2.2 ELEMENTS/ ATOMS LECTURE
1/16
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
17 Terms
what makes up matter?
elements
can elements be broken down into simpler substances by ordinary chemically methods?
no
what are the 4 elements that make up the human body?
CHON. Carbon, hydrogen, oxygen, and nitrogen
define atom
derived from greek word atomos, means indivisble, they make elements and each atom has unique properties . (can infer properties by periodic table)
how many and what parts make up an atom?
3 parts . protons , neutrons, and electrons
define amu
stands for atomic mass unit. weight used to describe 1 proton or neutron. (electrons are too small to be weighed)
define protons
has positive charge. is equal to one amu. located in nucleus.
define neutrons
has neutral charge. is slightly heavier than 1 amu. located in nucleus.
define electrons
located in outer electron shells. has negative charge . so small their weight is not considered.
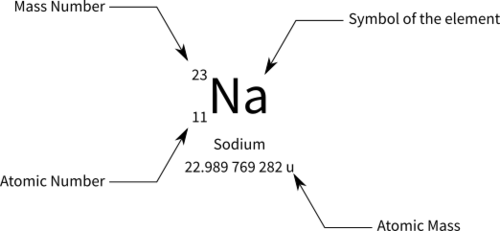
define atomic symbol
letter on periodic table that stands for element name
what is located in the nucleus?
protons and neutrons
Isotopes
an atom of the same element that has same # of protons with varying neutrons.
Radioisotope
isotopes that decay to stable form. Neutrons will decay to protons causing atom to change to different element. # of electrons DOES NOT equal number of protons for radioisotopes. This release of energy = radiation hence “radio” isotope.
If radioisotope loses half of itself this is called?
half life
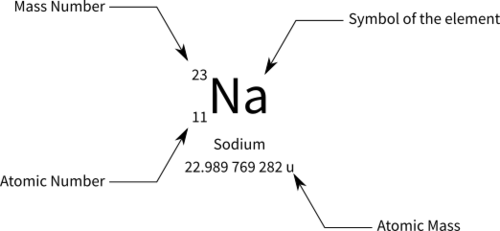
atomic number
number of protons in nucleus. written as subscript left of atomic symbol
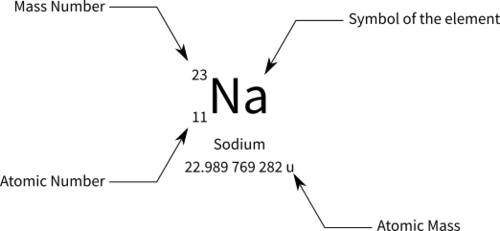
mass number
number of protons and neutrons in nucleus. total mass written as super script. (this is NOT the same as atomic wieght)
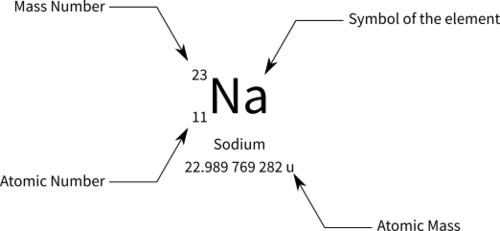
Atomic weight
average weight of all mass numbers , isotopes, etc recorded.