Chemistry of Life - Unit 1
1/31
Earn XP
Description and Tags
These flashcards cover key vocabulary terms and concepts related to the Chemistry of Life, crucial for understanding biological macromolecules and their functions.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Polarity
The property of water molecules that causes them to have a partial positive charge on one side and a partial negative charge on the other, leading to hydrogen bonding.
Hydrogen Bonding
A weak bond between a hydrogen atom and another atom, often seen in the interactions between water molecules.
Dehydration Synthesis
A chemical reaction that joins two molecules by removing a water molecule, forming a covalent bond between monomers.
Hydrolysis
A chemical reaction that breaks down a compound by adding water, splitting it into smaller molecules.
Macromolecules
Large biological molecules, such as carbohydrates, proteins, lipids, and nucleic acids, that are crucial for life.
Cohesion
The property of water that allows molecules to stick to each other, resulting in surface tension.
Adhesion
The property of water that allows it to stick to other types of molecules, which is important for processes like capillary action.
Monosaccharides
The simplest form of carbohydrates, consisting of single sugar molecules, which are the building blocks of polysaccharides.
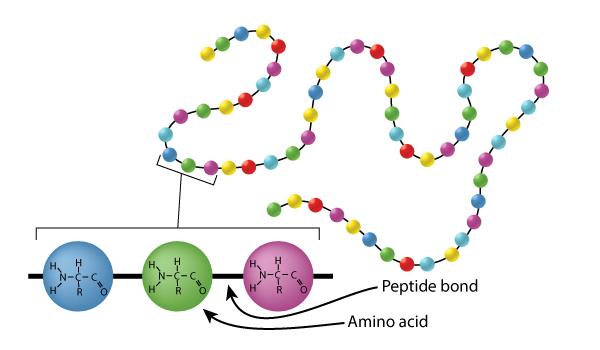
Amino Acids
The building blocks of proteins, consisting of a central carbon, an amine group, a carboxyl group, and a variable R group.
Lipids
Nonpolar molecules, including fats and oils, that are essential for energy storage and cell membrane structure.
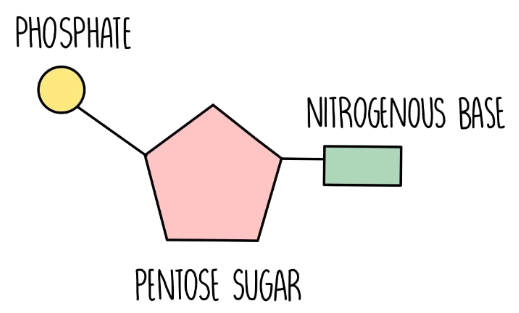
Nucleotides
The building blocks of nucleic acids, composed of a five-carbon sugar, a phosphate group, and a nitrogenous base.
Carbohydrates
Organic compounds made of carbon, hydrogen, and oxygen, which serve as energy sources and structural components in organisms.
Proteins
Biologically important macromolecules made up of amino acids that perform various functions including enzymatic activity, structural support, and signaling.
Surface Tension
cohesion + adhesion — allows water to move up plants from roots
Monomer
A small, simple molecule that can join together with other similar molecules to form a polymer.
Polymer
A large molecule made up of repeating structural units (monomers) connected by covalent bonds.
Peptide Bond
A covalent bond formed between two amino acids.
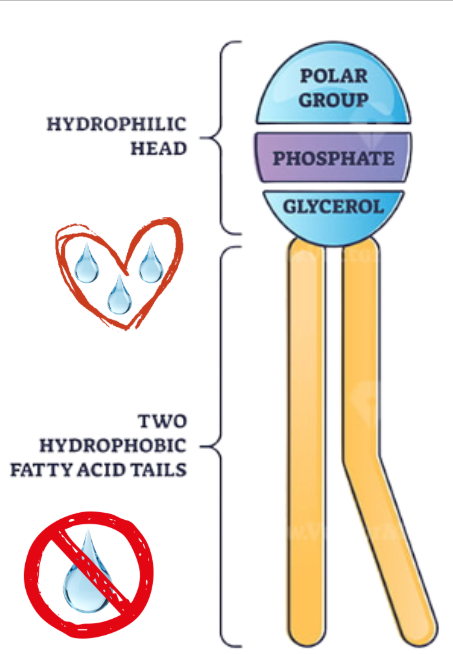
Phospholipid
A type of lipid that consists of two fatty acids, a glycerol unit, and a phosphate group, forming cell membranes through bilayers.
Nucleotide
The basic building block of nucleic acids, consisting of a five-carbon sugar, a phosphate group, and a nitrogenous base.
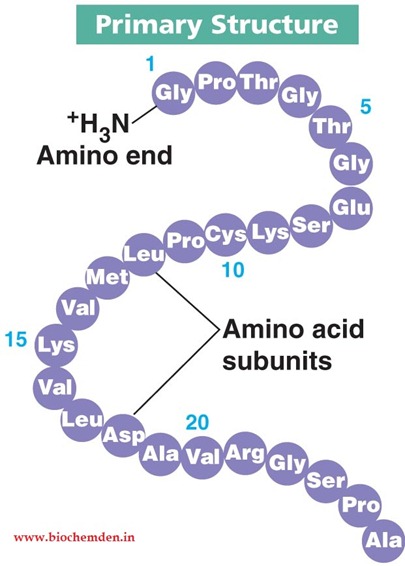
Primary Protein Structure
The linear sequence of amino acids in a protein, which determines its unique characteristics and function.
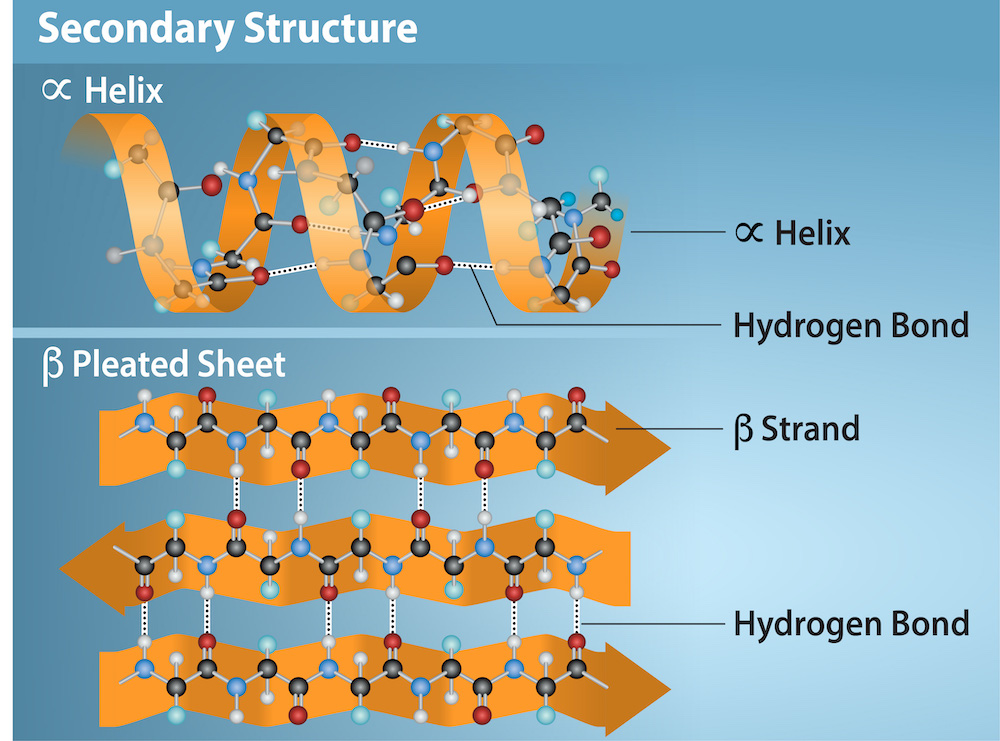
Secondary Protein Structure
The localized folding of a polypeptide chain into alpha helices and beta sheets, stabilized by hydrogen bonds.
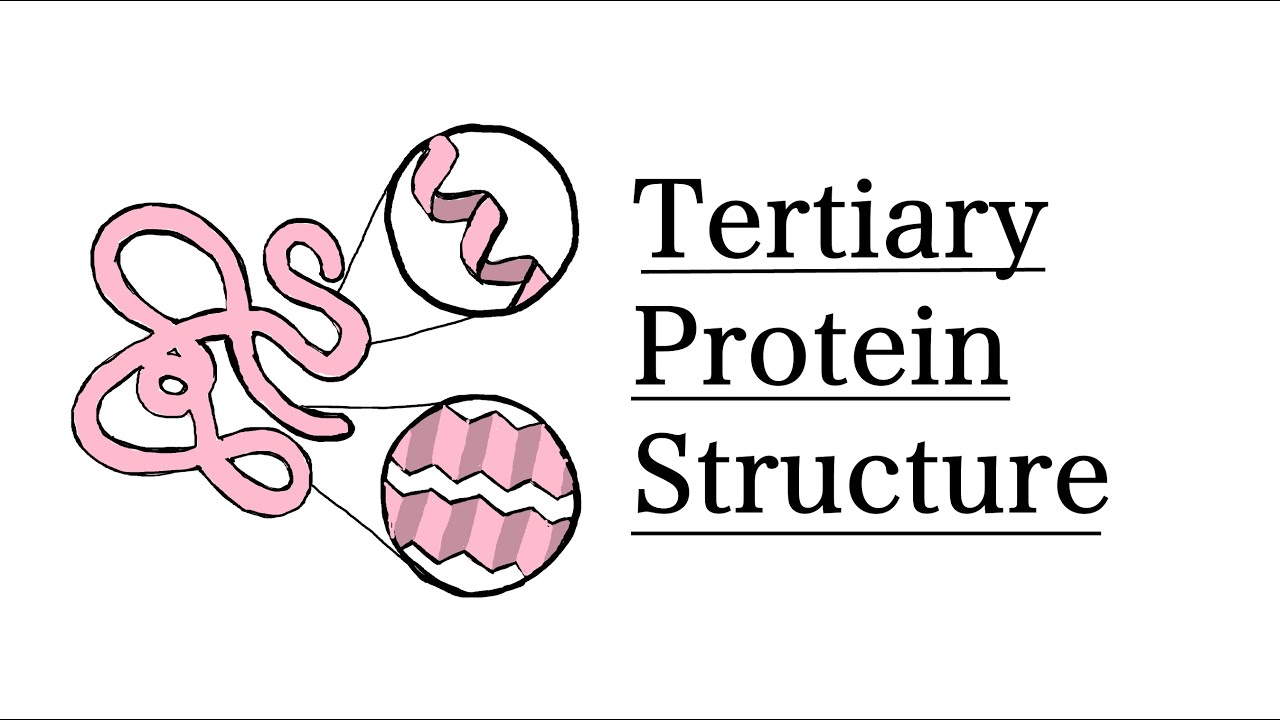
Tertiary Protein Structure
The overall 3D shape of a protein formed by the folding and twisting of the secondary structures due to various interactions (e.g., hydrogen bonding, ionic bonding).
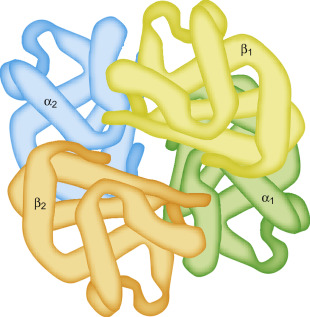
Quaternary Protein Structure
The arrangement and interaction of multiple polypeptide chains in a single protein complex, contributing to its function.
Electronegativity
A measure of the tendency of an atom to attract a bonding pair of electrons towards itself.
Covalent Bond
A chemical bond formed when two atoms share one or more pairs of electrons.
Ionic Bond
A chemical bond formed through the electrostatic attraction between oppositely charged ions after one atom transfers electrons to another.
Carboxyl Group
A functional group (-COOH) consisting of a carbon atom double-bonded to an oxygen atom and single-bonded to a hydroxyl group, characteristic of organic acids.
Amino Group
A functional group (-NH2) consisting of a nitrogen atom bonded to two hydrogen atoms, acting as a base and a building block for proteins.
Phosphate Group
A functional group (-PO4) that contains phosphorus and oxygen, playing a critical role in energy transfer (ATP) and nucleic acid structure.
Hydroxyl Group
A functional group (-OH) consisting of an oxygen atom bonded to a hydrogen atom, commonly found in alcohols and involved in hydrogen bonding.
Hydrophobic
Substances that repel water and are typically nonpolar, not easily dissolving in water.
Hydrophilic
Substances that attract water and are usually polar or charged, readily dissolving in water.