cell bio and neuroscience exam #3
1/80
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
Pseudounipolar neurons
Sensory neurons— One process extending from cell body, sensory receptors at one end and dendrites at the other
Afferent fibers
Send sensory info from skin into CNS
Dorsal root ganglia
Cell bodies outside CNS
Sensory transduction
Converting the energy of a stimulus into an electrical signal
Stimulus alters the permeability of ion channels in aferente nerve endings
Mechanoreceptors
Specialized receptor cells that encapsulate afferent fibers; specialized to detect touch, pressure, vibration
Transduction in a mechanosensory afferent
Stimulus causes ion channels to open, resulting in an action potential if threshold is reached
Dermatomes
Innervation arising from a single dorsal root ganglia and its spinal nerve
Axon diameter in afferent fibers
largest: supply sensory receptors in muscles
Slightly smaller: touch
Smaller: pain and temperature
Afferent fibers receptive field
the area of skin surface over which the stimulus results in a significant change in the rate of action potentials
Size of afferent receptive fields
dense innervation (lots of afferent fibers) have small receptive fields —> smaller 2-point discrimination threshold
Less densely innervated areas have larger receptive fields
Two-point discrimination
The minimum interstimulus distance required to perceive two simultaneously applied stimuli as distinct
Dense innervation = more fibers = smaller 2pt discrimination
Location of stimuli in receptive field
stimuli at edge of receptive fields spike at slower rate
Central areas = faster rate
Rapidly adapting afferent fibers
Detect changes in ongoing stimulation
Fire rapidly when stimulus introduced, then falls silent after continuous stimulation
Slowly adapting afferents
Detect spatial attributes, size & shape
Generate a sustained discharge in the presence of an ongoing stimulus
Types of mechanoreceptors in the skin
Merkle cell afferents, meissner afferents, pacinian afferents, ruffini afferents
Merkle cell afferents
epidermis (sweat ridges)
Info about Form and texture (edges, points, curvature)
Meissner afferents
dermis (but superficial)
Info about texture object movement across skin (slippage, grip control)
Rapidly adapting
Pacinian afferents
deep in the dermis
Rapidly adapting, very sensitive
Detect vibrations from objects being gripped, important for skilled tool use
Ruffini afferents
Dermis
Slow adapting
Responsive to internally generated stimuli, involved w muscle receptors for representing finger position (skin stretch)
Mechanoreceptors for proprioception
Muscle spindles, golgi tendon organs, joint receptors
Muscle spindles
In skeletal muscles, signal changes in muscle length
Golgi tendon organs
In tendons to inform CNS about changes in muscle tension
Joint receptors
In joints, appear to be importance for judging the position of the fingers. Little contribution to limb proprioception
Dorsal column-medial lemniscal pathway
mechanosensory info ascends spinal cord and crosses over at caudal medulla before reaching brain
Synapses into ventral posterior lateral nucleus of the thalamus, then to primary somatic sensory cortex
Hippocampus
Receives sensory info for learning and memory
Mechanosensory receptors in the face
Trigeminal ganglion
Info enters through cranial nerve, not spinal cord, goes to thalamus
Proprioceptive pathways for the upper and lower body
From muscle spindle afferents to spinal cord, to ipsilateral (same side) cerebellum
dorsal spinocerebellar tract
proprioceptive pathway for upper body, info ascends spinal cord to ipsilateral cerebellum
Cerebellum
Regulates the timing of muscle contractions necessary for voluntary movement. Motor learning
Somatic sensory components of the thalamus
Ventral posterior complex receives info from body and posterior head (VPL) and face (VCM)
Projects directly onto cortical neurons in the primary somatosensory cortex (SI)
Primary somatosensory cortex
Broadmann’s areas: 3a, 3b, 1, and 2
3b is first step in cortical processing, heavy projections onto areas 1 & 2
Functional changes in somatic sensory cortex following amputation of a digit
Neural plasticity of somatic sensory cortex following allows remaining digits brain regions to adapt
Functional expansion of a cortical representation by repetitive behavioral task
(Monkey experiments) After differential stimulation, a larger region of the cortex contained neurons activated by the digits used in the task
Phantom limb syndrome
When an amputee has sensation in their amputated limb
Representation of the face is right next to the somatosensory cortex allows neuroplasticity: the area of the brain corresponding with the face took over the region associated with the amputated hand
Can be treated with mirror box therapy, visual feedback
Blindsight
One cannot consciously perceive an object in one side of visual field, but can answer questions about it
Two pathways link eyes to visual cortex, newer one damaged while primitive one intact
nociceptors
unspecialized nerve endings that initiate pain sensation
axons are lightly myelinated or unmyelinated, relatively slow conduction compared to mechanosensory afferents
thermoreceptors vs nociceptors
at a certain temperature, nociceptor reaches threshold and fires meanwhile the thermoreceptor plateaus in response magnitude
first and second pain
first pain is sharp, initial sensation. second pain is delayed, longer lasting.
Type A𝛿 fibers/nociceptors
fibers are myelinated
nociceptors specialized for heat and mechanical stimuli
Type C fibers/nociceptors
fibers are unmyelinated, slower conduction
nociceptors respond to all types of pain: thermal, mechanical, and chemical (polymodal)
Capsaicin
‘hot’ taste
binds to TRPV1 vallinoid receptor channel
TRP (transient receptor potential)
TRPV2, TRPA1— chemical irritants (tear gas, exhaust, cigarettes)
ASIC3— muscle/cardiac pain related to pH changes from ischemia (lack of O2)
capsaicin as a topical analgesic
Repeated application desensitizes pain fibers, prevents neuromodulators from being released by nerve terminals
wide-dynamic range neurons
receive all types of input
also receive visceral (internal) sensory input
likely involved in Referred pain
Referred pain
feeling pain away from its point of origin (i.e. heart attack in jaw, arm)
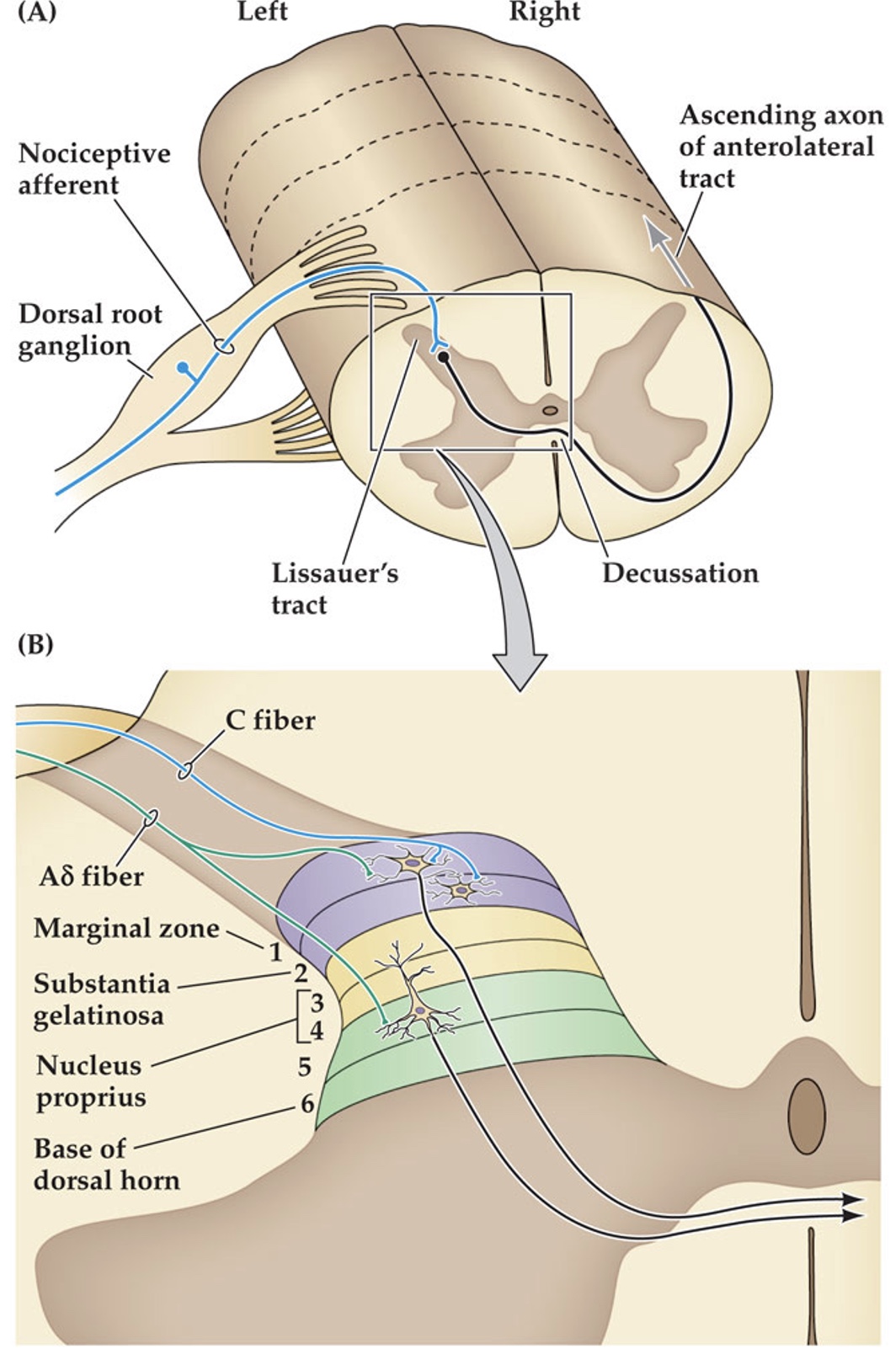
dorsal horn, dorsal root ganglia
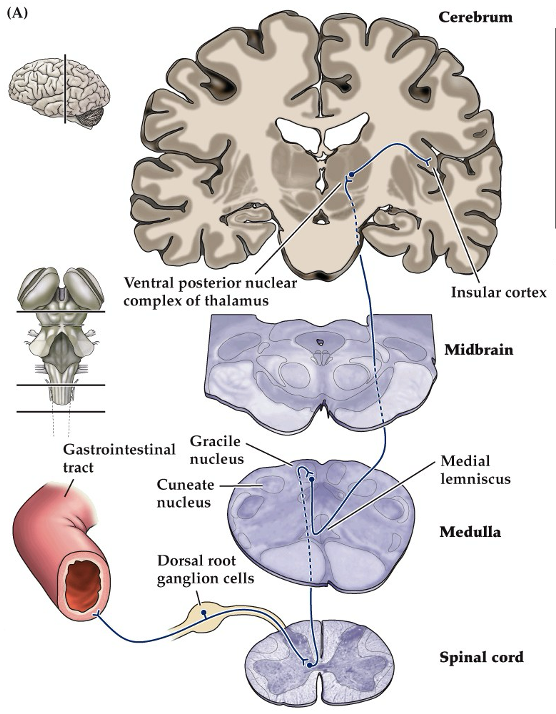
a visceral pain pathway in the dorsal column-medial lemniscal system. only pathways involving visceral (internal) pain from the pelvis & lower abdomen shown
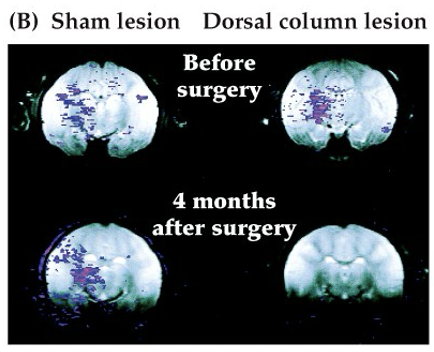
proves existence of visceral pain pathway: activity in thalamus follows noxious stimuli. after a dorsal column lesion, this activity is abolished (but not by a sham lesion)
midline myelotomy
lesion part of the dorsal column pathway to alleviate visceral pain, inserting needle into the spinal cord
anterolateral system
pain pathway
pain and temperature info crosses over immediately in the spinal cord
ascends all the way to cerebrum
pain and temperature info from the face
descends before ascending in order to cross over at caudal and middle medulla, then reaches cerebrum
lesion in the left lower thoracic spinal cord
will impair nociceptive afferents from the right, and mechanosensory afferents from the left
will not impair arms/upper body because info enters above the lesion
lesion in the right lower thoracic spinal cord
will impair nociceptive afferents from the left, and mechanosensory afferents from the right
will not impair arms/upper body
location of lesion in spinal cord impacts
If lesion is moved higher up, we would see deficits higher up in the body (arms, upper body)
distinct aspects of pain
anterolateral system has sensory-discriminative processing and affective-motivational processing
sensory-discriminative
location, intensity, quality of noxious stimuli. Spinothalamic tract to somatosensory cortex (S1, S2)
affective-motivational
unpleasant feeling, fear, anxiety, autonomic activation
hypothalamus and endocrine system generates stress response (cortisol)
amygdala generates fear, anxiety
heightened awareness, alertness
Pain Matrix
brain areas associated with the experience of pain; somatosensory cortex, amygdala, insular cortex, anterior cingulate cortex
pathway that transmits visceral pain info to the brain
dorsal column-medial lemniscal pathway (same as mechanosensory info)
observations that support the discovery of dorsal column visceral sensory projection
neural response to noxious stimuli
neural responses reduced by lesions to dorsal column
infusion of drugs that block nociceptive synaptic transmission blocks neural response to nociceptive information, but not cutaneous
hyperalgesia
following a painful stimulus, subsequent stimuli are perceived as more painful
peripheral sensitization
results from interaction of nociceptors with inflammatory molecules, causing hyperalgesia
inflammatory response to tissue damage
nociceptors release peptides and neurotransmitters (Substance P, CGRP, ATP)
non-neuronal like mast cells, platelets, macrophages cells release other molecules (cytokines, histamine, prostaglandins)
released molecules interact with receptors on nociceptive fibers
allodynia
stimuli that are normally innocuous become painful, occurs immediately after a painful event. can be induced by repeated presentation of a static stimulus feeling increasingly painful
central sensitization
due to immediate onset, activity dependent increase in excitability of neurons in the dorsal horn of the spinal cord
Activity of nociceptive afferents that was subthreshold before become sufficient to generate APs in dorsal horn neurons, leading to an increase in pain sensitivity and allodynia
neuropathic pain
increased pain sensitization can persist when afferent fibers or central pathways are damaged (diabetes, shingles, MS, AIDS). chronic, intensely painful experience that is difficult to treat with conventional analgesics
descending control of pain perception
top-down/psychological influence on the pain matrix, perception of pain depends on its context. signals originate in brain and descend to dorsal horn of spinal cord, where they modulate incoming signals.
placebo effect
physiological response varies depending on context accompanying pharmacologically inert “remedy”
gate theory of pain
synaptic interactions w/ the dorsal horn modulate perception of pain
descending pathways that produce pain relief
stimulation of certain midbrain regions results in pain relief. Periaqueductal grey matter (PAG), dorsal raphe, and locus coeruleus
opioid system
opioids are generally inhibitory. exogenous opioids are powerful analgesics, brain regions particularly affected by opioid drugs are sources of descending projections like PAG; dorsal horn also sensitive
endogenous opioid receptors
Mu, Delta, Kappa
endogenous opioid peptides
Enkephalins, endorphins, and dynorphins (agonists to Mu, delta, and kappa)
endocannabinoids
also suppress nociceptive neurons
effect of opioids at the synapse
closing of Ca2+ channels in the presynaptic neuron, decreasing neurotransmitter release
opening of K+ channels, causing efflux and hyperpolarization
opiate effects on respiration
opiate agonists cause respiratory depression
addiction to opiates
euphoria from GABA inhibitory interneurons in the ventral tegmental area. opioids bind to mu receptors, suppressing GABA and increasing dopamine
Desensitization/down-regulation of receptors
results from regular opioid use
withdrawal
symptoms are opposite to physical effects
naloxone
preferentially binds to opioid receptors, stronger affinity
enkephalin neurons in the spinal cord
local circuit interneurons release inhibitory enkephalin (opioid peptide), which projects to dorsal horn and decreases pain perception