A level Chem 1.3 Bonding
1/49
Earn XP
Description and Tags
if you find it useful, please leave a 5 star rating :)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Which species has a square planar shape? (1)
A NH4+
B SF4
C XeF4
D PCl4+
C XeF4 (1)
What formula can you use to quickly determine the number of lone pairs on the central atom? (1)
Number of lone pairs on central atom=0.5x[Valence electrons on central atom-(Number of bonded atoms)+Charge] (1)
In which molecule are all the atoms in the same plane? (1)
A CH3CHO
B CH3NH2
C C6H5Cl
D C6H5CH3
C C6H5Cl (1)
Sodium fluoride contains sodium ions (Na+ ) and fluoride ions (F– ). Na+ and F– have the same electron configuration. Explain why a fluoride ion is larger than a sodium ion (2)
Fluoride ion has (two) fewer protons/lower nuclear charge (1)
Weaker attraction between nucleus and (outer) electrons (1)
The ion H2F+ is formed when hydrogen fluoride gains a proton as shown in the equation
HF+ + H+ → H2F+
Name the type of bond formed when HF reacts with H+
Explain how this bond is formed. (2)
Type of Bond: Coordinate bond / dative (covalent) bond (1)
Explanation: A (lone) pair of electrons is donated from F (1)
hydrogen ion (H⁺) has no electrons
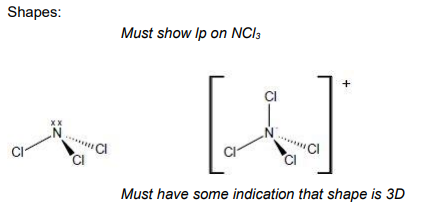
This question is about shapes of molecules and ions. Draw the shape of NCl3 and of NCl4 +
Include any lone pairs of electrons that influence the shape.
Name the shape of NCl3 State and explain the bond angle in NCl4 (5)
Name of shape of NCl3 = Pyramidal allow tetrahedral (1)
Bond angle =109.5
(4 bp and 0 lp) electron pairs repel equally / electron pairs repel to be as far apart as possible (1)
This question is about pentan-2-ol and pent-1-ene.
(a) The boiling point of pentan-2-ol is 119 °C
The boiling point of pent-1-ene is 30 °C
Explain why pentan-2-ol has a higher boiling point than pent-1-ene (3)
idea that pentan-2-ol has stronger intermolecular forces (1)
pent-1-ene has van der Waals’ forces (1)
pentan-2-ol (also) has hydrogen bond (1)
The melting point of XeF4 is higher than the melting point of PF3 .
Explain why the melting points of these two compounds are different.
In your answer you should give the shape of each molecule, explain why each molecule has that shape and how the shape influences the forces that affect the melting point. (6)
Level 3 5-6 marks
All stages are covered and the description of each stage is generally correct and virtually complete. Answer is communicated coherently and shows a logical progression from stage 1 to stage 2 and stage 3
Stage 1
XeF4 4 bonded pairs and 2 lone pairs around Xe (1)
PF3 3 bonded pairs and 1 lone pair around P (1)
Stage 2
XeF4 is square planar (1)
PF3 is pyramidal (allow tetrahedral) (1)
Electron pairs repel as far as possible /lone pair repels more than bond pair (1)
Stage 3 IMF
XeF4 has vdw forces and PF3 has dipole-dipole forces (and vdw) (1)
Stronger/more intermolecular forces in XeF4 (1)
Due to larger Mr or more electrons (1)
Which compound has the highest boiling point?
A CH3CH2CH2Br
B CH3CH2CH2F
C CH3CH2CHO
D CH3CH2COOH (1)
D as Propanoic acid is a carboxylic acid. In addition to van der Waals forces and dipole-dipole interactions, it can form strong hydrogen bonds between molecules (1)
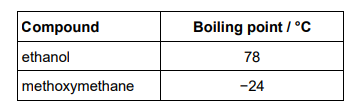
Methoxymethane (CH3OCH3) is an isomer of ethanol. The table shows the boiling points of ethanol and methoxymethane.
In terms of the intermolecular forces involved, explain the difference in boiling points. (3)
Hydrogen bonds (between ethanol molecules) (1)
(permanent) dipole-dipole OR van der Waals force (between methoxymethane molecules) (1)
Hydrogen bonds are stronger/est intermolecular force (1)
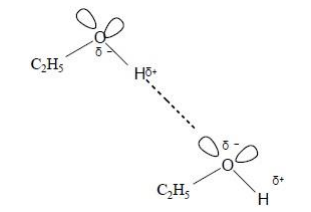
This question is about structure and bonding.
Draw a diagram to show the strongest type of interaction between two molecules of ethanol (C2H5OH) in the liquid phase.
Include all lone pairs and partial charges in your diagram. (3)
two lone pairs on each O atom and δ+ and δ- on each H-O bond (1)
dotted/broken line shown between lone pair on one molecule and the correct H on another (1)
O........H–O in straight line (1)
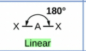
Sketch a linear bond shape and state how many bonded pairs and lone pairs are in the structure (2)
2 bonded pairs and no lone pairs (1)
Sketch a trigonal planar bond shape and state how many bonded pairs and lone pairs are in the structure (2)
3 bonded pairs and no lone pairs (1)
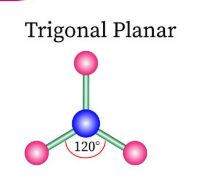
Sketch a tetrahedral bond shape and state how many bonded pairs and lone pairs are in the structure (2)
4 bonded pairs and no lone pairs (1)
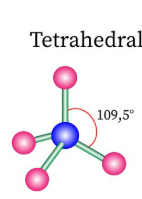
Sketch a trigonal bipyramidal bond shape and state how many bonded pairs and lone pairs are in the structure (2)
5 bonded pairs and no lone pairs (1)
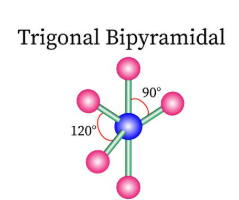
Sketch a octahedral bond shape and state how many bonded pairs and lone pairs are in the structure (2)
6 bonded pairs and no lone pairs (1)
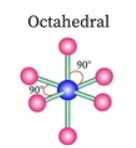
Sketch a square pyramidal bond shape and state how many bonded pairs and lone pairs are in the structure (1)
5 bonded pairs and 1 lone pair (1)
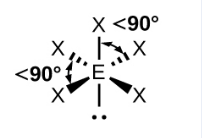
Sketch a square planar bond shape and state how many bonded pairs and lone pairs are in the structure (1)
4 bonded pairs and 2 lone pairs (1)
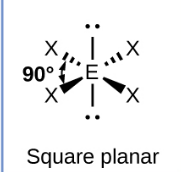
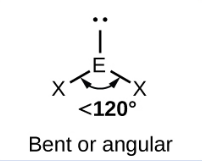
Sketch a Bent bond shape with 2 bonded pairs and state how many lone pairs are in the structure (1)
1 lone pair (1)
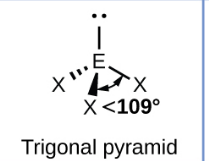
Sketch a trigonal pyramidal bond shape and state how many bonded pairs and lone pairs are in the structure (2)
3 bonded pairs and 1 lone pair (1)
Sketch a See Saw bond shape and state how many bonded pairs and lone pairs are in the structure (1)
4 bonded pairs and 1 lone pair (1)
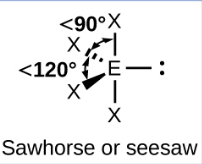
Sketch a T-shaped bond shape and state how many bonded pairs and lone pairs are in the structure (1)
3 bonded pairs and 2 lone pair (1)
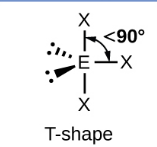
Sketch a Bent bond shape with 2 bonded pairs and state how many lone pairs are in the structure (1)
2 lone pairs (1)
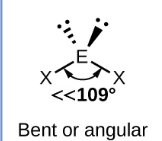
Sketch a linear bond shape with 2 bonded pairs and state how many lone pairs are in the structure (1)
3 lone pairs (1)
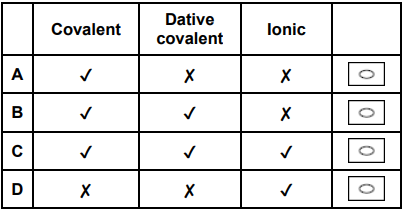
Which row shows the bonding in ammonium chloride? (1)
C (1)
Ionic bonding does not require a metal.
It only requires a positively charged ion and a negatively charged ion
Explain how permanent dipole-dipole forces arise between hydrogen chloride molecules. (2)
Difference in electronegativity leads to bond polarity (1)
(dipoles don’t cancel therefore the molecule has an overall permanent dipole) and there is an attraction between ∂+ on one molecule and ∂− on another (1)
There are two lone pairs of electrons on the oxygen atom in a molecule of oxygen difluoride (OF2).
Explain how the lone pairs of electrons on the oxygen atom influence the bond angle in oxygen difluoride. (2)
Lone pairs repel more than bond pairs (1)
bond angle will be lower (than regular tetrahedral angle) / bond angle of 103-106°(1)
Silicon tetrafluoride (SiF4) is a tetrahedral molecule.
Deduce the type of intermolecular forces in SiF4
Explain how this type of intermolecular force arises and why no other type of intermolecular force exists in a sample of SiF4 (3)
Van der Waals forces (1)
(Uneven distribution of electrons in) one molecule induces dipole in neighbouring /another/nearby molecule (1)
symmetrical molecule / dipoles cancel OR
no hydrogens bonded to F (N or O), therefore no hydrogen bonding (1)
Which molecule does not have a permanent dipole?
A CH3Br
B CH2Br2
C CHBr3
D CBr4 (1)
D (1)
Explain why the C–Cl bond is polar. (2)
Chlorine has a higher electronegativity (than carbon) (1)
So the electron density is unsymmetrical/so chlorine becomes δ- and carbon becomes δ+ (1)
There are van der Waals forces between non-polar molecules.
Explain what causes these forces. (3)
(Random) movement of electrons (in one molecule creates a dipole) (1)
Induces a dipole in another molecule (1)
(These temporary) dipoles in different molecules attract (1)
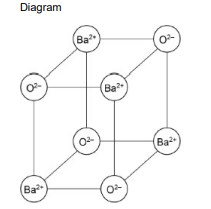
Barium reacts with oxygen to form barium oxide.
Barium oxide has a high melting point and an ionic lattice structure similar to that of sodium chloride.
Draw a 3D diagram to show how the particles are arranged in a barium oxide lattice. You should draw eight particles. (2)
Alternating ions with correct charges on ions (1)
Cubic 3D arrangement with minimum of 8 particles (1)
SF6 and SF3+ have different shapes and different bond angles.
Deduce the shape of SF6 and the shape of SF3+
State the bond angle in SF6 and the bond angle in SF3+
Justify the bond angles by referring to electron pairs (6)
SF6 is octahedral (either in words or as a structure) (1)
SF6 bond angle is 90° (1)
SF6 all the bond pairs repel equally (1)
SF3+ is (trigonal) pyramidal (1)
SF3+ bond angle is 103-107°(1)
SF3+ lone pair-bond pair repulsion is greater than bond pair-bond pair
repulsion (so bond angle is reduced) (1)

Explain why tetrachloromethane has a higher boiling point than dichloromethane. (2)
van der Waals' forces between molecules in CCl4 stronger than (combined van der Waals' and) dipole-dipole forces between molecules in CH2Cl2 (1)
as CCl4 has (many) more electrons than CH2Cl2 (1)

Propan-1-ol has a higher boiling point than the other two compounds because of hydrogen bonding. Describe the hydrogen bonding in propan-1-ol. (2)
attraction between O lone pair (1)
and δ+ H of OH on another molecule (1)
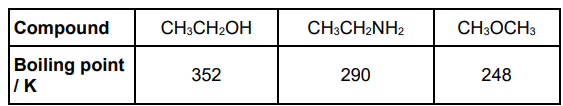
The table below shows some data about three compounds that all contain the same number of electrons.
All three compounds in the table above are polar.
Ethanol is the most polar and ethylamine is the least polar.
Explain why all three molecules are polar and why ethylamine is the least polar. In your answer refer to the shapes around, and relative electronegativities of, the most electronegative atoms.(4)
O AND N more electronegative than C and/or H (1)
CH3CH2OH and CH3OCH3 both v-shaped/non-linear/bent AND CH3CH2NH2 trigonal pyramidal(1)
shapes are not symmetrical (so molecules are polar) (1)
O more electronegative than N (so ethylamine is least polar) (1)
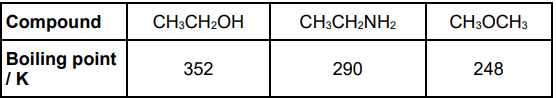
Explain the trend in the boiling points of the three compounds. Refer to the intermolecular forces in all three compounds in your answer. (3)
hydrogen bonding in CH3CH2OH and CH3CH2NH2 AND (permanent) dipole-dipole forces in CH3OCH3 (1)
hydrogen bonding stronger (than other (intermolecular) forces) (1)
hydrogen bonding stronger in CH3CH2OH than in CH3CH2NH2(1)
Which molecule has a permanent dipole?
A CF4
B PCl5
C CO2
D Cl2O (1)
D (1)
Which compound contains a co-ordinate bond?
A HF
B NH3
C CHCl3
D NH4Cl (1)
D (1)
Which change occurs when water is vaporised?
A An exothermic change occurs.
B Covalent bonds are broken.
C Intermolecular forces are overcome.
D The total energy of the molecules decreases. (1)
C (1)
Which species has a shape that is influenced by the presence of one or more lone pairs of electrons around the central atom?
A AlCl3
B ClF3
C IF6+
D PCl6– (1)
B (1)
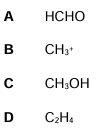
Which of these species is not planar?
(1)
C (1)
What is the formula of calcium nitrate(V)?
A CaNO3
B Ca(NO3)2
C Ca2NO2
D Ca(NO2)2 (1)
B (1)
Calcium nitrate(V) means:
Calcium forms a Ca²⁺ ion
Nitrate(V) is the ion NO₃⁻
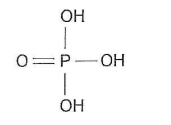
Cyclohexene is prepared by the dehydration of cyclohexanol using concentrated phosphoric acid as a catalyst. The structure of concentrated phosphoric acid is shown
Identify the factors that influence the boiling points of each of the compounds in this reaction mixture State how and explain why cyclohexene can be separated from the reaction mixture (6)
Cyclohexene has van der Waals forces between molecules (1)
Cyclohexanol has hydrogen bonds between molecules (1)
Phosphoric acid have hydrogen bonds between molecules (1)
Cyclohexene has the weakest forces (1)
So it has the lowest boiling point (1)
it’s easily separated by distillation (1)
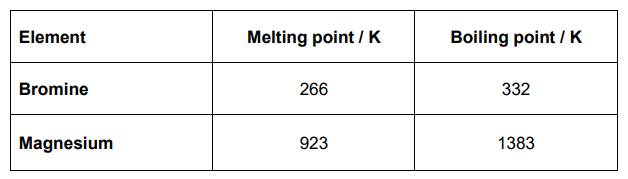
Table 2 shows some data about the elements bromine and magnesium.
In terms of structure and bonding explain why the boiling point of bromine is different from that of magnesium. Suggest why magnesium is a liquid over a much greater temperature range compared to bromine (5)
Bromine is (simple) molecular / simple molecules (1)
Magnesium is metallic/consists of (positive) ions in a (sea) of delocalised electrons (1)
Br2 has weak van der Waals forces between the molecules (1)
so more energy is needed to overcome the strong (metallic) bonds
Liquid range
Mg has a much greater liquid range because forces of attraction in liquid/molten metal are strong(er) (1)
Ammonia reacts with aluminium chloride as shown by the equation:
NH3 + AlCl3 → H3NAlCl3
Explain how the value of the Cl-Al-Cl bond angle in AlCl3 changes, if at all, on formation of the compound H3NAlCl3 (2)
Aluminium is now surrounded by 4 electron pairs/bonds or is tetrahedral (1)
Therefore Cl-Al-Cl bond angle decreases / changes (from 120° in AlCl3 ) (1)
Explain why sodium metal is malleable (can be hammered into shape). (1)
Layers can slide over each other – idea that ions/atoms/particles move (1)
State the meaning of the term electronegativity (2)
Ability/power of an atom/element/nucleus to withdraw electron density or electron cloud or a pair of electrons (towards itself) (1)
From a covalent bond or from a shared pair of electrons (1)
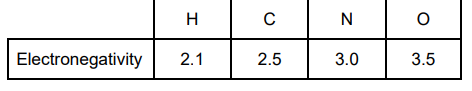
Use the values in the table to explain how the strongest type of intermolecular force arises between two molecules of ammonia. (3)
There is a large electronegativity difference between N + H / N is very electronegative (1)
Forms N δ– / H δ+ or dipole explained in words (1)
Lone pair on N forms weak bonds with H (δ+) (1)
Explain, in terms of electronegativity, why the boiling point of H2S2 is lower than H2O (2)
Electronegativity of S lower than O or electronegativity difference between H and S is lower (1)
No hydrogen bonding between H2S2 molecules (1)