Writing Chemical Equations
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
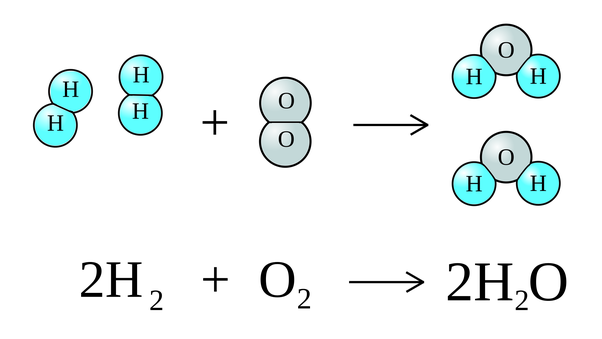
Numbers written before a molecular formula mean the number of ————— of that substance
molecules
e.g.
2NaCl means 2 molecules of sodium chloride
Numbers written as a lowercase after an atomic symbol mean the number of ———— of that element within a single molecule of that substance
atoms
e.g.
H2O means that in a single molecule of water, there are 2 hydrogen atoms and a single oxygen atom
2H2O means ….
there are two water molecules, each with 2 hydrogen atoms and a single oxygen atom for a total of 4 hydrogens and 2 oxygens
what is a word equation
Provides a simple way to describe chemical reactions by stating the reactants and products.
e.g
Solid sodium Metal reacts with Oxygen in the atmosphere to form sodium oxide solid.
what is a chemical equation
Chemical equations describe a chemical reaction, with the reactants to the left of the arrow and products to the right.
Formulae are used to represent the chemicals involved.
The physical states of the chemicals are often included.
Numbers are written in front of the formulae in order to balance the numbers of atoms on each side of the equation.
e.g.
2Na(s) + O 2(g) → 2Na 2O(s)
what are the reactants
the substances that enter a chemical reaction
what are products
are formed as the result of reactants reaction