Acids
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
What does a pH less than 7 show?
Increasing acidity
What does a pH greater than 7 show?
increasing alkalinity
What does a low conc of H+ ions mean for the pH?
The pH is high
What does a high conc of H+ ions mean for the pH?
The pH is low (acidic)
What does a change in one pH number mean for the H+ concentration?
there is a 10x difference
e.g. if pH decreases by 1 then H+ conc increase by a factor of 10
How are hydrogen concentrations of 10-1 to 10-14 converted into pH values of 1-14?
Using a negative logarithm
What is the mathematical relationship between pH and H+ concentration?





What is a monobasic acid?
an acid that contains only 1 hydrogen ion which can be donated to a base
In aq solutions what happens to a strong monobasic acid?
It completely dissociates
If a strong monobasic acid completely dissociates in aq solution how does the concentrations of the H+ ions and the acid compare?
They are the same

How can the pH of a strong acid be calculated?
Directly from the concentration of the acid because the concentration of H+ ions = the concentration of the acid






What do strong and weak acids describe?
The extent of dissociation of an acid
What is a weak acid? And what is the general form it can be shown in?
An acid which partially dissociates in aq solutions (therefore it is a reversible reaction)

What is Ka?
The acid dissociation constant - it can be used for acid-base equilibria
What is the formula for the acid dissociation constant? and how do you calculate the units

True or false Ka changes with temperature
true
Why are record Ka value standard at a temperature?
because Ka changes with temperature
What temperature are Ka values standardised at?
25 degrees Celsius
How does the value of Ka relate to the equilibrium constant?
the larger the Ka, the further to the right the equilibrium is the lower the Ka the further to the left the equilibrium is
How does the Ka relate to dissociation and acid strength?
The larger the Ka value the greater the dissociation and the greater the acid strength and vice versa
Because it is difficult to compare numbers with negative indices what is Ka converted to? And what is the formula?
a negative logarithm called pKa




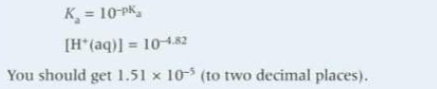
The stronger the acid the ____________ the Ka value and the ____________ the pKa value
The stronger the acid the larger the Ka value and the smaller the pKa value
The weaker the acid the __________ the Ka value and the __________ the pKa value
The weaker the acid the smaller the Ka value and the larger the pKa value
What are pKa values used for?
comparing the strength of weak acids particularly in biological systems
In a weak acid what does the concentration of H+ ions depend on?
the concentration of the acid and the acid dissociation constant Ka
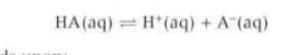
When HA molecules dissociate H+ and A- ions are formed in equal quantities so what are the start an equilibrium quantities of HA? And therefore how can Ka be written?

What is approximation 1 of the Ka equation?
The dissociation of water is negligible
HA dissociates to produce equilibrium concentrations of H+ and A- that are equal. there will be a very small concentration of H+ from the dissociation of water but this will be negligible compared with the H+ concentration from the acid.

When does the first approximation not work?
for very weak acids or very dilute solutions because if pH>6 the [H+] form the dissociation of water will be significant compareed with dissociation of the weak acid
What is the second approximation of the Ka equation?
The concentration of acid is much greater than the H+ concentration at equilibrium

When can the second approximation work and when is it not justified?
The approximation will work for weak acids with small Ka values. It breaks down when the concentration of H+ becomes significant and there is a real difference between [HA(aq)] at equilibrium and ([HA(aq)] at start - [H+(aq)]at equilibrium)
The approximation is not justified for the stronger weak acids with a
Ka > 10-2 moldm-3 and for very dilute solutions
By applying the 2 approximations how can the Ka expression be greatly simplified?



How can the Ka of a weak acid be determined? (in practice)
By preparing a standard solution of the weak acid of known concentration
How can you measure the pH of a standard solution?
Using a pH metre




What is the Arrhenius definition of acids and bases?
acids dissociate and release H+ ions in aq solution and alkalis dissociate to release OH- ions in aq solution
What is an alkali?
A soluble base
What is a Bronsted-Lowry acid and a Bronsted-Lowry base?
Bronsted-Lowry acid - a proton donor
Bronsted-Lowry base - proton accepter
What is a conjugate acid base pair? Use HCl as an example
Contains 2 species that can be interconverted by the transfer of a proteins.
E.g. with HCl: in the forward direction, HCl releases a protein to form its conjugate base, Cl- and in the reverse direction Cl- accepts a protein to form its conjugate acid HCl
What is the ionic equation for neutralisation?
H+ + OH- —> H2O
Describe the ionisation of water
water ionises slightly, acting as both an acid and a base
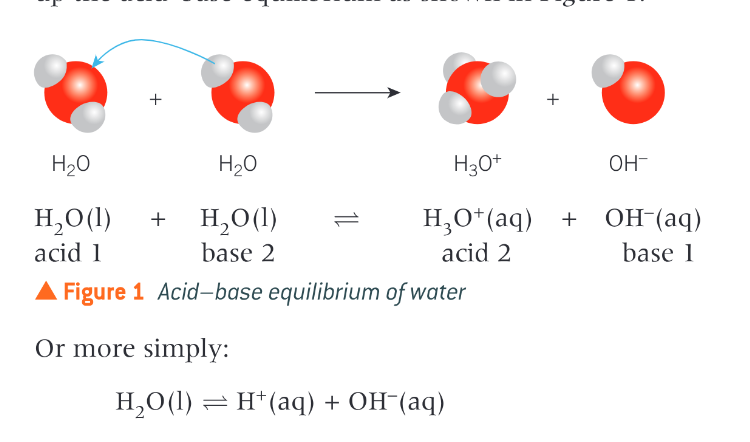
What is Kw and what does it do?
Kw is the ionic product of water - the ions in water (H+ and OH-) multiplied together
control the concentration of H+ and OH- ions in aq solutions
Describe the pH of pure water at 298K (in the terms of ions)
on dissociation water is neutral, it produces the same number of H+ and OH- ions
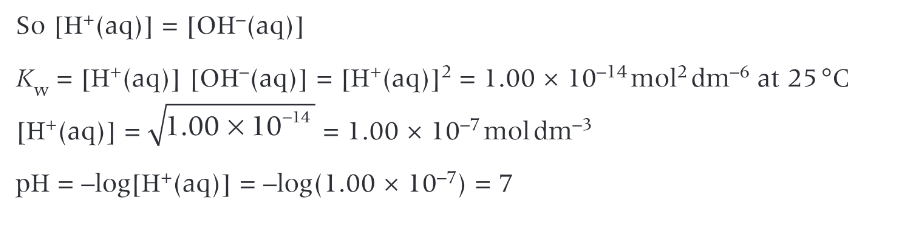
what is a strong base?
an alkali that completely dissociates in solution
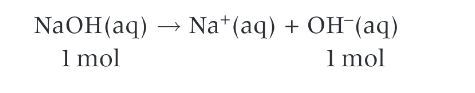
What is a monoacidic base?
where one mole of a base releases one mole of OH- ions
What can the pH of a strong base be calculated by?
the concentration of the base
the ionic product of water
What is the hydronium ion? when is it formed?
H3O+
formed when H2O has accepted a proton to form its conjugate acid
What is a monobasic acid?
when one mole of an acid dissociates to form one mole of protons
What is a buffer solution?
a system that minimises pH changes when small amounts of an acid or a base are added. They contain 2 componants to do this
a weak acid - which removes added alkali
and its conjugate base which removes added acid
True or false a buffer can be used up
true
How is a buffer made from a weak acid and one of its salts?
a buffer solution can be prepared by mixing a solution of a weak acid and a solution of one of its salts e.g. ethanoic acid and sodium ethanoate. When ethanoic acid is added to water the acid partially dissociates and the amount of ethanoate ions in solution is very small. When added to water the salt completely dissolves, so the salt is the source of the conjugate base
How is a buffer made by partial neutralisation?
a buffer solution can also be prepared by adding an aq solution of an alkali such as NaOH, to an excess of weak acid. The weak acid is partially neutralised by the alkali forming the conjugate base. Some of the weak acid is left over unreacted. The resulting solution contains a mixture of the salt of the weak acid and any unreacted weak acid.
How do the acid and conjugate base paur remove acid and alkali (not process just equilibrium)
by shifting the buffer’s equilibrium system to the right or left
What happens when acid is added to a buffer?
Conjugate base removes added acid
[H+] increases
H+ ions react with he conjugate base, A-
the equilibrium position shifts to the left, removing most of the H+ ions
![<p>Conjugate base removes added acid</p><ol><li><p>[H+] increases </p></li><li><p>H+ ions react with he conjugate base, A-</p></li><li><p>the equilibrium position shifts to the left, removing most of the H+ ions </p></li></ol><p></p>](https://assets.knowt.com/user-attachments/2a7c176b-05b6-46c5-ab58-9cd3c5ab7906.png)
What happens when alkali is added to a buffer
Weak acid removes added alkali
[OH-] increases
the small concentration of H+ ions react with OH- ions H+ + OH- → H2O
The HA dissociates, shifting the equilibrium position to the right to restore most of H+ ions
![<p>Weak acid removes added alkali</p><ol><li><p>[OH-] increases</p></li><li><p>the small concentration of H+ ions react with OH- ions H+ + OH- → H2O</p></li><li><p>The HA dissociates, shifting the equilibrium position to the right to restore most of H+ ions </p></li></ol><p></p>](https://assets.knowt.com/user-attachments/30f9377a-1eaf-4496-8c3e-3803e3b2c0b1.png)
What are the concentrations of weak acid and conjugate base when a buffer most effective? and why
when there are equal concentrations of the weak acid and its conjugate base
when [HA]=[A-]
the pH of the buffer solution is the same as the pKa value to HA
the operating pH is typically over about 2 pH units, centred at the pH of the pKa value
![<p>when there are equal concentrations of the weak acid and its conjugate base</p><p>when [HA]=[A-]</p><ul><li><p>the pH of the buffer solution is the same as the pKa value to HA</p></li><li><p>the operating pH is typically over about 2 pH units, centred at the pH of the pKa value </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/ed680ff7-b33f-449b-aaa8-b13a1de2675a.png)
in buffers does [H+] = [A-]. give a reason
no
A- has been added one of the components of the buffer
What is the formula to work out the pH of a buffer solution>
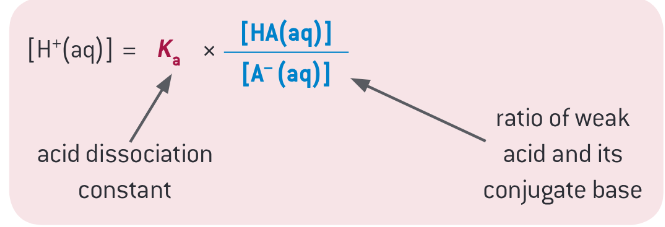
What happens to the buffer equation when the concentrations of HA and A- are the same?
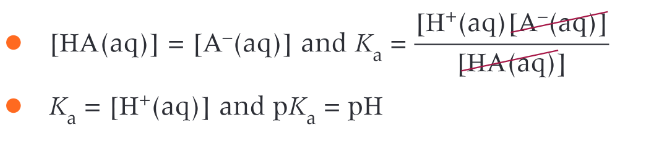

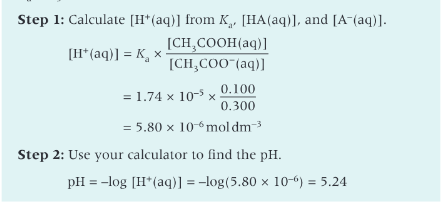




What pH does blood need to be maintained at?
between 7.35 and 7.45
normal blood should have a pH of 7.40
How is blood pH controlled?
by a mixture of buffers, with the carbonic acid-hydrogencarbonate buffer system being the most important (H2CO3/HCO3-)
What happens if the pH of the blood falls below 7.35?
people can develop a condition called acidosis, which can cause fatigue, shortness of breath and in extreme cases, shock or death
What happens if the pH of the blood rises about 7.45?
people can develop a condition called alkalisis, which can cause muscle spasms, light-headness and nausea
How does the carbonic acid buffer system work - what happens when acid and alkali is added?


How does the body prevent H2CO3 build up?
by converting it to carbon dioxide gas which is exhaled by the lungs

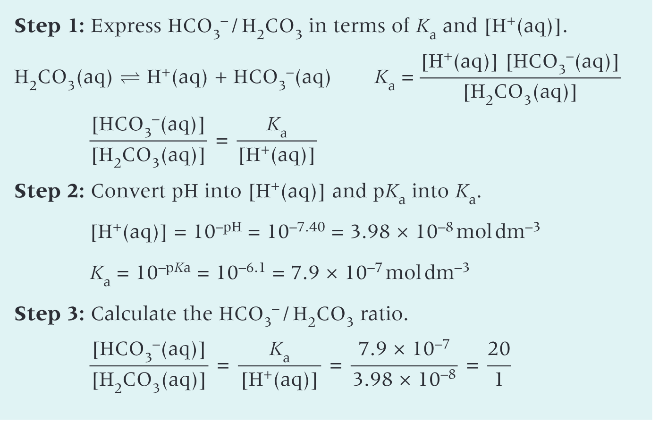
How would you use a pH metre to monitor the pH of an aq base being added to an acid solution - practical - carry out the practical so you can plot a titration curve with your results

What is an alternative automatic method for using the pH metre that allows automatic readings

Describe a titration curve
when the base is first added the acid is in great excess and the pH increases very slightly, As the vertical section is approached the pH starts to increase more quickly, as the acid is used up more quickly
eventually the pH increases rapidly during addition of a very small volume of base, producing a vertical section, only drops of solution will be needed for the whole vertical section
after the vertical section th pH will rise very slightly as the base is now in great excess
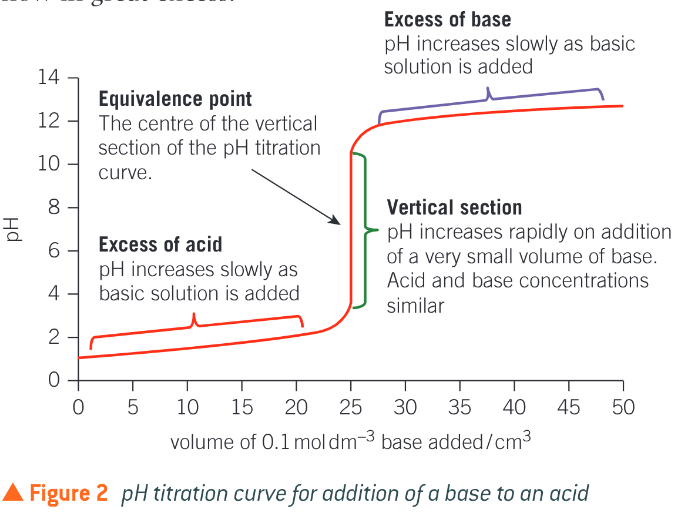
What is the equivalence point of the titration?
The centre of the vertical section of the pH titration curve. The volume of one solution that exactly reacts with the volume of the other solution. The amounts used match the stoichiometry of the reaction. - does not always mean it is when the solution is neutral
What does a base to acid titration curve look like?
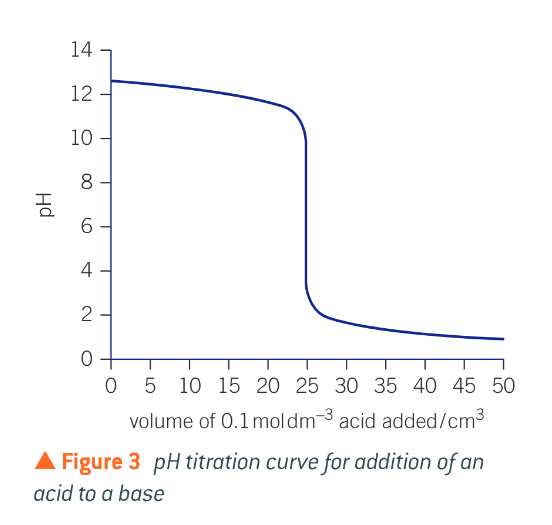
What is an acid-base indicator?
a weak acid that has a distinctively different colour from its conjugate base
What are the colour changes for methyl orange?
weak acid HA - red
conjugate base A- yellow
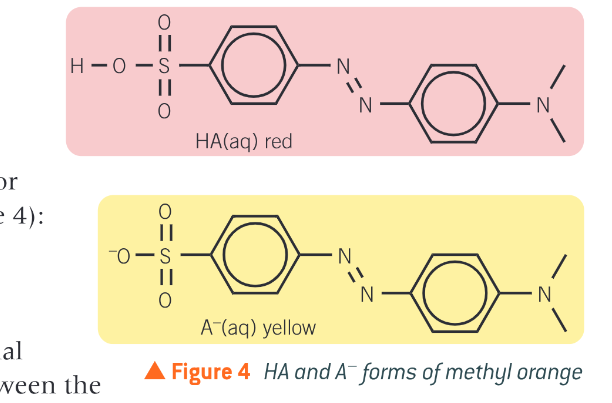
How do you determine the end point of a titration from an indicator? And how is this shown in methyl orange
at the end point the indicator contains equal concentrations of HA and A- and the colour will be in between its two extreme colours. For methyl organe the colour it its end point is orange.
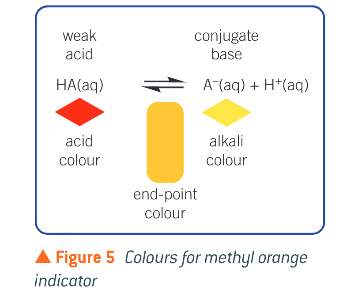
What happens to the weak acid inidicators as they change colour?
an indicator is a weak acid. The equilibrium position is shifted towards the weak acid in acidic conditions or towards the conjugate base in basic condition, changing the colour as it does so.

What is special at the end point of a acid base indicator - Ka and pKa
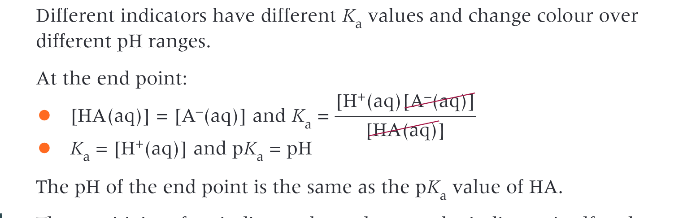
How do you choose an indicator?
you must use an indicator that has a colour change which coincides with the vertical section of the pH titration curve. Ideally the end point and equivalence point would coincide but this my not be possible..
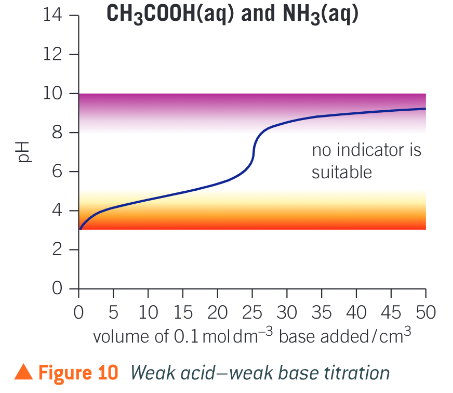
What indicator is suitable for a weak acid weak base titration?
no indicator as there is no vertical section
what does a strong acid strong base titration curve look like?
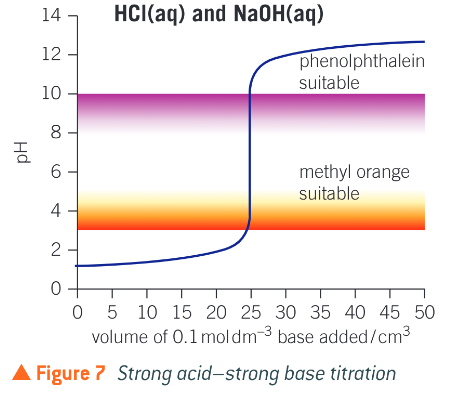
What does a weak acid strong base titration curve look like?
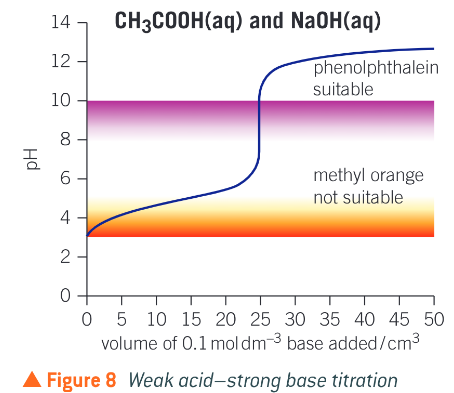
What does a strong acid weak base titration curve look like?
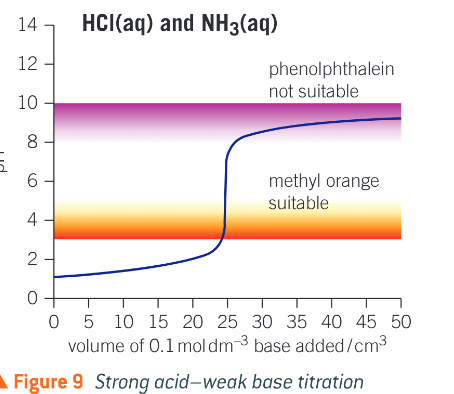
What does a weak acid weak base titration curve look like?