Unit 9: Global Change
{{9.1-9.2 Stratospheric Ozone Depletion{{
^^Good Ozone^^
- The protective layer in the stratosphere
^^Bad Ozone^^
- “Ground-level”; photochemical oxidant (sunlight reacts with SO2 and NOs)
^^The function of the Ozone Layer^^
- Absorbs the sun’s UV Rays, allowing just enough for life on earth
^^Formation of the Ozone Layer^^
- Step 1 of Ozone Formation: O2 + UV-C → O + O
- Step 2 of Ozone Formation: O +O2 → O3
^^What are CFCs:^^
- Chlorofluorocarbons
- Nontoxic, nonflammable, very stable
- Contains C, CL, and F
- Found in refrigerants, propellants (aerosols), foam, and packing materials
- So Cl acts as a catalyst—it aids another reaction but is not getting used up itself
- 1 Cl atom can break down 100,000 ozone molecules before it bonds with another Cl atom, creating Cl2 and stopping the reaction
^^How do CFCs break down ozone?^^
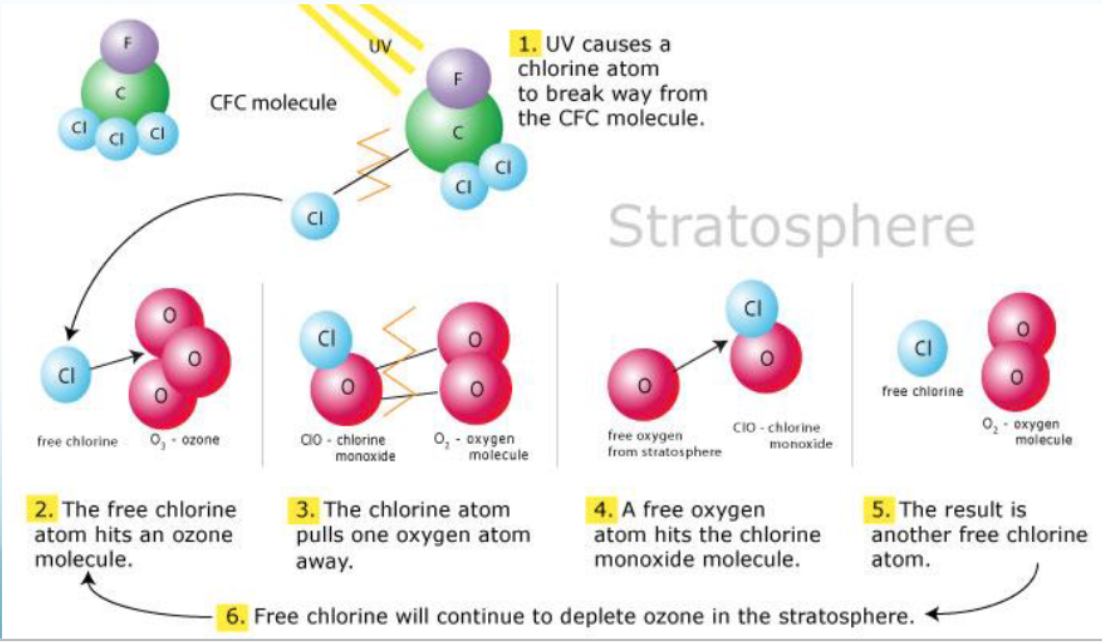
- UV causes a chlorine atom to break away from the CFC molecule
- The free chlorine atom hits an ozone molecule
- The chlorine atom pulls one oxygen atom away.
- A free oxygen atom hits the chlorine monoxide molecule
- The result is another free chlorine atom
- Free chlorine will continue to deplete ozone in the stratosphere
^^International Action- The Montreal Protocol^^
- Montreal Protocol: Gradually eliminating the production and consumption of ozone-depleting substances to limit their damage to the earth’s ozone layer.
- Considered to be the most successful environmental global action.
^^Reversing Stratospheric Ozone Depletion^^
- Stop producing ozone-depleting chemicals immediately
- It will take at least 60 years to 1980 levels
- Agreements with a prevention approach
- Montreal protocol: Cut emissions of CFCs
- Copenhagen Amendment: Accelerated phase-out of CFCs
^^Natural Capital Degradation→Effects of Ozone Depletion^^
- Human Health and Structure
- Worse sunburns
- More eye cataracts and skin cancers
- Immune system suppression
- Food and Forests
- Reduced yields for some crops
- Reduced seafood supplies due to smaller phytoplankton populations
- Decreased forest productivity for UV-sensitive tree species
- Wildlife
- More eye cataracts in some species
- A shrinking population of aquatic species sensitive to UV radiation
- Disruption of aquatic food webs due to shrinking phytoplankton populations
- Air Pollution and Climate Change
- Increased acid deposition
- Increased photochemical smog
- Degradation of outdoor painted surfaces, plastics, and building materials
- While in the troposphere, CFCs act as greenhouse gases
{{9.3 The Greenhouse Effect{{
Core Case Study: Melting Ice in Greenland
- World’s largest island: Mostly covered by ice glaciers
- Glaciers melting at an accelerating rate in summers
- Atmospheric warming is a crucial factor
- Greenland’s ice loss: Responsible for nearly one-sixth of the global sea-level rise over the past 20 years
^^How and Why Is the Earth’s Climate Changing?^^
- Scientific evidence strongly indicates that the earth’s atmosphere is warming at a rate that is likely to lead to significant climate change
- Weather: Short-term changes
- Climate: Average weather conditions of a particular area over 30 years or more
^^Climate and the Natural Greenhouse Effect^^
- The greenhouse effect is a natural process
- Heat-holding gases absorb heat
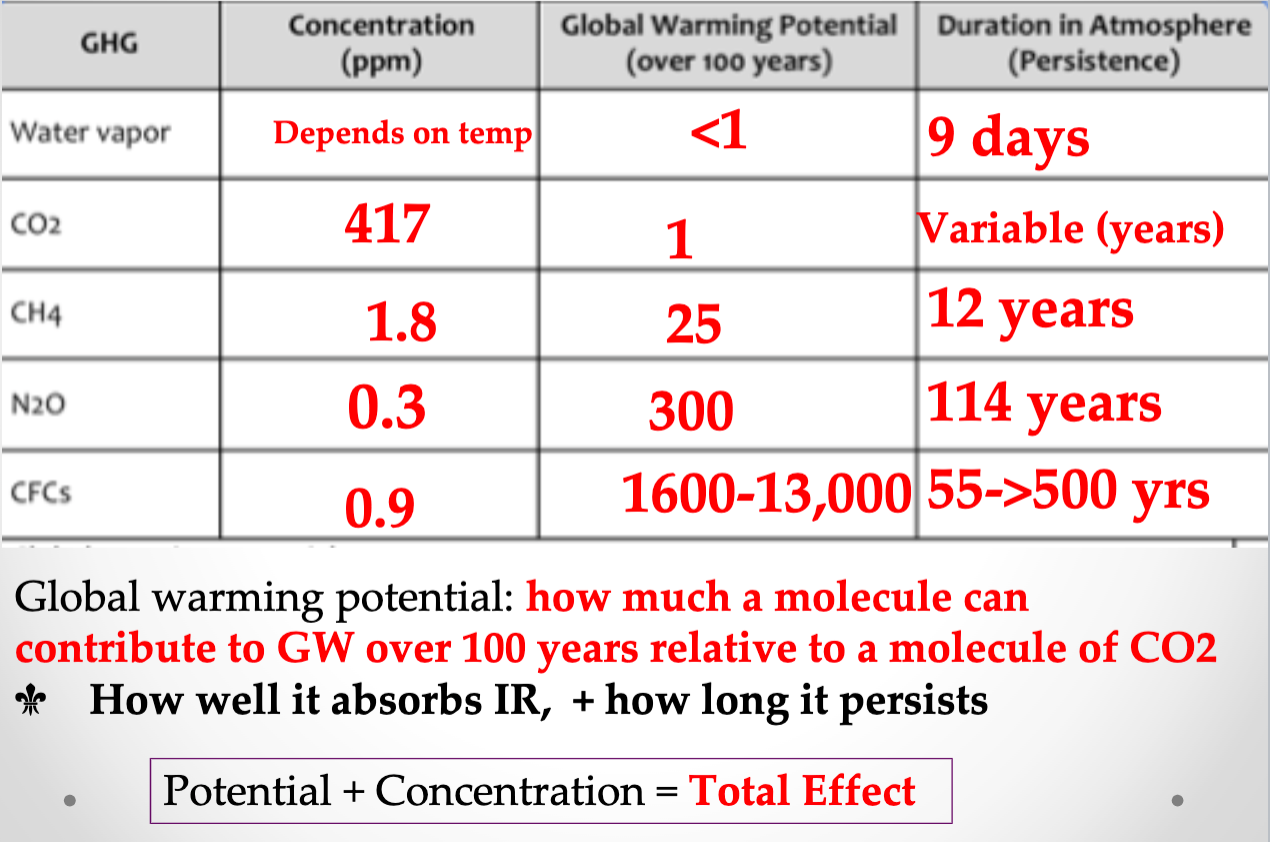
- CO2, CH4, N2O
- Historical changes in the amount of carbon dioxide in the atmosphere correlate to changes in global temperatures
^^Earth’s Greenhouse Effect^^
- Incoming solar radiation: UV and visible light
- 1/3rd reflected back into space
- The remaining light is absorbed by clouds and the planet’s surface
- These become warmer, emit infrared radiation
- This radiation is either absorbed by the GHGs or emitted into space
- More GHGs = more IR absorbed & emitted back to earth
This process should be in equilibrium; short term, inputs might be higher or lower, which causes global warming/cooling (ice ages)
- N2 and O2 = 99% of the atmosphere- NOT GHGs
- H2O Vapor: Absorbs more IR than anything else, but does not persist
- CO2
- CH4 (methane)
- H2O (nitrous oxide)
- O3 (ozone)
- CFCs (not natural)
^^Natural Sources of GHGs^^
- Volcanoes: ash reflects incoming radiation, so a cooling effect
- Decomposition & Digestion: Dead organic matter is converted into CO2 unless there is not enough O2. If there is not enough O2, it’s converted to CH4 (methane)
- Methane sources
- Wetlands
- Termites
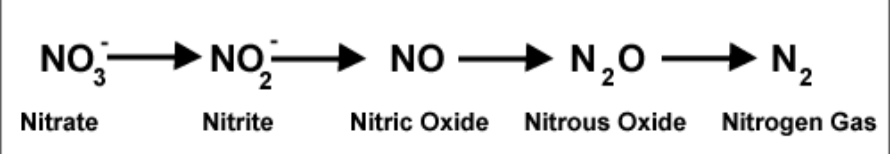
- Denitrification: How we get N2O or nitrogen cycle
- Evaporation and Evapotranspiration: water vapor
^^Anthropogenic Sources of GHGs^^
- Fossil Fuels: CO2, methane, N2O
- PM lowers albedo (black soot) causing a cooling effect
- Agriculture
- Overirrigation: low O2 so methane-like wetlands
- Fertilizers: identification so an increase in N2O
- Livestock: cattle and sheep create methane
- Deforestation: less photosynthesis, more CO2 burning releases PM, methane, N2O as well
- Landfills: Methane
- Modern chemicals like CFCs: HCFCs (have replaced CFCs) also GHGs
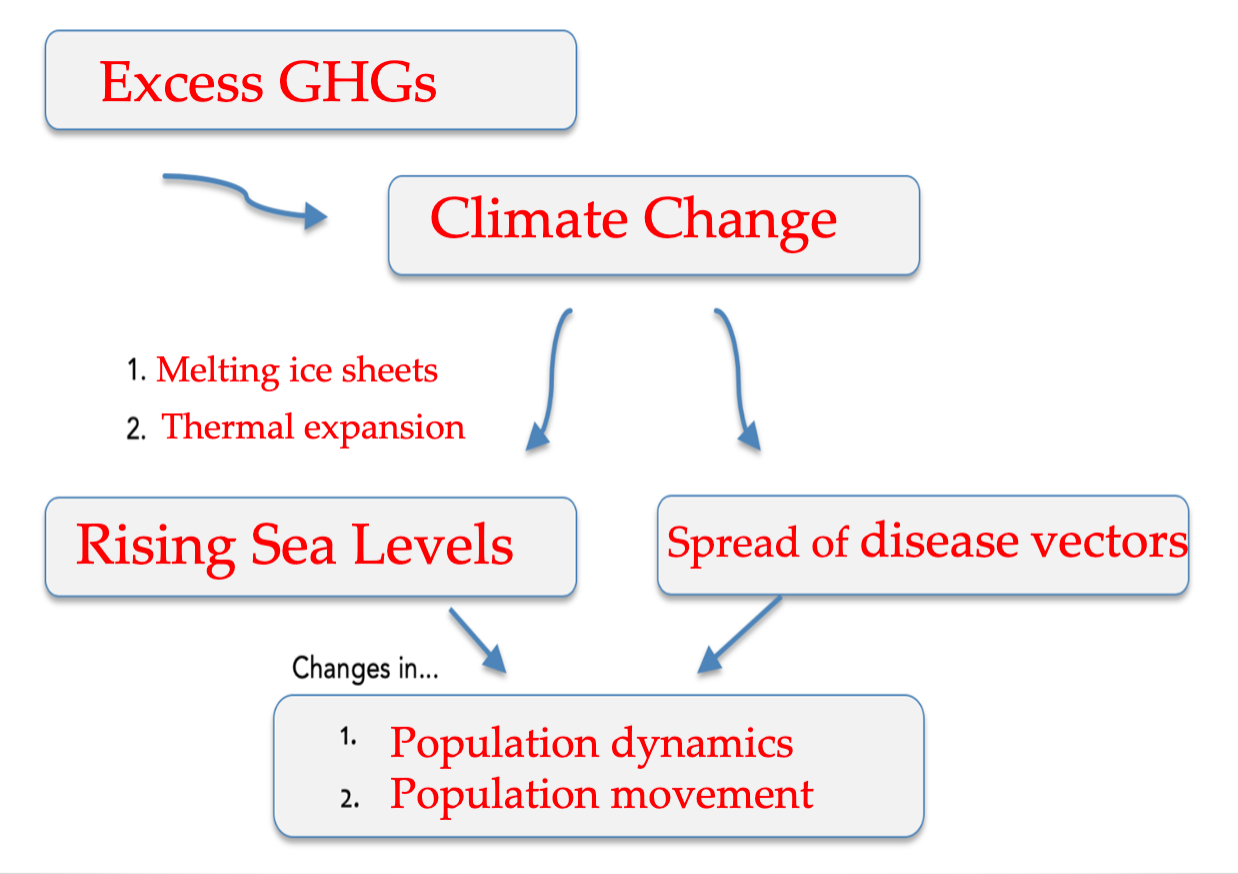
^^Threats to Human Health and the Environment^^
- Thermal expansion of ocean water
- Disease vectors spreading from the tropics to the poles
- Rising sea levels
- Population movements
- Global climate change
- Population dynamics
- Excess GHGs in the atmosphere
- Melting ice sheets
{{9.5-9.7 Global Climate Change{{
- Global change: Planetary scale changes in Earth systems land, air life, soil, atmosphere, oceans, humans
- Global climate Change: changes in temperature, precipitation, wind, storms, currents, etc.
- Global warming: Increase in average temp of earth over time
^^Causes^^
- Increasing CO2 concentration
- Keeling Cure: First to accurately measure CO2
- CO2 levels vary seasonally and increase overall
- Why is there a seasonal fluctuation?
- Vegetation, most of it is in the northern hemisphere so overall there is a huge decrease in our spring/summer
- Why is there a latitudinal difference?
- 90% of the world’s population is in the northern hemisphere
- 60% of the world’s land
- However, phytoplankton (single-celled algae) produce > 40% of the world’s oxygen
- Why does it increase overall?
- Increased emissions from fossil fuel combustion
- Net destruction of vegetation
- Temperature Increases: Global temperatures have increased on average 1.4F, but regionally up to 4F
- Changing species compositions
- Proxy data: Preserved physical characteristics of the environment that can stand in for direct measurement
- Foraminifera: different species prefer different temps; very narrow range
- Fossilize well
- Sedimentary layers of the ocean floor
- Ice cores
- Data from 500,000 years
- Air bubbles are trapped each year as layers of ice laid down
- Ice in warmer temps has more O-18 isotopes (heavy)
- For half a million years, CO2 has never been above 300 ppm; last 60 years has risen to >400 ppm
- Methane and N2O have also increased
- Tree rings
- Coral growth rings
- Pollen spores
- What caused CO2/Temp shifts before humans?
- Path of orbit/position relative to the sun
- Orbital tilt
^^Developing countries overtook developed in CO2 emissions^^
- Industrialization happens first→ through fossil fuel combustion in stage 2
- There is no money or political will to pass progressive environmental laws until phases III or IV
^^We use climate models to make predictions^^
- Air/ocean temperatures
- Concentrations of CO2, other GHGs
- Amount of vegetation/sea ice
^^Effects of Global Warming^^
- Polar ice melting
- Sea level rise
- Polar bear habitat is lost
- Glaciers melting
- Lack of water supply
- Permafrost melting
- Lakes drain deeper into the ground, lose water
- Cause erosion with loss of structural support
- Organic matter decomposes and releases methane
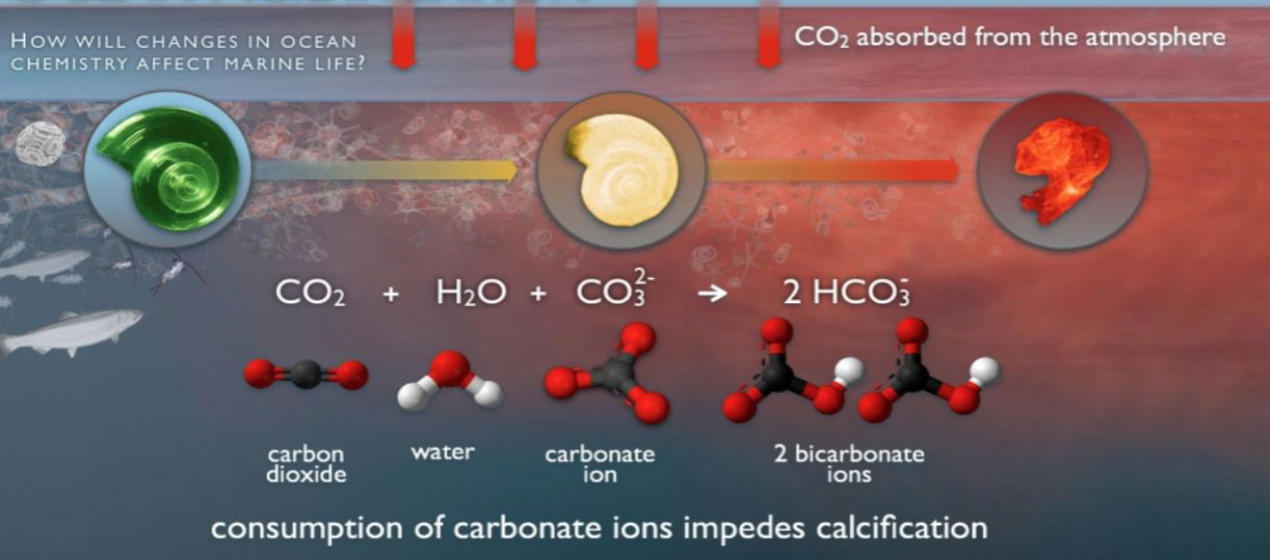
- Ocean acidification: CO2 dissolves in water to form H2CO3 (carbonic acid). Dissolves shells/skeletons of marine organisms (crustaceans, mollusks, coral)
- Sea level Rise
- Has risen by 9 in (2.2 cm)
- By 2100, 7-23 in. more
- Total volume increases as land ice melts
- Water expands as it gets warmer (thermal expansion)
- Effects→flooding of coastal nations and towns, saltwater intrusion into aquifers, increased erosion
- 100 million people live within 3 ft. of sea level
^^Effects on Organisms^^
- Ranges have shifted toward both poles
- Plants flower earlier, birds, migrate earlier, insects emerge earlier
- Coral bleaching: range of temperature tolerance is small
- Fragmentation prevents migration
^^Future Effects^^
- Heat waves
- Increased energy demand
- Risk of death to poor and elderly
- Damage to crops and increased irrigation needs
- Cold spells
- May have some positive effects
- Expands the range of pest species
- Precipitation Patterns
- Where there’s less, crops require more irrigation
- Where there’s more, there is flooding, landslides, erosion
- Storm Intensity
- Ocean warming causes more hurricanes
- Ocean currents
- Thermohaline circulation: freshwater dilutes the ocean and stops salt water from sinking near Greenland
^^Effects on Humans^^
- Relocation/Climate refugees
- Health→ heat waves, infectious diseases
- Tourism→ snow, coral reefs
^^Positive Effects of Climate Change^^
- Fewer deaths, crop damage due to cold
- New habitats become hospitable to humans
- Higher rainfall = recharge aquifers, more crops
^^1997: Kyoto Protocol^^
- Kyoto Protocol: Set goals for global emissions of GHGs to be reduced by 2012
- Different amounts required from different countries---US 7%
- Developing (China/India) had no limits
- The US would not ratify
^^Precautionary Principle^^
- Reduce emissions: Increase fuel efficiency, renewable energy
- Carbon sequestration: return agricultural lands to pasture/forest
- Capture CO2 from emissions and pump underground or into the ocean
{{9.8-9.9 Invasive and Endangered Species{{
^^Introduced Non-native Exotic^^
- Brought outside of historical range by humans
- does not necessarily have negative impacts
^^Invasive^^
- Has a negative impact on the ecosystem
- May spread rapidly
- Outcompete natives, disrupt food webs
- May have been introduced on purpose or accidentally
^^Illegal Trade^^
- Lacey Act (1900): (one of the earliest environmental laws) prohibits the transport of illegally harvested plants and animals
^^International treaty CITES (1973)^^
- Convention on International Trade in Endangered Species of Wild Fauna and Flora
- IUCN Red List: threatened species. In each country, an agency is assigned to monitor the import and export of species on the list. (US Fish & Wildlife)
- Illegal trade = $5-20 billion/yr
{{9.9 Endangered Species{{
^^Causes of Endangerment--Natural^^
- Starvation
- Excessive predation
- Loss of habitat through natural means
- Impediments to mating
- Natural disaster
- Being outcompeted by other similar species
^^Causes of Endangerment--Anthropogenic^^
- HIPPCO
- Habitat Destruction
- Degradation
- Fragmentation
- Invasive Species
- Pollution
- Population
- Climate Change
- Overexploitation
- Overhunting
- Overharvesting
- Overfishing
^^Selective Pressures^^
- Selective Pressures: Any factors that change the behaviors and fitness of organisms within an environment
- “Selective” = Natural selection = leads to evolution
- Not all species are equally impacted by selective pressures and ecosystem changes
- Adaptability
- Mobility
^^Protecting Endangered Species^^
- Criminalizing poaching
- Protecting animal habitats
- Factors influencing ecosystem conserve nation plans
- Size, shape, connectedness
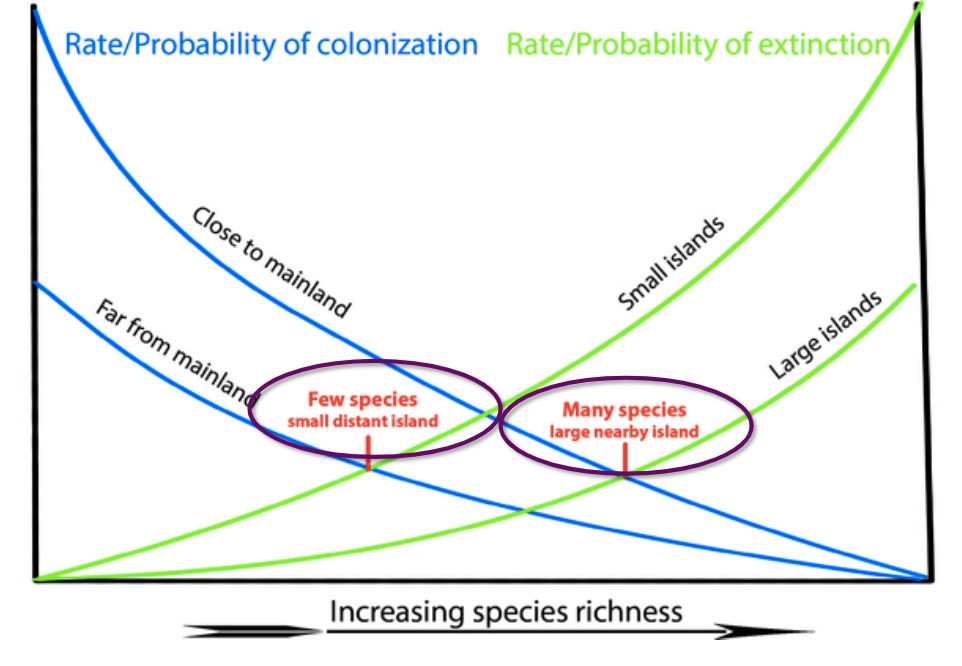
- Theory of island biogeography
- Metapopulations: Spatially separated pops of the same species, interact and interbreed
- SLOSS (Single Large or Several Small)
- Edge habitat
- Biosphere reserves: contain zones that vary in the allowed amount of impact. The central core, buffer zone, transition area
- Legislation
- Single species legislation
- More traditional option
- Become experts on important species
- Focus $/resources/efforts
^^Which species “deserve” this focused attention?^^
- Indicator: Quickly reflected changes in ecosystems functioning
- Keystone: Has a disproportionate, far-reaching impact on species in its ecosystem
- Flagship/Charismatic: Popular with humans due to their '“cuteness”, large size, and/or high intelligence. Serves to raise concern and awareness
- Umbrella: The conservation and protection of these species indirectly affect the conservation and protection of other species within their ecosystem
^^Limitations of the single-species approach^^
- Protecting HABITAT instead allows the ecosystem to be more flexible/natural
- Protects species/relationships we’re not even aware of
- Focus $/efforts on “hot spots” of biodiversity (rainforests, coral reefs)
^^Marine Mammal Protection Act (1972)^^
- Marine Mammal Protection Act (1972): Prohibits killing any marine mammals in the US and any import/export of MM body parts
^^Endangered Species Act (1973)^^
- Endangered Species Act (1973): Authorizes the USFWS to determine which species can be listed as threatened or endangered. Prohibits their harm as well as the trade of fur/other body parts
- Endangered: Danger of extinction within the foreseeable future
- Threatened: Likely to become endangered in foreseeable future throughout all or a significant portion of its range
^^Instrumental (Extrinsic) Value^^
- Goods and services
- Recreation
- Aesthetics
- Medicine/research
- Genetic info
^^Intrinsic Value^^
- Right to exist regardless of practical use to humans
- E.O Wilson
- Biophilia: inherent genetic kinship with the natural world
{{9.10 Human Impacts on Biodiversity{{
^^Why is our current estimate too low?^^
- Population growth, resource consumption, and climate change continue to increase
- The actual number of species is unknown
- Very expensive and difficult to accurately categorize all extant species
- New species are being discovered all the time
- Rates are even higher in the centers of highest biodiversity--a hotspot
- Terrestrial- rainforests
- Marine- coral reefs
- Potential habitats for new species are increasingly degraded and fragmented→speciation crisis
^^How does climate change impact habitat loss?^^
- Changes in temp and precipitation outside of species’ range of tolerance
- The sea level rising is destroying polar and coastal habitats