Chemistry - Physical
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
Name and angle for a molecule with 2bp and 0lp
Linear, 180 degrees
How is relative abundance of an ion determines in the TOF mass spectrometre?
Ions hit the detector and gain an electron. The size of the current is proportional to the relative abundance
Name and angle for a molecule with 2bp and 2lp/1lp
V-shaped or bent, 104.5/117.5 degrees
Name and angle for a molecule with 3bp and 0lp
Trigonal Planar, 120 degrees
Name and angle for a molecule with 3bp and 1lp
Trigonal Pyramidal, 107 degrees
Name and angle for a molecule with 4bp and 0lp
Tetrahedral, 109.5 degrees
Name and angle for a molecule with 4bp and 2lp
Square Planar, 90 degrees
Name and angle for a molecule with 4bp and 1lp
Trigonal Bipyramidal, 180 and 117.5 degres
Name and angle for a molecule with 5bp and 0lp
Trigonal Bipyramidal, 120 and 90 degrees
Name and angle for a molecule with 6bp and 0lp
Octahedral, 90 degrees
How does electronegativity change as you go down a group?
Electronegativity decreases, as there are more shells so more shielding and increased atomic radius. The attraction between the nucleus and outer electron decreases so it is harder to pull electrons in.
How does electronegativity change as you go across a period?
It increases as nuclear charge increases. This means that electrons are attracted more strongly so it makes the atom more electronegative.
What is dative bonding? In ammonium ion from ammonia for example?
When both of the electrons in a bond are supplied by one atom, it is indicated by an arrow from the lone electron pair. The N in ammonia will supply both electrons to the extra H+ ion to form a dative bond to make the ammonium ion (NH4+)
What is electronegativity?
The power of an atom to attract a pair of electrons in a covalent bond towards itself.
How is dipole-dipole bond formed?
They occur between polar molecules and arise because of a great difference in electronegativity. The positive reigon of one molecule is attracted to the negative reigon of another molecule.
How does hydrogen bonding occur?
A hydrogen directly bonds to a FON element which has a lone pair of electrons
How to vdw forces arise?
It acts as an induced dipole. In any molecule, electrons are constantly moving around randomly. At one point the electron in one atom or molceule will repel a neighbouring electron from another atom or molecule. Parts of the molecule become more or less negative and a temporary dipole is induced. An attraction between the delta positive and negative reigons form.
How are vdw forces stronger?
When a molecule has more electrons or is bigger.
What affects ionisation energy?
Increasing atomic radius and a greater positive charge.
What affects the strength of the metallic bond?
More protons, more delocalised electrons and a smaller ion increase the strength
What is average bond enthalpy?
The energy needed to break one mole of a covalent bond in a molecule in the gaseous state.
What does an exothermic reaction tell you about the system?
There is a negative enthalpy change so temperature of system increases
What does an endothermic reaction tell you about the system?
Enthalpy change is positive so temperature of the system decreases.
What is the standard enthalpy of formation?
The enthalpy change when one mole of a product is formed from its constituent elements with all reactants and product in their standard states in standard conditions. These being 298k and 100kPa
What is the standard enthalpy of combustion?
The enthalpy change when one mole of a compound is completely burned in oxygen will all substances in their standard states in standard conditions.
How can you improve the experiment for enthalpy changes?
Add insulation to the sides of the container, use a copper calorimeter (to improve heat transfer to water), stir constantly (improved heat distribution)
Equation for bond enthalpy
Bonds broken-bonds made
Why is average bond enthalpy inaccurate?
They are a generalised value as the true bond enthalpy is different for the same bond in a different environment within the same molecule.
What is the rate determining step?
The slowest step in a reaction and the step which has the highest activation energy.
What is the rate of reaction?
Change in concentration per unit time
Why do some collisions not lead to a reaction?
The particles do not collide in the right direction, they do not have the minimum kinetic energy to collide.
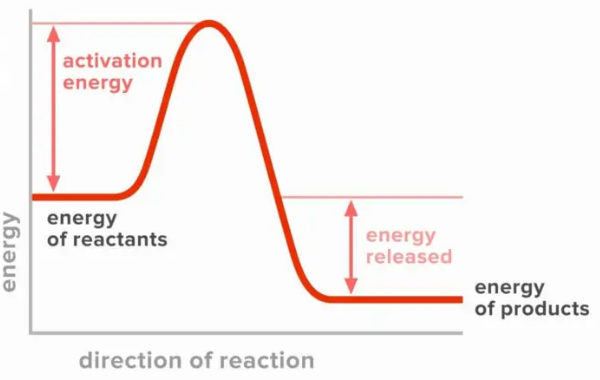
What type of reaction is this?
exothermic
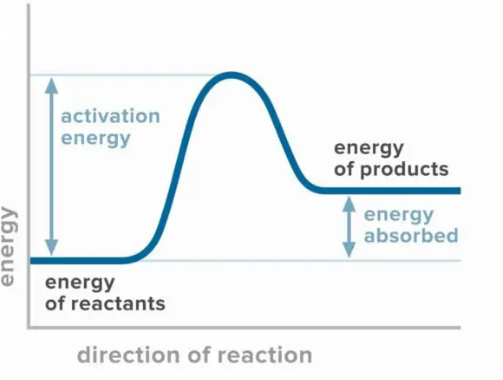
What type of reaction is this?
endothermic
What does the maxwell boltzman distribution curve show?
The distribution of energy between molecules in a solution at a specific temperature
Why does the MBD curve start at 0?
No particles have zero kinetic energy
What does the area under the curve of the MBD curve show?
The total number of molecules
What does the peak of the MBD curve show?
The most probable energy
Where would the mean energy of the particles be shown on the MBD curve?
A line slightly right to the most probable energy
What can the particles to the right of the AE in the MBD curve do?
Start a reaction because they have enough energy to collide
Why do small increases in temperature lead to higher increases in rate?
Increasing temperature means there are many more particles with energy greater than or equal to the activation energy so there are more frequent successful colisions per unit time.
How do catalysts affect rate?
They increase the rate of reaction by providing an alternative pathway that has a lower activation energy. The catalyst is unchanged at the end.
How will the MBD curve change with the effect of the catalyst?
Area to the right of the line for activation energy increases
In an experiment to determine rate of reaction, when samples of the reaction mixture is removed for a titraiton, what must be done to each sample before its titrated?
The sample must be quenched by dilution to stop the rate of reaction increasing between the point where you remove the sample and when you actually titre it.
What is enthalpy change?
The heat energy change at a constant pressure
What is the enthalpy of lattice formation?
enthalpy change when one mole of a solid ionic compound is formed from its gaseous ions under standard conditions
What is the lattice enthalpy of dissociation?
Enthalpy change when one mole mole of a solid ionic compound dissociates into its gaseous ions under standard conditions
What is the enthalpy of atomisation?
Enthalpy change when one mole of gaseous atoms are made from an element in its standard state.
What is the first electron affinity?
Enthalpy change when an electron is added to one mole of gaseous atoms to form gaseous ions with a 1- charge
What is the enthalpy of solution?
Enthalpy change when one mole of a solid ionic compound fully dissolves in a minimum volume of solvent to ensure no further enthalpy change is observed.
What is the enthalpy of hydration?
Enthalpy change when one mole of gaseous ions are dissolved in water to form an aqueous solution.
What is bond dissociation enthalpy?
Enthalpy change when one mole of gaseous molecules form 2 moles of gaseous atoms.
What is first ionisation energy?
Enthalpy change when one mole of electrons are removed from one mole of gaseous atoms to form one mole of gaseous ions each with a 1+ charge.
Why is the electron affinity for Cl/F a negative value?
There is an attraction between the nucleus and the added electron. Energy is released when an electron is gained.
Why is the second electron affinity a positive value?
Adding a negative electron to an already negatively charged ion means there is a repulsion between them. Energy is gained when an electron is added.
If the experimental value for enthalpy of lattice formation (from the BHC) is different to the theoretical value, what does it say about the compound?
It contains some covalent character as well as some ionic bonding.
How is the amount of covalent character in an ionic bond affected?
The amount the positive ion polarises the negative ion
How are lattice enthalpies affected?
The size of the ion, the larger the ion the less negative the lattice enthalpy so the lattice becomes weaker.
The charges of the ion, the bigger the charge the more negative the latice enthalpy so the lattice becomes stronger
What characteristics would a covalent character lattice have?
Small cations, big anions
What characteristics would a ionic character lattice have?
Large cation, small anions
What does a small difference in lattice enthalpy show?
The structure is more perfectly ionic
What is Entropy?
A numerical measure of disorder within a system
What is the order of the states with increasing entropy
solid, liquid, gas
Why is entropy 0 at 0K?
particles in a solid have no energy so will not vibrate
Why does entropy increase with temperature?
Particles gain energy so move further apart, increasing the state of disorder
Why is entropy change biggest from liquid to gas and not solid to liquid?
Its biggest at the boiling point due to there being a bigger change in disorder as particles fully spread out from a liquid to a gas.
What is the equation for entropy change? And the units.
Entropy of (products - reactants) in J per kelvin per mole
How to calculate enthalpy of solution?
Enthalpy of lattice dissociation + sum of individual enthalpy of hydrations
What is the electrode potential of a half cell?
A measure of how easily the half cell gives up electrons
Will a more negative half cell be reduced or oxidised?
Oxidised
Will a more positive half cell be reduced or oxidised?
Reduced
In electrochemical cells, in what direction do we write out half equations?
In the reduction direction
Why is the standard hydrogen electrode used?
It is used as a reference to measure other electrode potentials as by defenition the SHE has a E value equal to 0.00V
To compare E values, what conditions are required?
298K, 100kPa, 1 moldm^-3 concentration
In the Electrochemical series, describe the best oxidising agent.
It is the substance that in its own half equation gains electrons and its electrode potential being the most positive.
In the electrochemical series, describe the best reducing agent.
It is the substance that in its own half equation loses electrons and its electrode potential being the most negative.
In terms of oxidation and reduction how would you calculate the E cell value?
Electrode potential of (reduction - oxidation)
In cell notation, what electrode goes on the LHS of the double line?
The more negative electrode potential (as it undergoes oxidation)
In cell notation, if the oxidised species and reduced species are in the same state, what is used?
A comma is used to separate them
If 2 non-metals are part of a half cell, what is added at the end of the cell notation?
A platinum electrode (solid)
What sign will a E cell value take if the reaction between two substances is feasible?
Positive
What solution is used to form the salt bridge in an electrochemical cell?
Potassium Nitrate
What is the equation for the oxygen half equation in a hydrogen fuel cell?
2H2O + O2 + 4e- → 4OH-
What is the equation for the hydrogen half equation in a hydrogen fuel cell?
H2 + 2OH- → 2H2O + 2e-
Why is a carbon rod kept in a rechargeable fuel cell?
To allow electrons to flow
Why is a non-reactive porous separator kept within the fuel cell?
To allow ions to pass through as it is semi-permeable
Why is platinum used as a metal for the electrode in a cell?
They are inert, have a high electrical conductivity and are durable so last longer that standard carbon electrodes.
Why is potassium nitrate used as a solution for the salt bridge?
It is inert and allows ions to move between solutions
What is Kw equal to?
[H+] [OH-]
In the dissociation equation there is H2O, why is it not present in the Kw equation?
The concentration of H2O is very high and becomes a constant along with Kc
Kw varies with temperature, what is the value of Kw at 25oC?
1×10^-14
Why can the equation of Kw be rearranged to Kw = [H+]2 ?
In the dissociation of water equation, both ions are produce in equal amounts of 1 mole each