2.4D Addition and Condensation Polymers
1/12
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
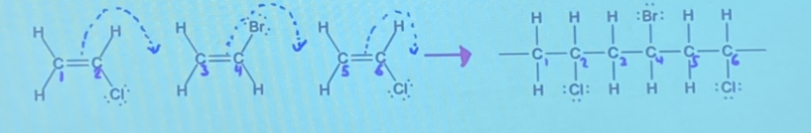
What are Addition Polymers?
Very long organic molecules formed through addition reactions between monomers with unsaturated carbon-carbon bonds.
Condensation Polymers
Very long organic molecules formed through condensation reactions between monomers with two reactive functional groups, resulting in the release of a small molecule.
Functional Group
A specific group of atoms within a molecule that determines its chemical properties and reactivity.
Carboxylic Acid
A functional group consisting of a carbon atom double-bonded to an oxygen atom and single-bonded to a hydroxyl group.
Alcohol
A functional group consisting of a hydroxyl group (-OH) bonded to a carbon atom.
Amine
A functional group consisting of a nitrogen atom bonded to one or more carbon atoms.
Esterification
The reaction between a carboxylic acid and an alcohol, resulting in the formation of an ester.
Peptide Bonds
The bonds formed between a carboxylic acid and an amine during a condensation reaction.
Condensation with Two (Different) Monomers
The reaction between a monomer with the same functional group at either end and a monomer with a different functional group at either end.
Polyester
A polymer formed from condensation reactions that produce ester linkages between monomers, with monomers having an -OH and a -COOH functional group.
Polyamide
A polymer formed from condensation reactions that produce amide linkages between monomers, with monomers having an -NH2 and a -COOH functional group.
Hydrolysis
The reverse of condensation reactions, where the linkage formed by condensation is split up by a molecule of water.
Addition Polymers vs
Addition polymers contain only carbon-carbon single bonds and are slow to break down, while condensation polymers can be made to be biodegradable and can be broken down by certain bacteria through hydrolysis.