1.2 Conservation & Dissipation of Energy
1/21
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is the law of conversation of energy?
Energy can be transferred usefully, stored or dissipated, but cannot be created or destroyed.
In all system changes energy is ___________, so that it is stored in less ________ ways.
This energy is often described as being ‘________’.
dissipated
useful
wasted
How does a pendulum show the conservation of energy?
At its highest point of its swing it has maximum gravitational potential energy.
As it falls it gains kinetic energy which is its greatest at the mid part of its swing.
As it moves past the midpoint it decreases in kinetic energy and increases again in gravitational potential energy.
What energy transfers happen to a bungee jumper after jumping off the platform?
When the rope is slack, energy is transferred — _____________ __________ energy store → _________ energy store as the jumper _____________ towards the ground (due to the force of ________).
When the rope tightens, it ________ the jumper’s fall — the force of the rope reduces the _________ of the jumper.
Jumper’s __________ energy store decreases, rope’s __________ ____________ energy store increases as rope ____________.
Eventually the jumper comes to a stop — energy in __________ energy store of the jumper all transferred to the ___________ __________ energy of the rope.
After reaching the bottom, rope recoils and pulls jumper back up — as jumper rises energy in ___________ ___________ energy store of rope decreases and jumper’s ___________ energy store increases (until rope becomes ________).
After rope becomes ________ (and at the top of the ascent), jumper’s __________ energy store = 0 — jumper’s ______________ __________ energy store increases through the ascent.
gravitational potential
kinetic
accelerates
gravity
slows
speed
kinetic
elastic potential
stretches
kinetic
elastic potential
elastic potential
kinetic
slack
slack
kinetic
gravitational potential
What happens when there are energy transfers in a closed system?
There is no net change to the total energy.
What are two ways to reduce unwanted energy transfers?
Lubrication
Thermal insulation
How can lubrication reduce unwanted energy transfers?
Oil/grease can be used — slightly separates moving parts
Reduces friction when objects rub against each other
Decreases the amount of energy wasted as heat to the surroundings
How can thermal insulation reduce unwanted energy transfers?
Uses materials with low thermal conductivity — e.g. double glazing or loft insulation
Often works by trapping pockets of air — very poor thermal conductor
Slows down the rate of energy transfer — reduces the amount of useful thermal energy lost from a system
The higher the __________ _________________ of a material the higher the rate of energy transfer by ______________ across the material.
thermal conductivity
conduction
What is the rate of cooling of a building affected by?
Thickness of its walls
Thermal conductivity of its walls
How is the rate of cooling of a building affected by the thickness and thermal conductivity of its walls?
Higher thermal conductivity of the walls = higher rate of energy transfer — building will cool down faster
Using materials with low thermal conductivity slows down this heat loss
Thicker walls reduce the rate of thermal energy transfer — lowers the rate of cooling
What is the independent variable in RP 2 (Activity 1)?
Type of insulating material
What is the independent variable in RP 2 (Activity 2)?
Thickness of material
What is the dependent variable in RP 2?
Temperature decrease over time
What are the control variables in RP 2 (Activity 1)?
Volume of water
Initial temperature of the water
Thickness of the insulation
What are the control variables in RP 2 (Activity 2)?
Volume of water
Initial temperature of the water
Type of insulating material
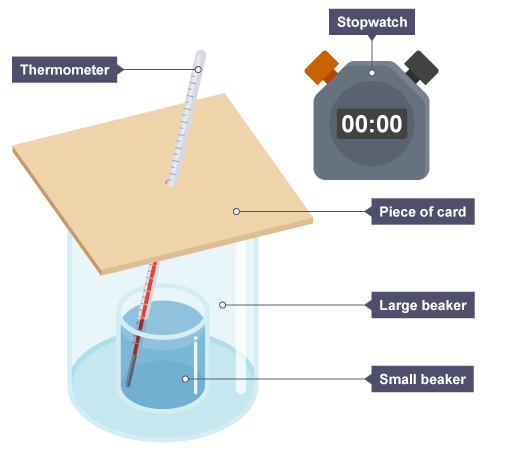
Describe a method for investigating the effectiveness of different materials as thermal insulators. (6)
Place a small beaker inside a larger beaker and pack the gap with an insulating material — leave one setup with no insulator as a control
Pour a fixed volume of hot water into the small beaker
Place a cardboard lid on top with a thermometer placed through a hole
Record the initial temperature (this should be the same for each material) and start a stopwatch
Record the temperature every 3 minutes for 15 minutes
Calculate the total temperature drop for each material (initial temperature - final temperature) — the one with the lowest value is the best thermal insulator
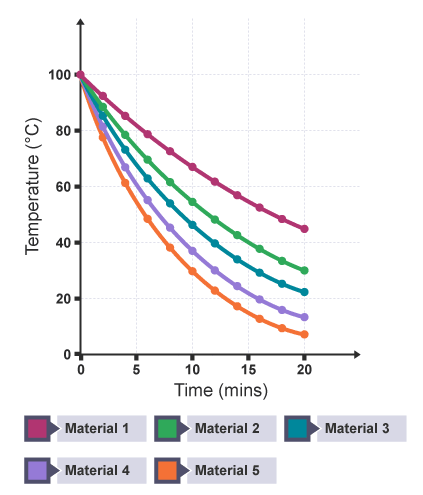
This graph shows results for the practical investigating the effectiveness of different materials as thermal insulators.
What does this graph show? (4)
The curve which takes the longest time for the water temperature to drop (the shallowest) should be the material that is the best insulator.
The temperature falls quickly at high temperatures and slowly at low temperatures.
When the beaker is at a high temperature, there is a big difference between the temperature of the beaker and the temperatures of the surrounding air — this means that there is a high rate of transfer.
When the beaker is at a lower temperature, there is less difference between the temperature of the beaker and the temperatures of the surrounding air — this means that there is a lower rate of transfer.
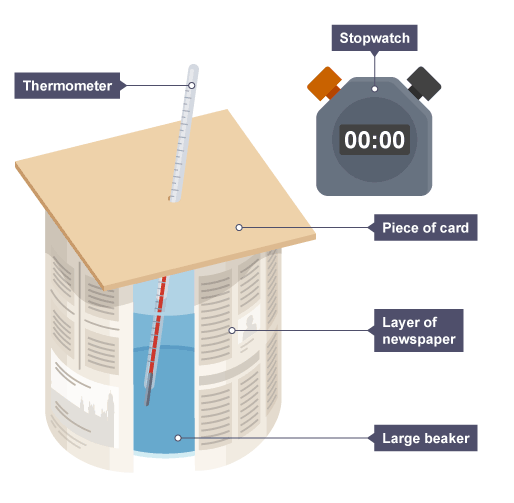
Describe a method for investigating how the thickness of a material affects the thermal insulation. (6)
Wrap a beaker in a single layer of an insulator — e.g. newspaper
Pour a fixed volume of hot water into the beaker
Add a cardboard lid and insert a thermometer into the water
Record the initial temperature (this should be the same for each round of the experiment) and start a stopwatch
Record the temperature every 3 minutes for 15 minutes
Repeat the experiment, adding more layers of the same material — compare the overall temperature drops (initial temperature - final temperature)
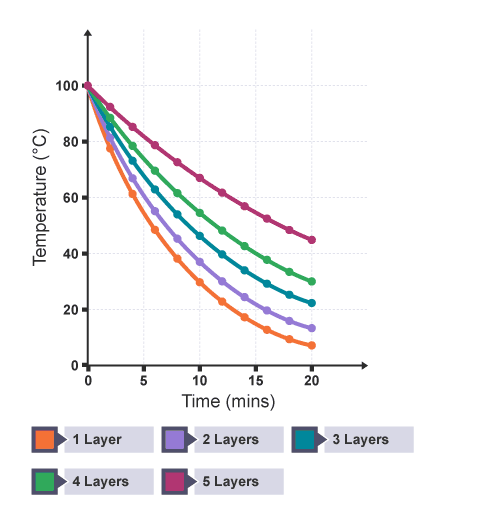
This graph shows results for the practical investigating how the thickness of a material affects the thermal insulation.
What does this graph show? (5)
The curve which takes the longest time for the water temperature to drop (the shallowest gradient) shows the amount of layers that provide the best insulation.
The curve for no insulation has the steepest gradient at any given time interval.
As the number of layers increases, the gradient of each curve decreases at any given time interval.
Having more layers increases the insulation which means temperature drops more slowly — the thickest insulation has the lowest rate of cooling.
The temperature falls quickly at high temperatures and slowly at low temperatures.
What is the equation for the energy efficiency for any energy transfer (energy)?
efficiency = (useful output energy transfer / total input energy transfer) × 100
What is the equation for the energy efficiency for any energy transfer (power)?
efficiency = (useful power output / total power input) × 100