3.2.5.4 formation of coloured ions
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
what are transition metals widely recognised in chemistry for?
for their vast colour-based properties
the same element can have different colours based upon what?
based upon what ligands are bonded around it
colour changes arise from a change in what 3 things?
oxidation state
coordination number
ligand
why do colour changes arise in transition metals?
due to their partly filled d sublevels
what can the electrons sitting in the d sub level do?
they can shift up + down between the unoccupied orbitals - this is called electron promotion
explain in detail why transition metals have different colours? ie the process behind it
the d orbitals split in energy levels
d electrons are promoted to higher energy d orbitals by absorbing energy in the form of light
the colour transmitted + observed is the light not absorbed
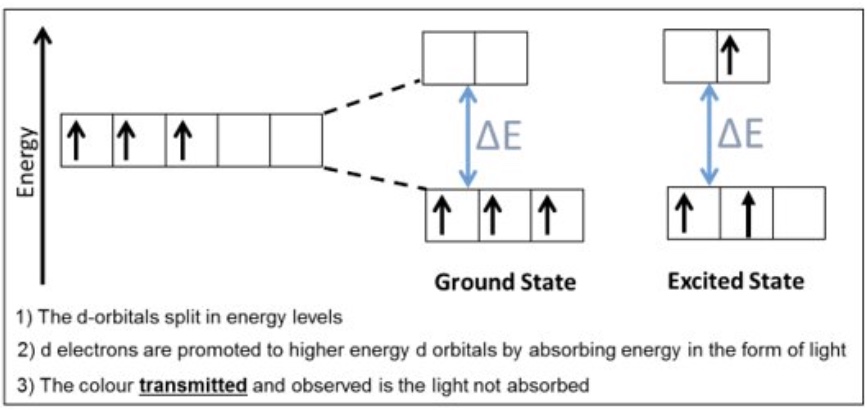
the d orbitals are at specific energy levels, this means what?
that the gap between them has a very specific energy gap (△E) value
therefore what must an electron be given to be promoted from the ground state to the excited state?
at least the △E value
what needs to happen to promote electrons?
the transition metal must absorb a specific frequency of light
give the equation(s) that link the frequency of light to the △E value
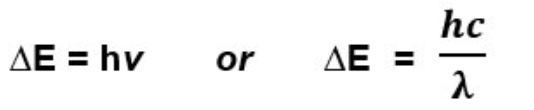
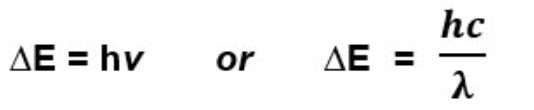
give the units for these equations

if the wavelength is given in nm, how do you convert to metres?
by x 10⁻⁹
explain how electron promotion relates to colour chemistry

a change in colour is due to what?
to the size of △E (energy gap) between the d orbitals changing
what happens if △E changes?
different frequencies of light will be absorbed + different frequencies of light will be transmitted (and hence observed) through the sample
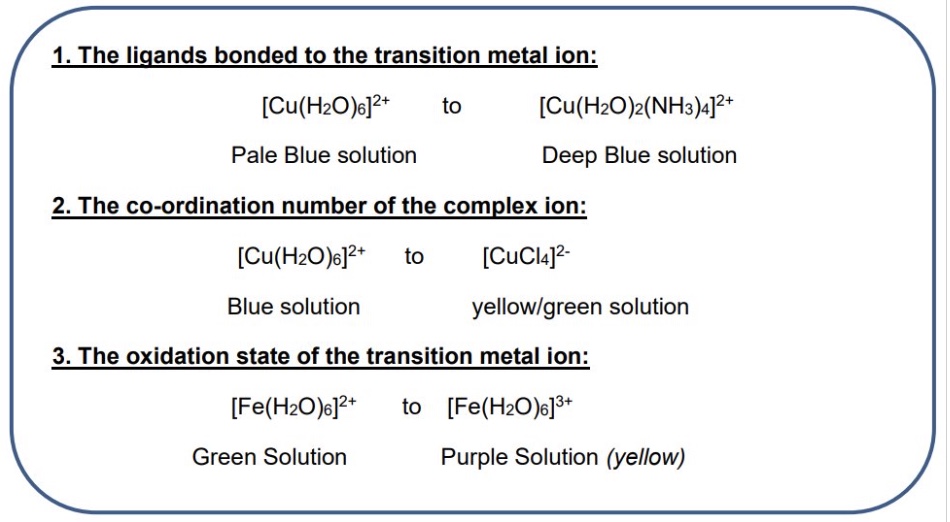
outline the 3 changes that can be made to transition metals to change the △E + so the colour of the complex - include examples
what is a spectrophotometer or colorimeter used to do?
to measure the absorbance of light in the visible (and ultra-violet) region and so the concentration of a complex ion
outline how a spectrophotometer/colorimeter works
it contains a coloured filter
the colour of the filter is chosen to allow wavelengths of light through that would be most strongly absorbed by the colour solution
light of increasing frequency is passed through a sample of coloured complex ion, some of the light is absorbed
what is the wavelength of a colorimeter?

what is the amount of light absorbed proportional to?
to the concentration of the complex ion
some complexes have only pale colours, how could the colour be intensified?
by adding a suitable ligand to intensify the colour
describe the method to determine the concentration of a transition metal ion

example of calibration curve
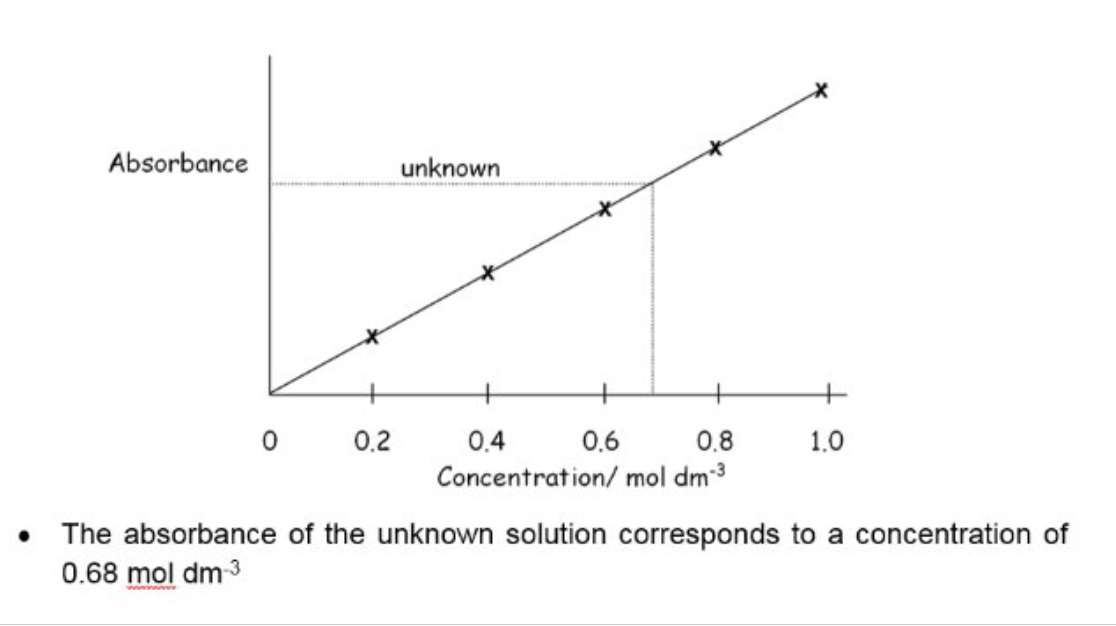
why is the important for each vial to be the same size?
because the amount of light absorbed will also be proportional to the distance travelled through solution

do question b
