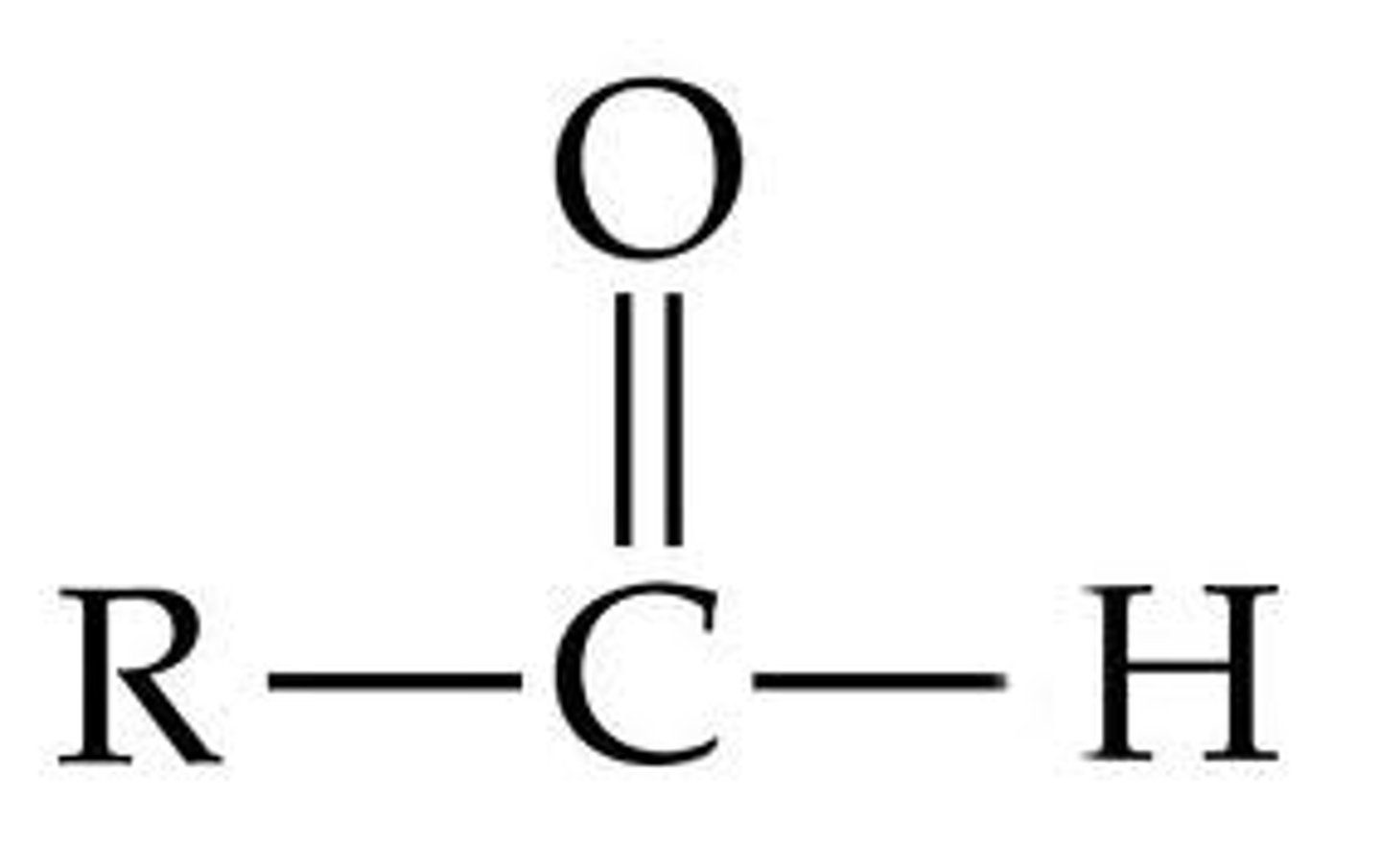
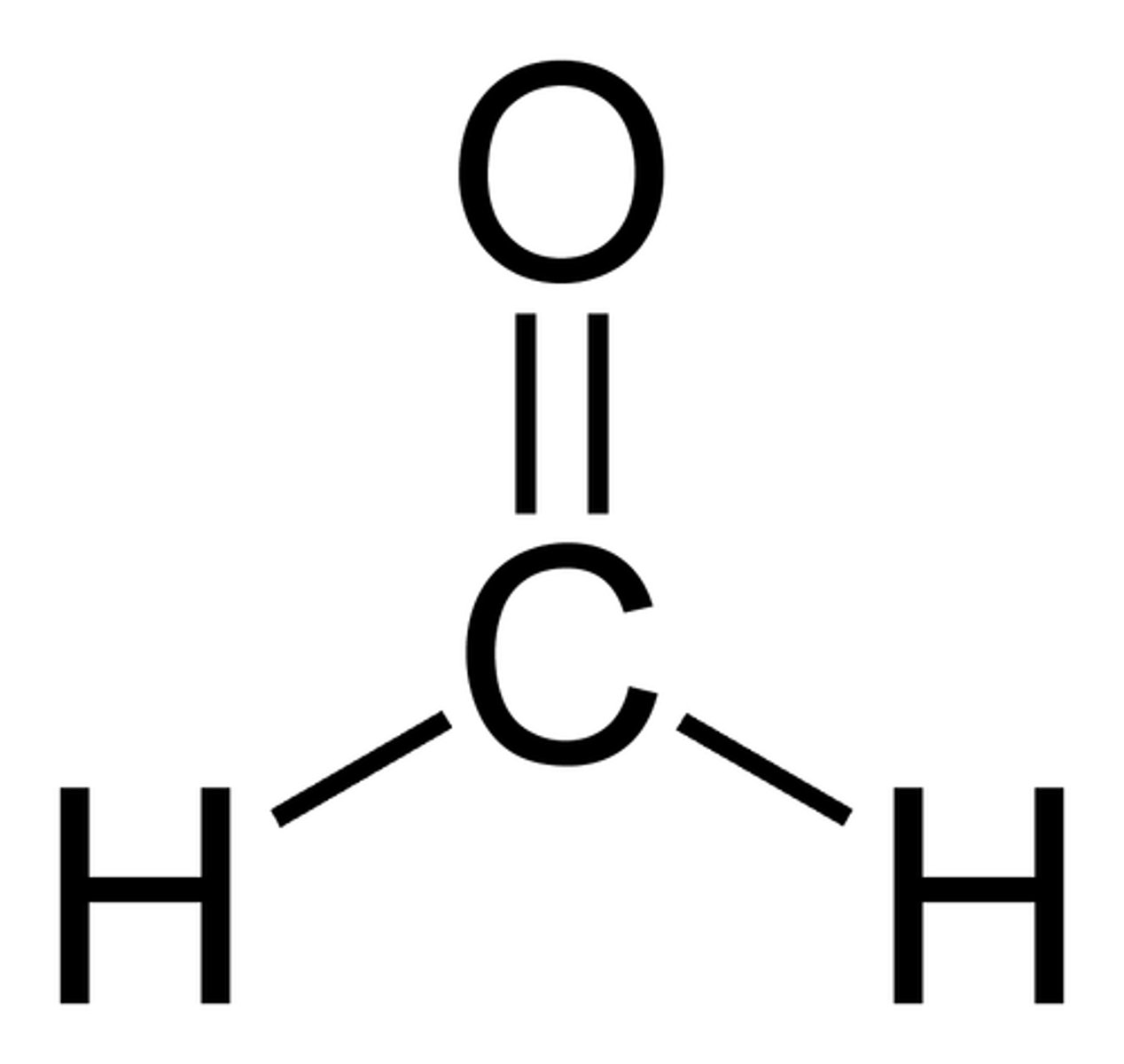
Formaldehyde
methanal
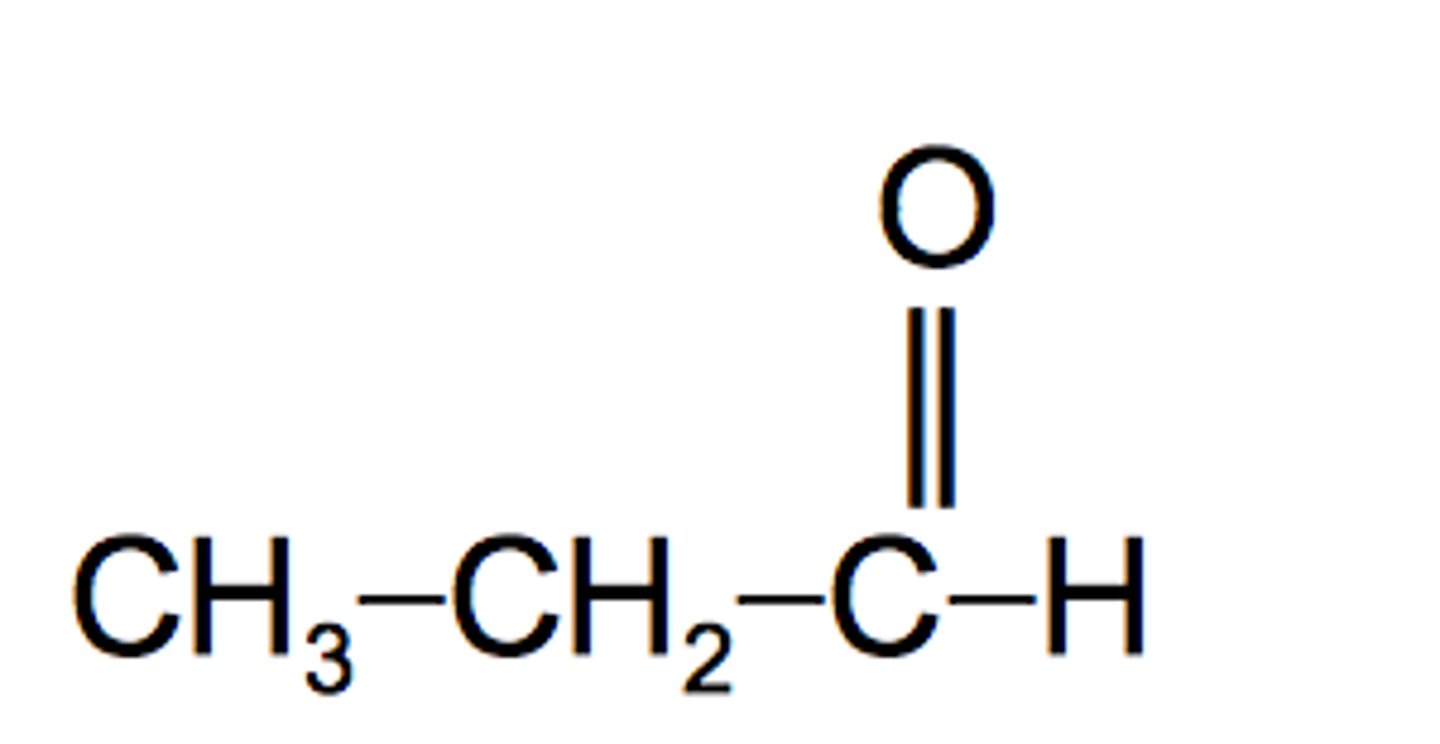
propionaldehyde
propanal
solubility in water lists
acids > alcohols > aldehydes + ketones > alkanes
how to synthesize aldehydes
oxidation of secondary alcohol
ozonolysis
oxymercuration
hydroboration
organocupurate/DIBAL-H + acid halides
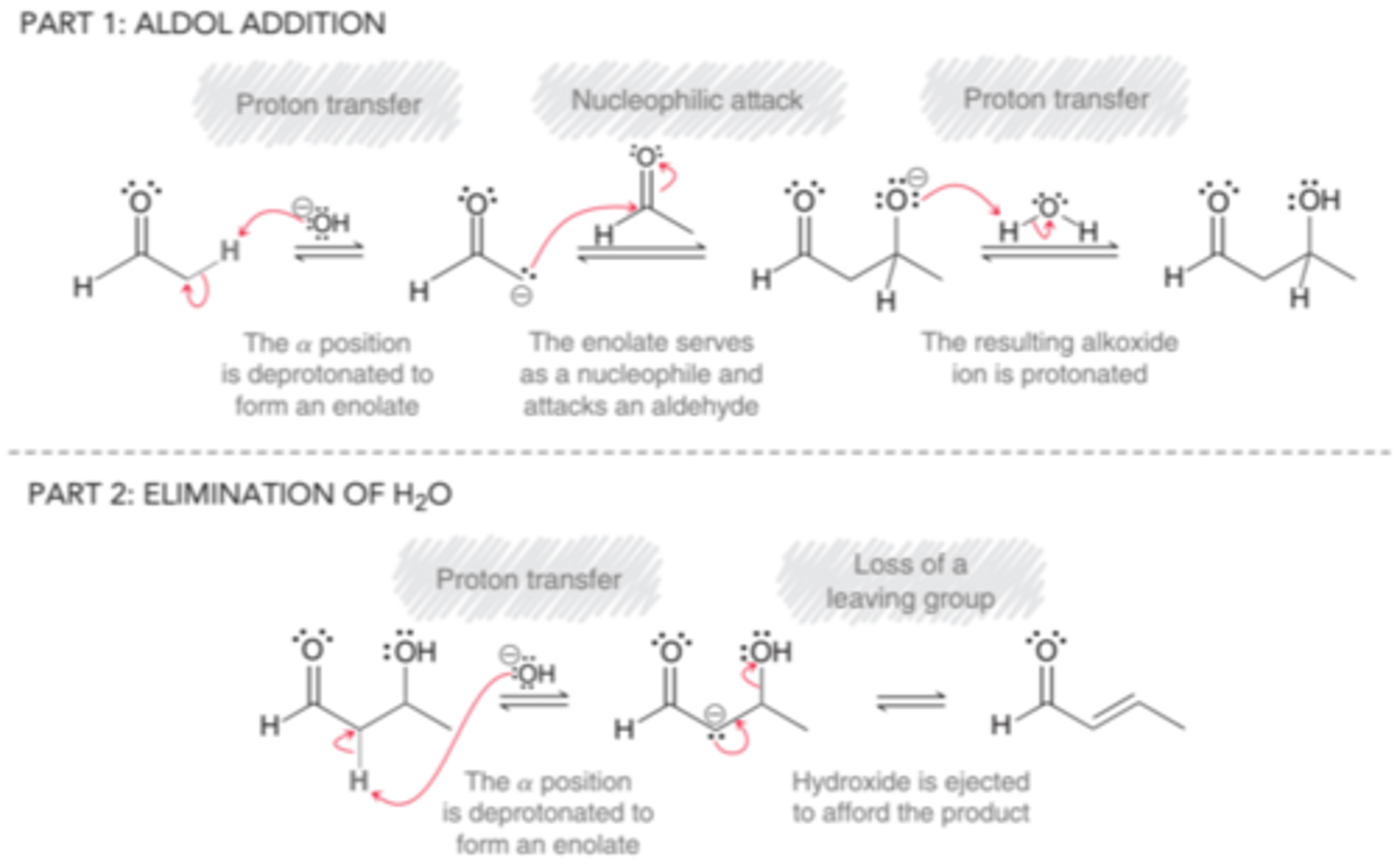
DIBAL-H
disabutylaluminum hydride
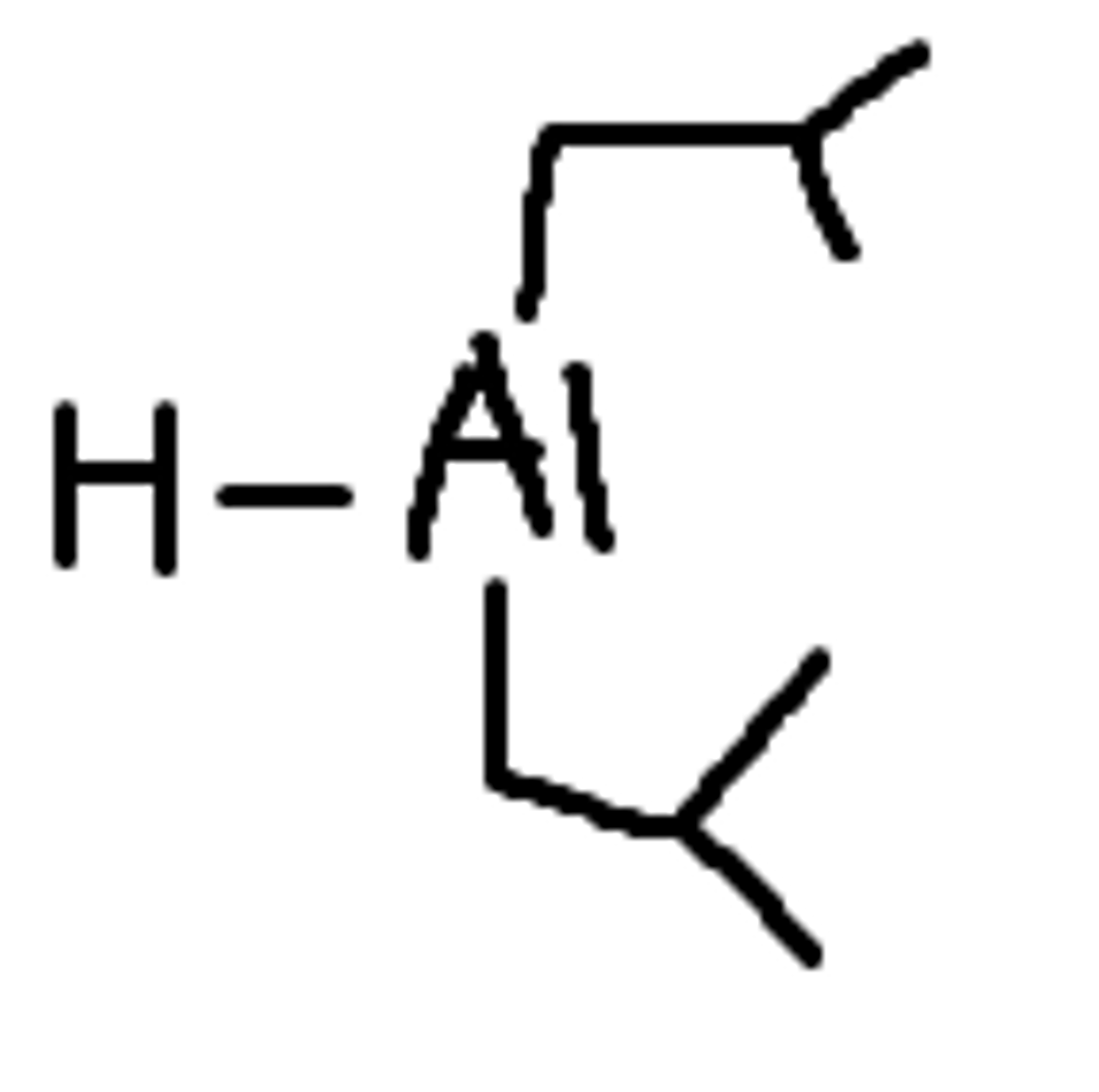
strong reducing agents
LiAlH4
weak reducing agent
Sodium Borohydride (NaBH4)
- Can only reduce aldehydes, ketones, and acids to alcohol
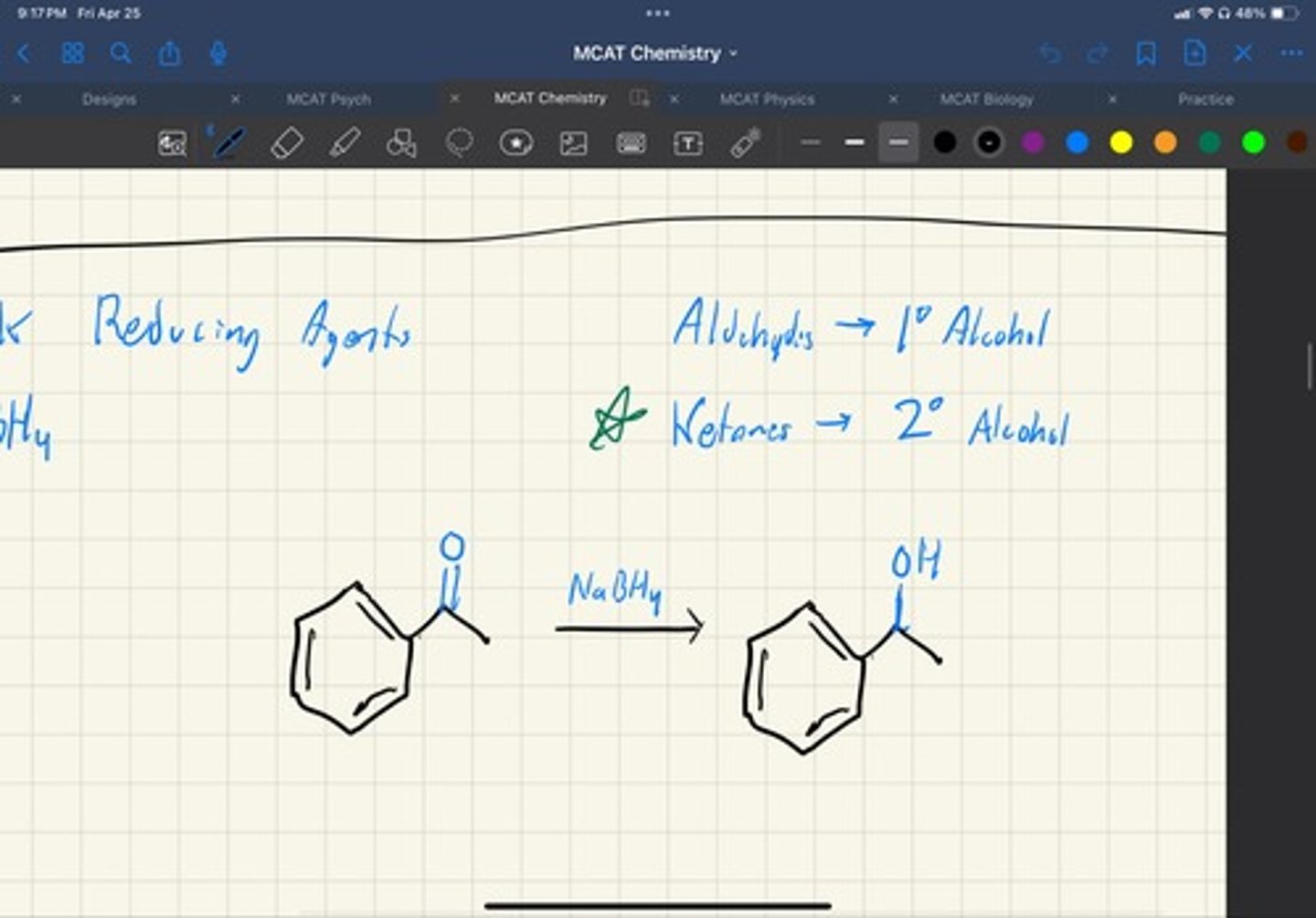
why are aldehydes more reactive
is is less sterically hindered and forms a less stable carbocation
strong acids make
weak conjugate bases
weak acids make
strong conjugate bases
Organometalics
RLi, R-MgBr
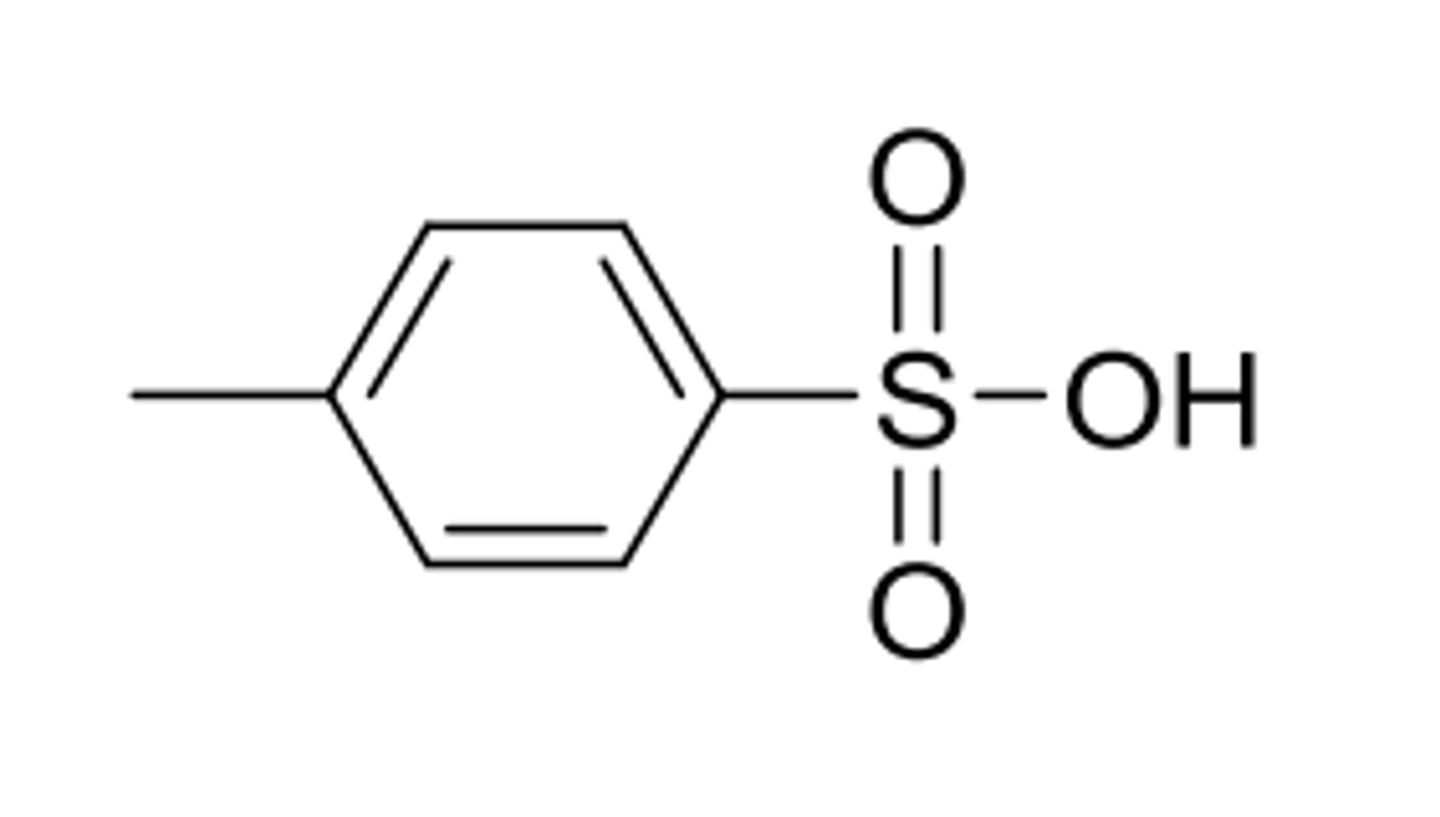
PTSA
p-toluenesulfonic acid
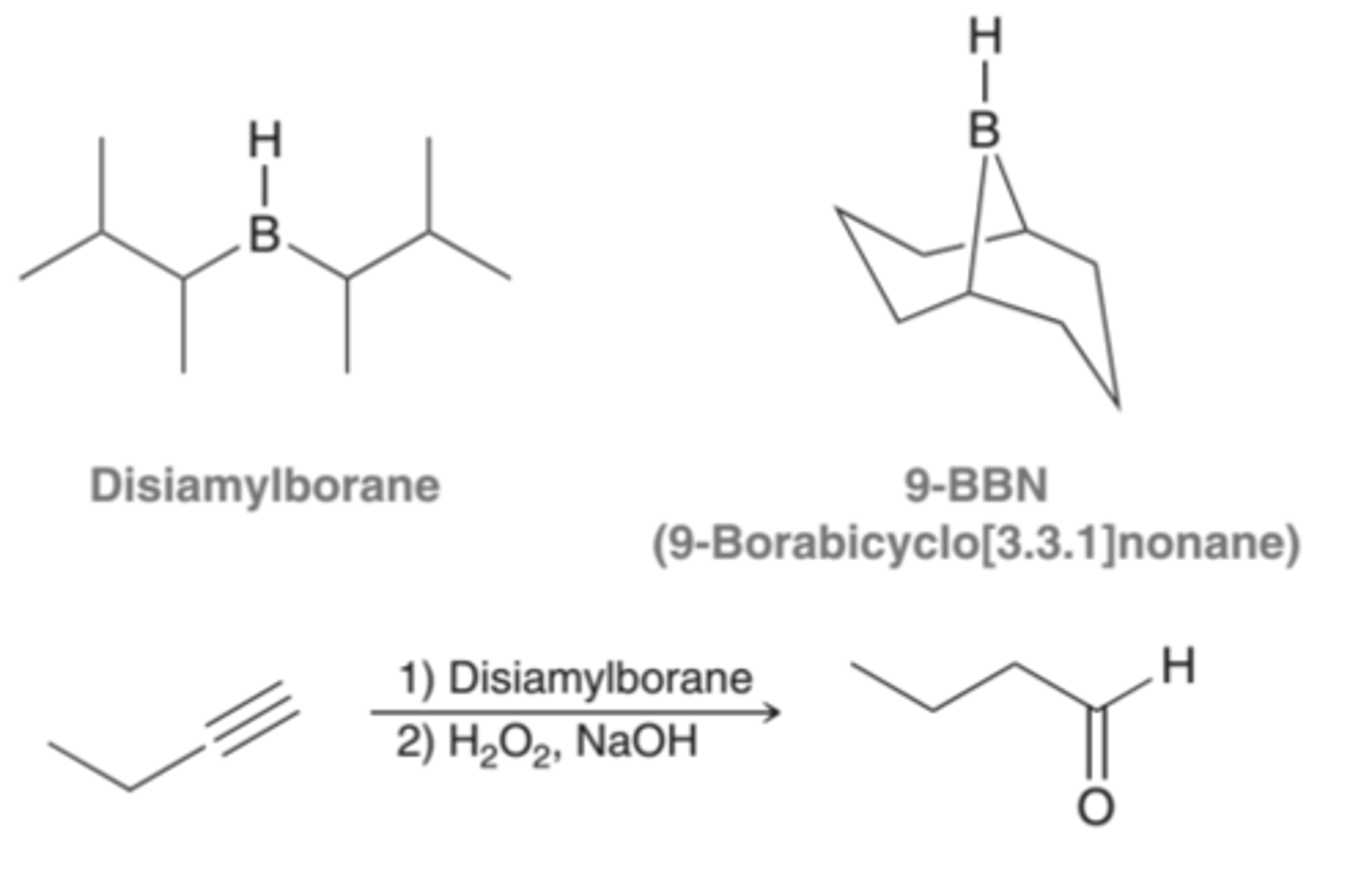
Reagents for Hydroboration-Oxidation
1. BH3, THF
2. H2O2, NaOH
Reagents for aldol condensation
NaOH, H2O, heat
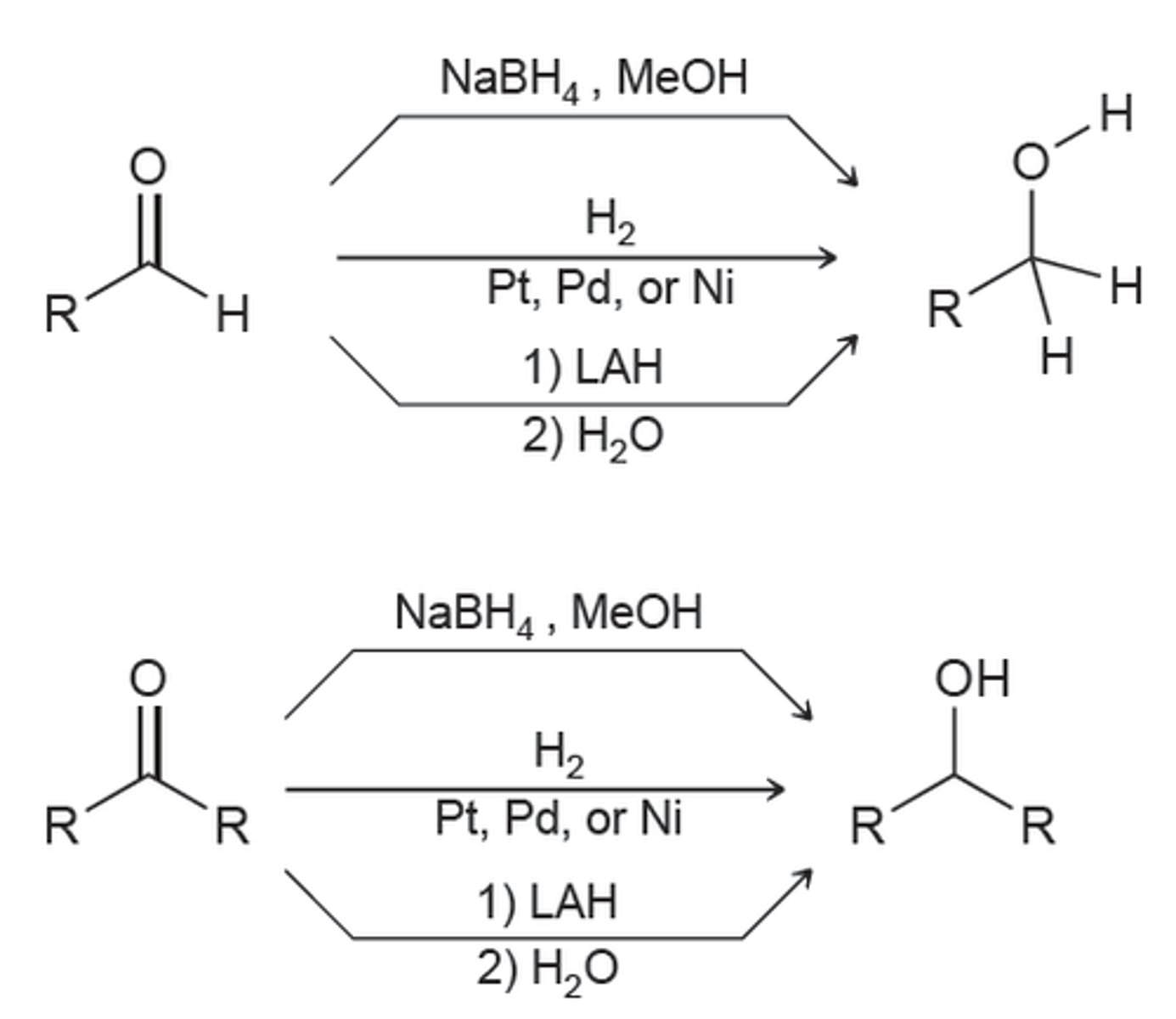
reagents for reduction
H2/Pt or Ni
NaBH4/H2O
LiAlH4/H2O
DIBAL-H
Reagents for nucleophilic addition
alcohol, organometallics, Grignard, amines, ammonia
nucleophilic addition reactions
Grignard, imines and enamine formation, acetal formation, hydride addition, halohydrins
2,4 DNP test
test for aldehydes and ketones

tollene's test
silver mirror test, checks for aldehydes
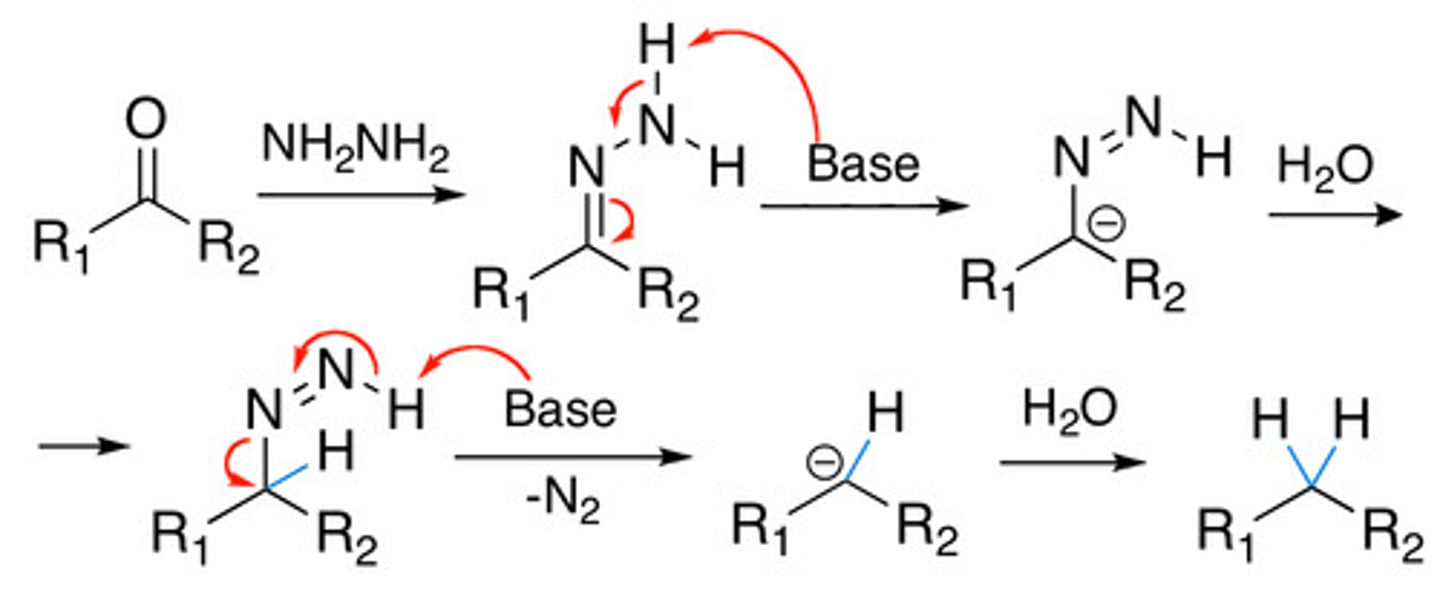
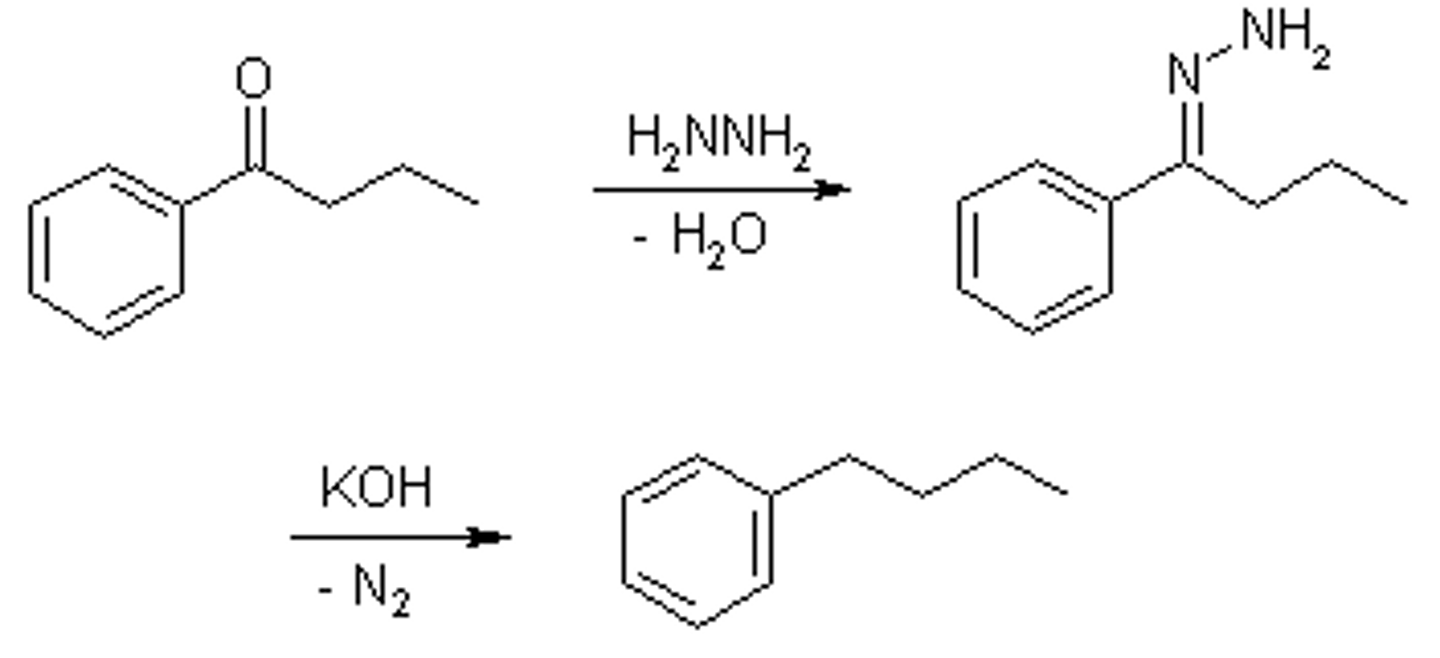
Reagents for wolf kishner
hydrazine/LDA to produce alkane
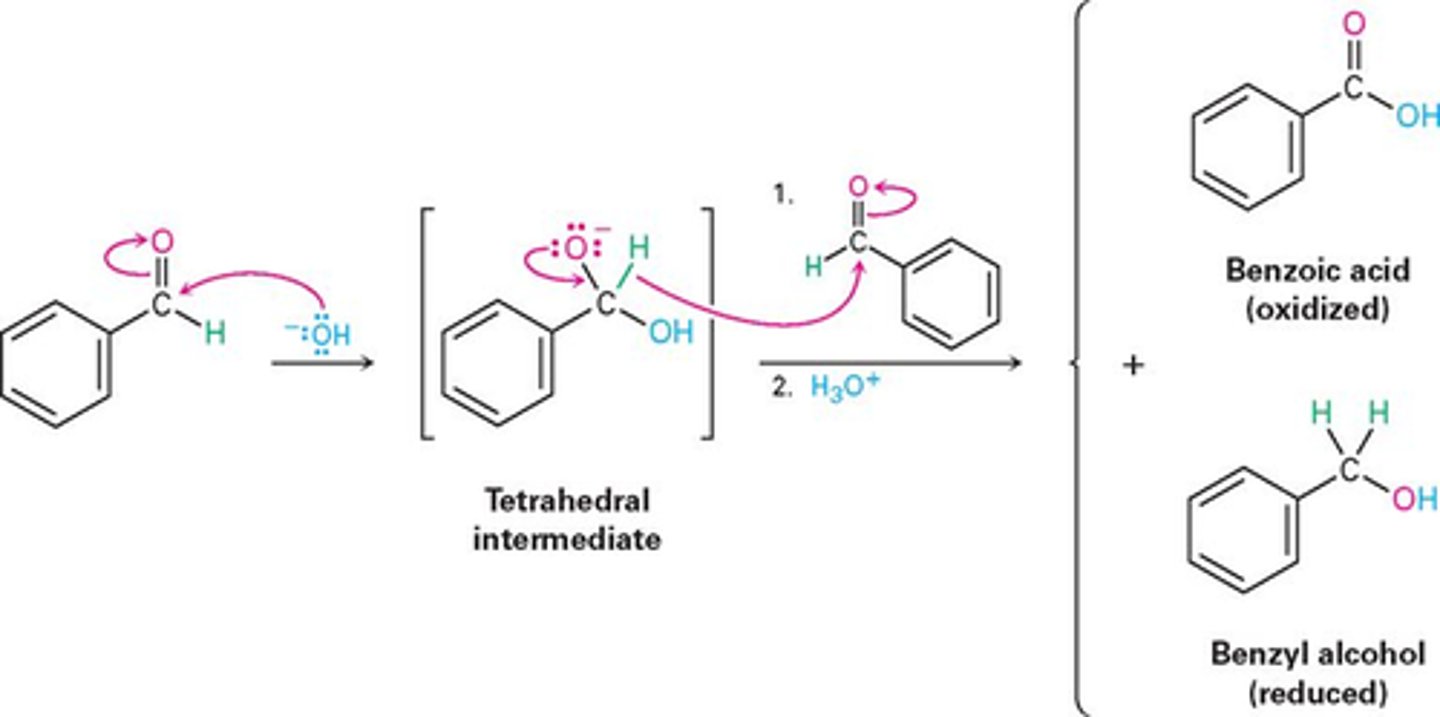
Cannizzaro Reaction
OH- with no alpha protons to produce alcohol and acid
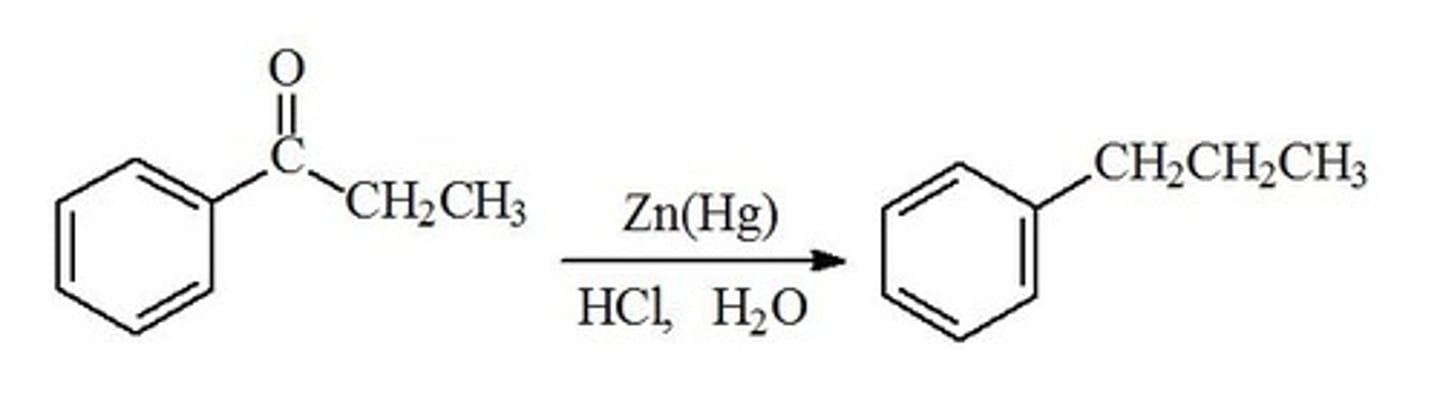
clemmenson reaction
Zn/Hg+ in HCl to produce alkane
witting reaction
1. Ph3P-CH2 (phosphonium ylide) to produce alkene
reagents for acid chloride to aldehyde
DIBAL-H
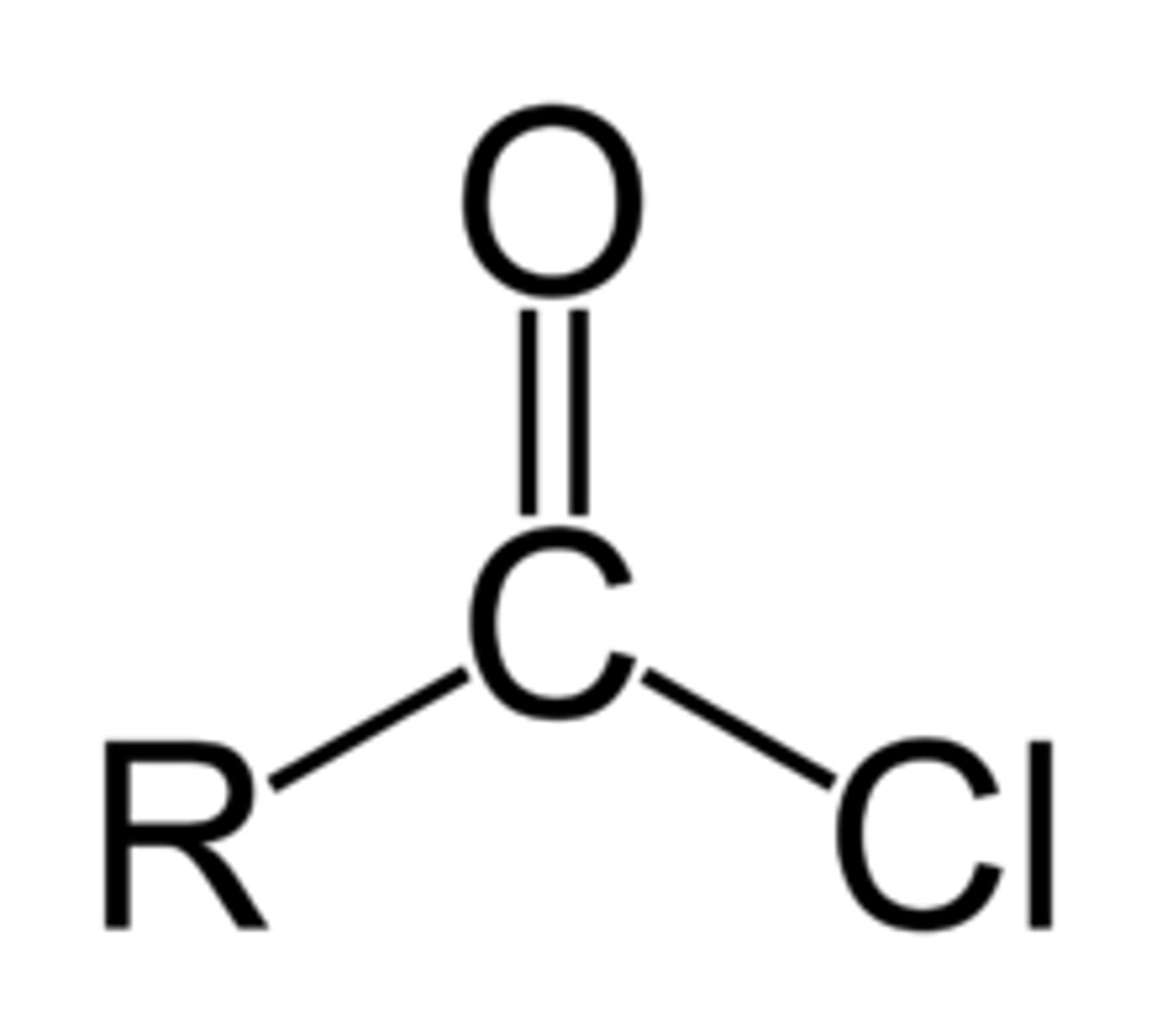
reagents for acid chloride to ketone
(CH3)2Li
molecular weight of benzaldehyde
106g/mol
molecular weight of acetone
58g/mol
Dibenzalacetone
product of 2 benzaldehyde and 1 acetone
color of dibenzalacetone
bright yellow