measuring electrode potentials and cell notation
1/15
Earn XP
Description and Tags
update w/ tricky examples and h2so4 conc thing
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
what is the standard electrode potential (Eθ)?
p.d. measured under standard conditions when the ½ cell is connected to a standard hydrogen electrode
a measure of where the PoE lies for each ½ cell
how can we relate electrode potentials to PoE?
the more +ve the electrode potential, the further right the PoE
how are ½ equations always written for standard electrode potentials?
always written as reduction (see image for e.g.)

what can we use to measure the Eθ for a single ½ cell?
standard hydrogen electrode = electrode consisting of H2 gas in contact w/ H+ ions on a platinum surface
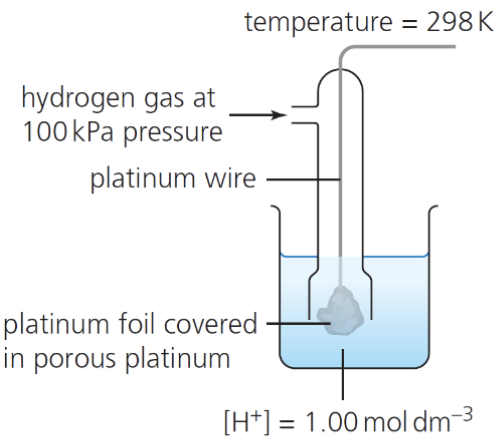
describe how we can use the standard hydrogen electrode to measure the Eθ for a single ½ cell:
H2 gas flows over the inert Pt electrode to establish the equilibrium 2H+ (aq) + 2e- ⇌ H2 (g)
the standard hydrogen electrode is connected to the ½ cell under investigation under standard conditions
the Eθ of the standard hydrogen electrode is 0.00 V by definition
so we can determine the Eθ by using the formula Ecell = Eright - Eleft
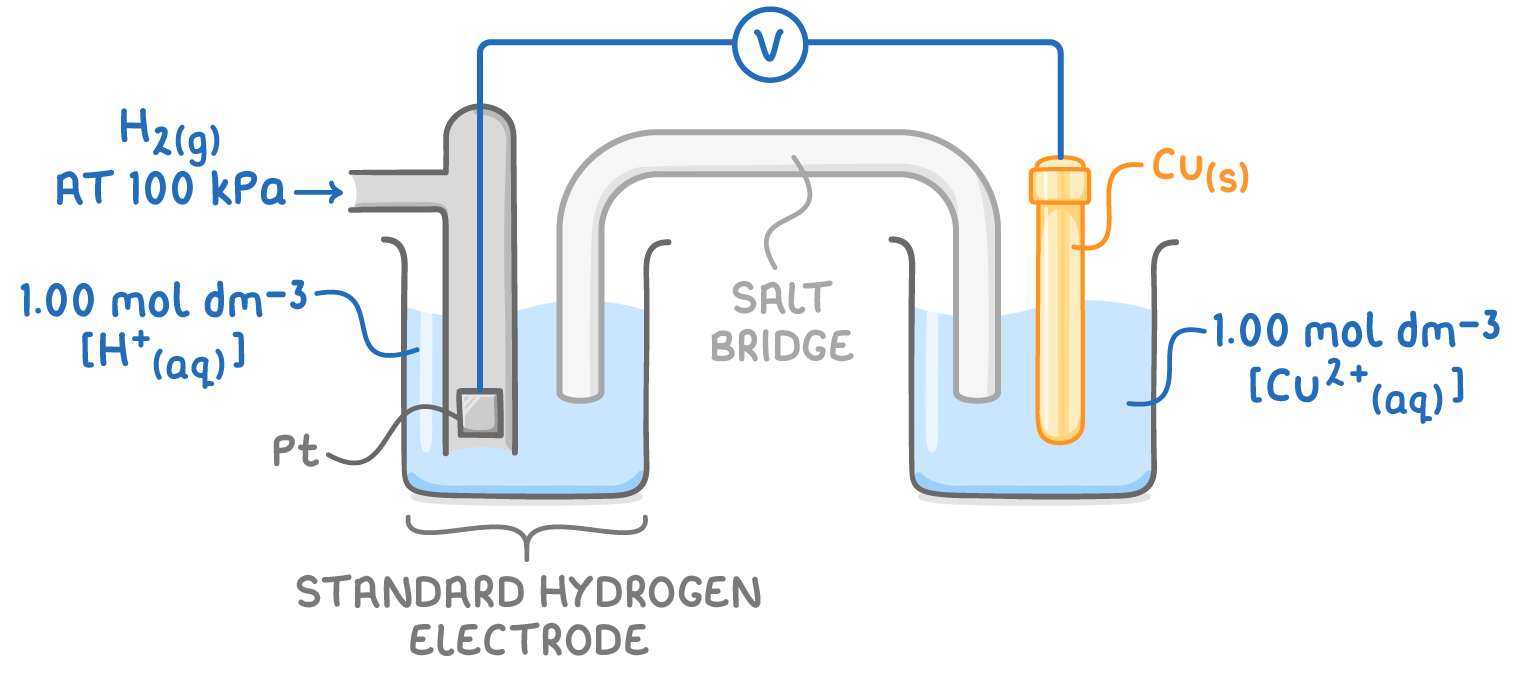
state the standard conditions necessary when using the standard hydrogen electrode:
298 K
H2 gas at 100 kPa
all solns have a conc of 1 mol dm-3
which side is the standard hydrogen electrode always positioned on?
LHS - regardless of Eθ values of other cells
give the formula used to calculate the EMF (Ecell) of a cell:
Ecell = Eright - Eleft
give the symbols used in cell notation and state what they represent:

which species is placed closest to the salt bridge || when writing cell notation?
species w/ the highest oxidation state (i.e. oxidised species)
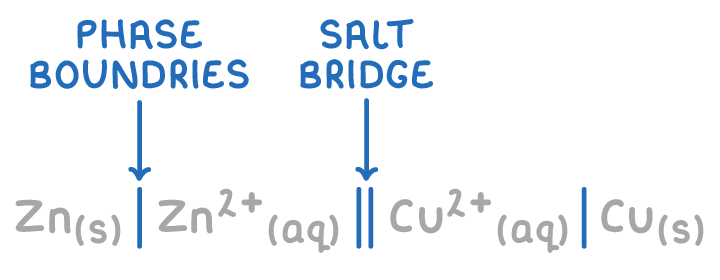
on which side of the salt bridge || do you write each species in cell notation?
as seen/given in a diagram:
-ve ½ cell (oxidised) on LHS
+ve ½ cell (reduced) on RHS
why is Pt used as the contact electrode in the standard hydrogen electrode?
inert
give and explain the cell notation for the standard hydrogen electrode being used to measure the EMF of a Cu/Cu2+ ½ cell:
Pt (s) | H2 (g) | H+ (aq) || Cu2+ (aq) | Cu (s) :
species w/ highest oxidation state closest so salt bridge
| separate species w/ diff states
Pt (s) included - used as a contact: solid contact must be used if there are only aq/gaseous species

you are provided with the Daniell cell referred to in part (b), including a Zn electrode of known mass - briefly outline how you could carry out an experiment to confirm your answer to part (b) (3)
allow cell to discharge until [CuSO4] = 0.5 mol dm-3
confirm using colorimetry
weigh mass of Zn electrode before and after experiment
![<ul><li><p>allow cell to discharge until [CuSO<sub>4</sub>] = 0.5 mol dm<sup>-3</sup></p></li><li><p>confirm using colorimetry</p></li><li><p>weigh mass of Zn electrode before and after experiment </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/07b94776-d6dd-4cbb-9f6e-5fa11fa6a424.png)

use data from the table above to explain why Au+ ions are not normally found in aq soln (2)
Eθ of Au+ bigger than Eθ of H2O
so Au+ ions oxidise H2O

use data from the table to predict and explain the redox reactions that occur when iron powder is added to an XS of aq silver nitrate (3)
Eθ of Ag+ bigger than Eθ of Fe2+
Eθ of Ag+ bigger than Eθ of Fe3+
so Ag+ oxidise Fe → Fe2+ and → Fe3+
