L17 - Gene Therapy I
1/46
Earn XP
Description and Tags
Biotech Exam II
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What is gene therapy?
a technique to treat a disorder by introducing, inactivating, or replacing genes in a patient’s cells instead of using drugs/surgery
Gene augmentation
involves adding a copy of a functional gene → cell with corrected function
loss of function genes
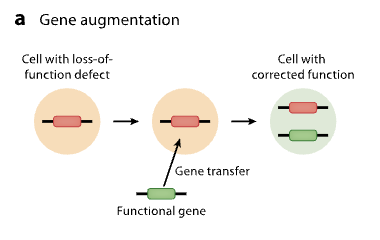
Gene suppression
involves adding an inhibitory sequence to silence a defective gene
gain of function genes
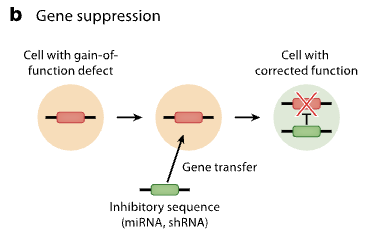
Genome editing
involves replacing a defective gene with a healthy copy
What are the requirements for successful gene therapy protocols?
selecting the right gene (need to understand disease)
identifying and accessing target cells that require treatment
appropriate gene delivery system
proof of principle, safety, and efficacy
suitable manufacturing and analytical processes
Properties of somatic gene therapy:
not inheritable
gene expression only occurs in the target cells
aim to cure disease only in patient, not in descendants
Properties of germline gene therapy
inheritable
genetic mods will be passed to the next generation
Advantages of targeting lymphocytes for somatic cell gene therapy
relatively long lived
readily obtainable from peripheral blood
easy to manipulate for gene transfer
no inactivation of gene expression during differentiation
can be depleted post-transfer
able to secrete large amounts of protein
potentially useful for manipulating immune responses (CAR-T cell therapy)
Ex vivo vs in vivo
ex vivo is done outside of body
in vivo is done inside of body
How does ex vivo gene therapy work?
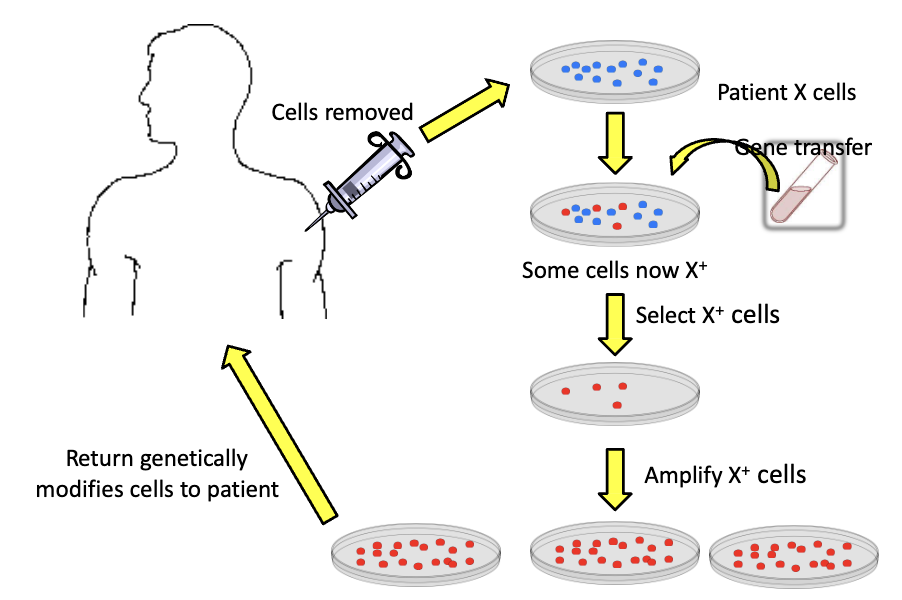
Advantages of ex vivo
does not require tissue specific vectors
very high transfer efficacy
target cells can be manipulated/amplified
Disadvantages of ex vivo
can be used for limited target cells (BCs)
cells need to function normally post transfer
in vitro artifacts
In vivo advantages
can target all body tissue
no in vitro artifacts
In vivo disadvantages
specificity of gene transfer can be an issue
less invasive
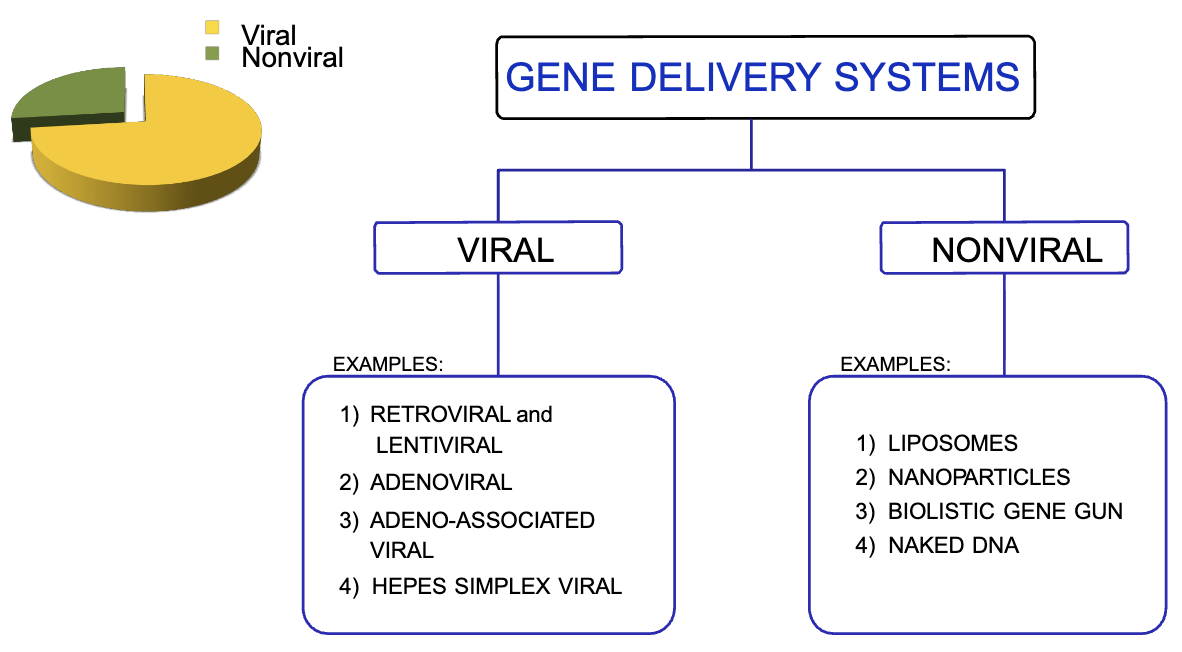
What are the two types of gene delivery systems?
viral (use of viral vectors)
nonviral (majority)
What are retro viruses?
single stranded positive sense RNA viruses
inserts a copy of its genome into the DNA of a host cell that it invades, thus changing the genome of that cell
infection persists indefinitely
What are the components of a retrovirus
gag gene - viral core structural proteins
pol gene - viral enzymes for infection
env genes - viral envelope for recognizing host cell receptors
2 copies of RNA genome
What is the retrovirus life cycle?
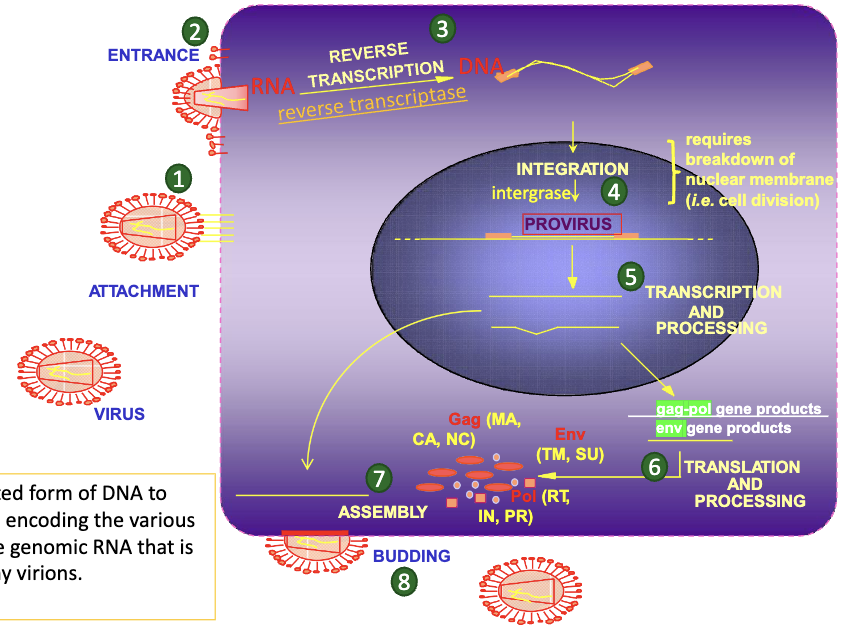
What is a provirus?
the integrated form of DNA to produce both mRNAs encoding the various viral proteins
What are the two categories of the retroviral genome?
Cis sequences - directly active as nucleic acids
trans sequences - the protein coding sequences (gag, pol, env)
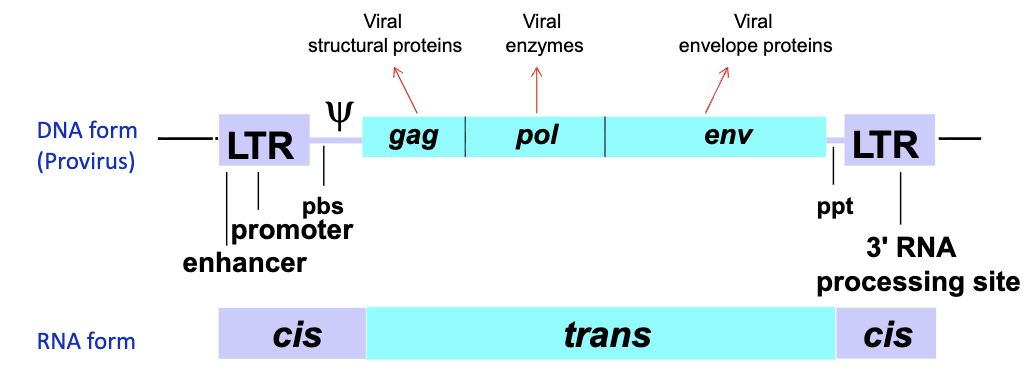
What are the different components of the cis sequence?
5’ LTR - acts as the promoter in DNA form and contains seqs important for reverse transcription in RNA form
PBS - first strand DNA synthesis during reverse transcription
psi sequence - directs packaging of genomic RNA into virion
ppt - primer binding site for second strand DNA synthesis
3’ LTR - in DNA acts as polyadenylation signal, in RNA important for reverse transcription
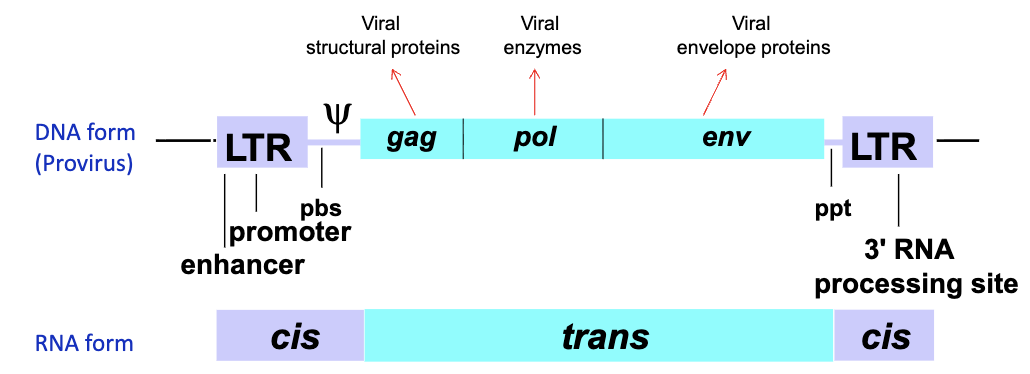
What would happen if the psi region is deleted from the viral genome?
you would have empty virus particles with no viral RNA (aka helper virus)
What would happen if you replace the trans sequence with therapeutic genes?
The resulting virus particle will contain the therapeutic gene (aka vector virus)
What could happen if you combined the helper and vector virus?
regeneration of the replication competent virus (wild type virus)
What is the main limitation of retrovirus in which all the trans sequences are replaced by the gene of choice?
they can express only one gene
What are some ways we can express multiple genes from one viral vector?
expression of different proteins from alternatively spliced messenger RNAs trabscribed from one promoter
use of IRES elements to allow translation of multiple coding regions from a single mRNA
use of the promoter in the LTR and internal promoters to drive transcription of different cDNAs
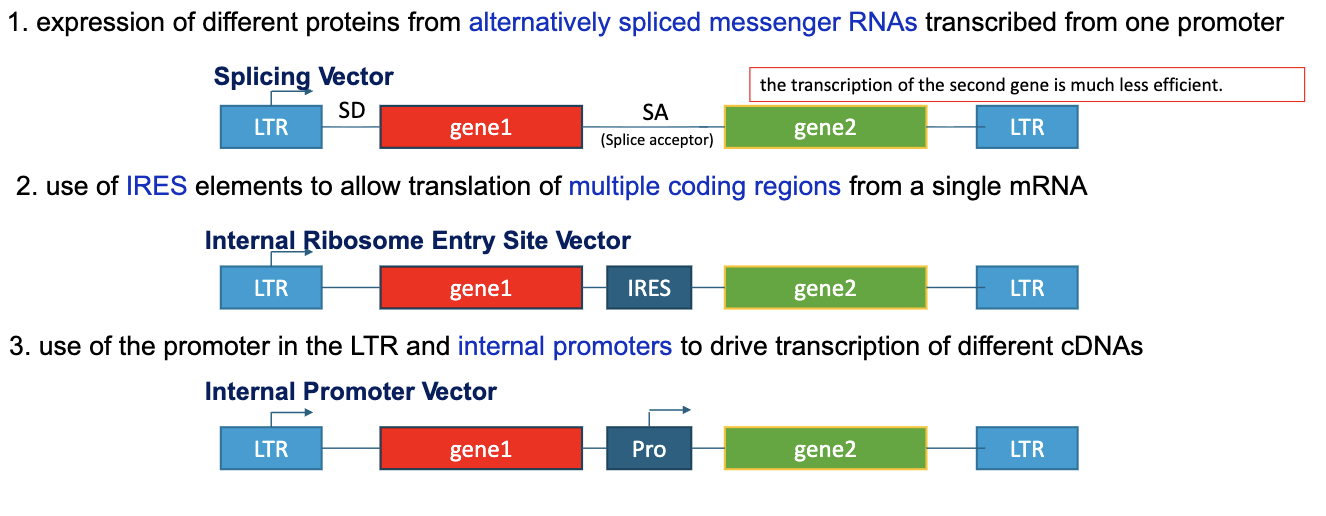
Typically what is gene 1 and gene 2 in viral vectors?
gene 1 is the desired gene and gene 2 is an antibiotic resistance gene, which serves as a marker gene for molecular detection of positive target cells in the host
What are pseudotyped viruses?
the envelope proteins consist of the parts of viral protein necessary for incorporation into the virion and the seqs meant to interact with specific host cell proteins
What are advantages of using retroviral vectors?
efficient and stable integration
controllable host range via envelop pseudotyping
capable of delivering up to 8 kbp of exogenous sequences
What are disadvantages of using retroviral vectors?
can infect only dividing cells
difficult to obtain high concentrations
8 kbp may not be enough
What are some safety concerns for using retroviral vectors?
production of replication-competent virus
insertional mutagenesis (activation of protooncogene)
What are lentiviral vectors?
retroviruses with the ability to infect both dividing and non-dividing cells
integrate permanently into host genome
What are some alternatives to HIV-1 based vectors?
HIV 2 based vector - lower transmissibility
SIV
FIV,EIAV - non-primate based vectors
What is the number 1 vector used in gene transfer in clinical trials?
adenovirus (non-enveloped double-stranded DNA virus)
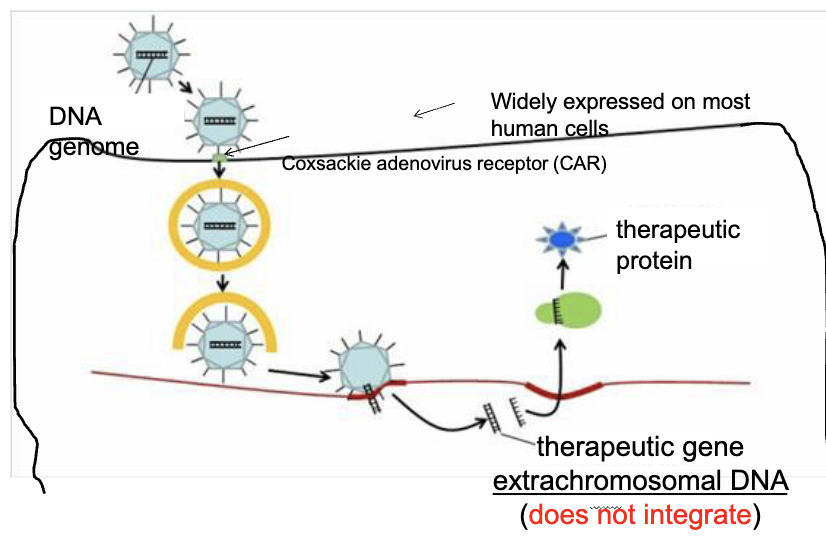
What is the adenoviral vector delivery system
bind to receptors and enter cell via endocytosis
travels to nucleus
DNA enters the nucleus, but does NOT integrates into genome
gene is transcribed and protein is produced
What is the structure of adenoviral vectors?
early genes (E1-4) - expresses non structural, regulatory proteins
late genes (L1-5) - viral structural protein required for viral genome packaging and assembly
E1 and E3 are replaced with the therapeutic gene
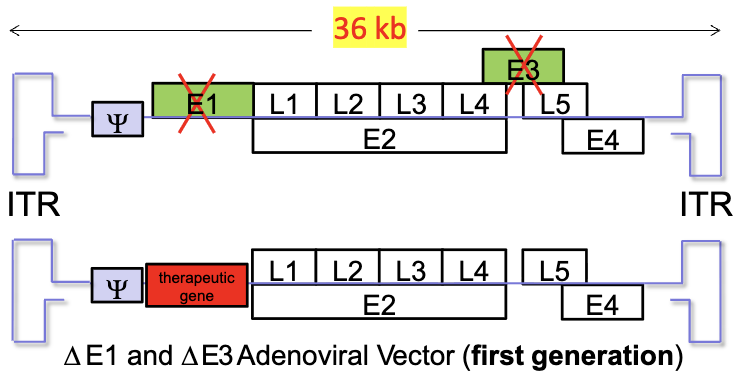
What are limitations of first generation adenoviral vectors?
pre-existing immunity
leaky expression of adenoviral proteins from genes that were not deleted
can result in destruction of cells expressing viral proteins
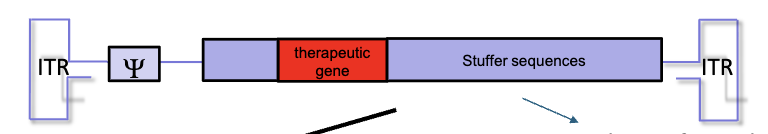
What are gutted adenoviral vectors?
have no viral protein genes present
contains the therapeutic gene and stuffer sequences to maintain right size for packaging
high capacity, can transduce multiple genes
extended time of gene expression
reduced immunogenicity
What are the main advantages of adenovirus vectors?
large capacity of transgene
high titers
ability to infect a broad range of cells (dividing and non dividing)
no evidence for chromosomal integration
stable and does not undergo rearrangement at a high rate
low pathogenicity
What are the main disadvantages of adenovirus vectors?
very immunogenic
does not integrate into host’s genome
transient expression
What are adeno-associated viruses (AAV)?
non-enveloped single stranded DNA virus
requires a helper virus to produce infectious particles
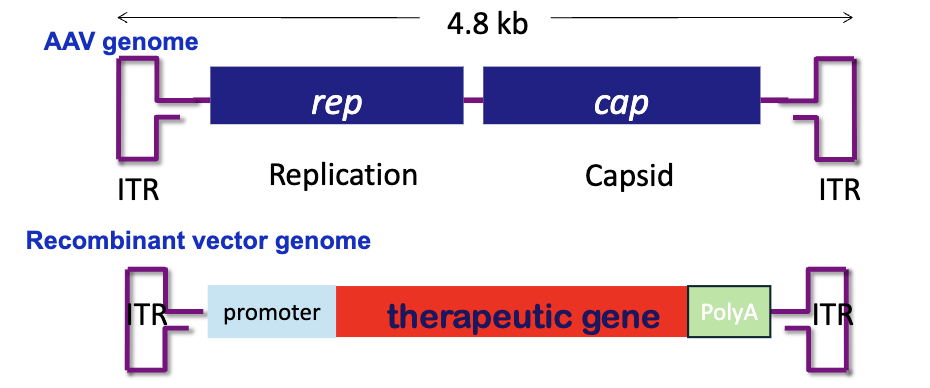
What is the main problem with conventional AAV?
slow and inefficient because the host cell must first convert the ssDNA into dsDNA for transcription to begin
What are self-complementary AAV (scAAV) vectors?
modified vector that contains two complementary copies of the transgene arranged in an inverted orientation
linked by mutated inverted terminal repeat (ITR) that allows for self-complementary folding
has a reduced cargo capacity
What are the main advantages of AAV vectors?
small, easy to manipulate
infect both non dividing and dividing cells
low immunogenicity
not associated with any known human disease
What are the disadvantages of AAV vectors?
limited packaging capacity
requires adenovirus as helper virus
What is herpes simplex type I virus (HSV)?
dsDNA virus
tissue specific gene transduction
treats CNS diseases
can be selectively depleted by treating with ganciclovir
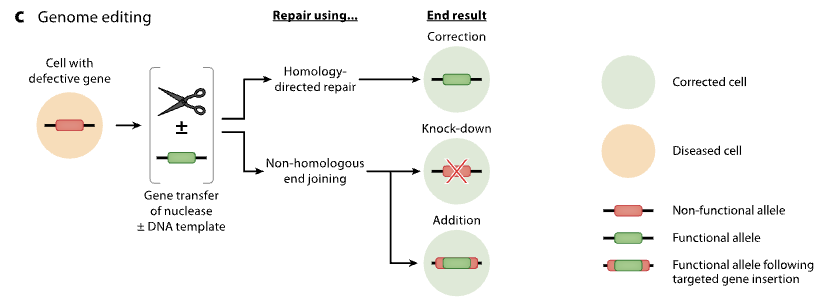
Comparation of different viral vectors