2.4D Addition and Condensation Polymers
Addition Polymers:
Polymers are created through polymerization reaction. Addition polymers are very long organic molecules formed via addition reactions between monomers with unsaturated (double bonds) carbon carbon bonds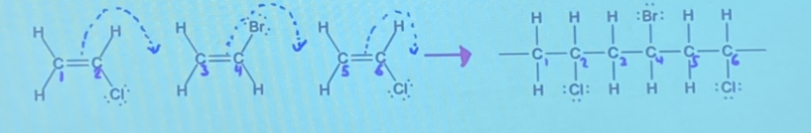
In this reaction, the double bonds break and the electrons in the bond are used to form a covalent bond with another monomer
Condensation Polymers:
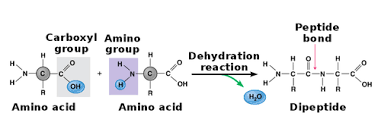
Condensation polymers are very long organic molecules formed as a resultt condensation reactions between monomers that have two reactive functional groups, one on each ed. For every one between monomers, a small molecule is released.
Functional Group Refresher:
Functional Group | |
|---|---|
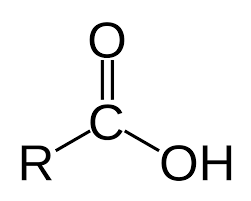
Carboxylic Acid |
|
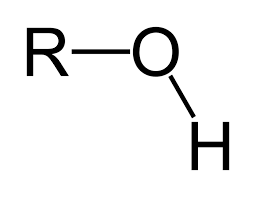
Alcohol |
|
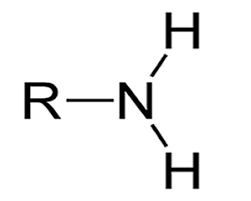
Amine |
|
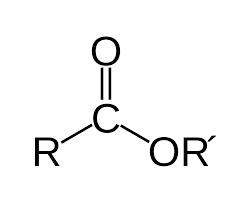
Esterification carboxylic acid + alcohol |
|
Peptide Bonds carboxylic acid + amine |
|
Condensation with Two (Different) Monomers:
One monomer with the same functional group at either end can react with a monomer that has a different functional group at either end.
e.g. a diol (two hydroxyl groups) and a dicarboxylic acid
Condensation of the Same Monomer: Polyesters
A polyester is a polymer formed from condensation reactions that produce ester linkages between monomers. Monomers have two functional groups: an -OH and a -COOH
Condensation of the Same Monomer: Polyamides
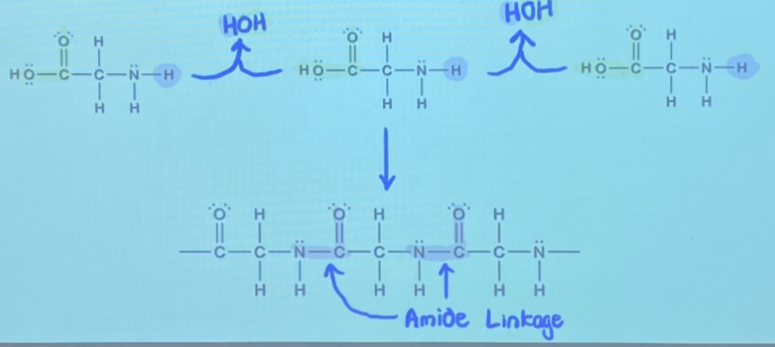
Polyamides are polymers formed by condensation reactions resulting in amide linkages between monomers. Monomers have two functional groups: an -NH2 and a -COOH
 Hydrolysis
Hydrolysis
The reverse of condensation reactions is called hydrolysis, where the linkage formed by condensation is split up by a molecule of water.
All biological macromolecules form by condensation reactions and break down by hydrolysis.
Addition Polymers vs. Condensation Polymers
Addition polymers contain only carbon-carbon single bonds; these bonds are very stable. Plastics made up of addition polymers are very slow to break down
Condensation polymers can be made to be biodegradable. Certain bacteria can hydrolyze the amide and ester bonds between monomers to break polymer chains into smaller chains.