Chemistry - Topic 17 - Organic II
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
59 Terms
what are optical isomers?
optical isomers are non-superimposable mirror images of each other
what is optical activity?
optical activity is the ability of a single optical isomer to rotate the plane of polarisation of plane-polarised monochromatic light in molecules containing a single chiral centre
why is there no optical activity observed from a racemic mixture of optical isomers?
optical isomers rotate the plane of plane-polarised light in opposite directions (therefore cancelling out rotational effects)
does a primary halogenoalkane go via SN1 or SN2? If reactant is chiral, what is the product?
SN2
if we start with a single enantiomer, the product is a single enantiomer but with the stereochemistry inverted
does a tertiary halogenoalkane go via SN1 or SN2? If reactant is chiral, what is the product?
SN1
if we start with a single enantiomer of a tertiary halogenoalkane, the product is a racemic mixture. this implies two different mechanisms are taking place
why is stereochemistry inverted during SN2?
two species involved in the slowest step
the nucleophile must attack from 180 degrees away from the leaving group in an SN2 mechanism. this leads to inversion of stereochemistry.
why is a racemic mixture formed via an SN1 mechanism?
trigonal planar around the central carbon atom
the nucleophile can attack from above or below the plane of the carbocation with 50:50 probability leading to a racemic mixture
why do tertiary halogenoalkanes undergo SN1 mechanisms?
tertiary carbocations are more stable than secondary, which are more stable than primary
therefore the C-Hal bond in a tertiary halogenoalkane is the weakest and most likely to break to form a carbocation. tertiary halogenoalkanes are also unlikely to do an SN2 mechanism because the delta positive carbon is crowded with alkyl groups so it is sterically difficult for the nucleophile to attack
what reducing agent and conditions can be used to reduce a carbonyl (aldehyde or ketone). what type of mechanism and reaction is this? what is formed?
dry lithium aluminium hydride (LiAlH₄), aka lithal or dry sodium borohydride (NaBH4)
in dry ether
followed by dilute HCl(aq) to protonate the intermediate formed (acidic work up)
if aldehyde or ketone is reacting then this is a nucleophilic addition (and a reduction), where the hydride ion, H- acts as the nucleophile
if a carboxylic acid is reacting then this is just a reduction
an alcohol is formed
how to test for a carbonyl?
2,4 - DNPH
positive test → red/orange/yellow crystals (it is a spectrum) - often yellow ppt on PPQs
this is a qualitative test
specific to PPQs:
add 2,4-DNPH and collect precipitate by filtration
recrystallise
measure melting point and compare with a data base
how do you identify a carbonyl compound?
take melting point of crystals and compare with data base values to identify the aldehyde/ketone
what type of mechanism and reaction occurs when a carbonyl reacts with HCN (a weak acid) in the presence of a KCN homogenous catalyst? what type of product is formed?
nucleophilic addition
HCN and KCN is a buffer
a hydroxynitrile is formed
why can nucleophilic addition lead to a racemic mixture as a product?
the carbonyl is trigonal planar about the C=O. therefore, the cyanide ion nucleophile can attack from above or below the plane with 50:50 probability
why will nucleophilic addition involving a carbonyl and HCN in the presence of KCN not occur at a low pH?
only HCN
(K+)
no cyanide ion as it has been protonated therefore no nucleophile
at low pH there are not enough deprotonated cyanide ions to act as a nucleophile in the first step/[CN-] is too low therefore rate would be too slow
why will nucleophilic addition involving a carbonyl and HCN in the presence of KCN not occur at a high pH?
at a high pH, there is not enough HCN/H+ to protonate the intermediate in the second step
too low [H+]
how to test for a methyl next to a carbonyl? give an example equation for testing for a methyl group in propanone with state symbols.
reactants: iodine and aqueous sodium hydroxide (alkali)
positive test → yellow precipitate (CHI₃ - triiodomethane, also known as iodoform) + antiseptic smell
also produces a salt of carboxylic acid
CH3COCH3(aq) + 3I2(aq) + 4NaOH(aq) → CH3COO(-)Na(+)(aq) + CHI3(s) + 3Na(+)I(-)(aq) + 3H2O(l)
what determines whether a hydroxide ion is a base or a nucleophile?
base → forms bond with H⁺
nucleophile → forms bond with delta positive carbon atom
state two methods in which you can increase the length of a carbon chain?
reacting a carbonyl with HCN in the presence of KCN (not efficient as can only add one carbon at a time)
using magnesium and dry ether to form a Grignard reagent (compound containing MgBr in dry ether), then react aldehyde with Grignard followed by an acidic workup (source of H⁺ ions)
reacting a compound containing a Br with magnesium makes delta positive carbon atom become delta negative, so that MgBr can break off and the carboanion can attack delta positive carbon atom of carbonyl
how to create a carboxylic acid from a Grignard? what type of mechanism and reaction is this? give an example balanced equation starting with C6H5MgBr (phenyl magnesium bromide).
react carbon dioxide (dry ice) with a Grignard
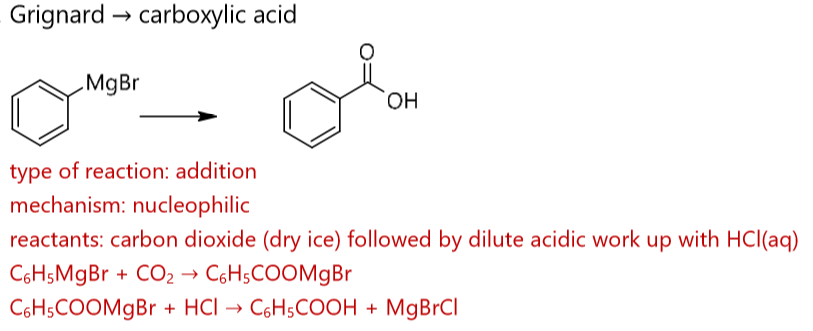
name eight ways to prepare a carboxylic acid.
complete oxidation of a primary alcohol
oxidation of an aldehyde
acid hydrolysis of a nitrile
alkaline hydrolysis of a nitrile followed by an acidic work up
reacting carbon dioxide with a Grignard reagent
reacting an acyl chloride with water
acid hydrolysis of an ester
acid-base reaction with a carboxylate salt
how do you form a carboxylic acid from a nitrile? state conditions and an example balanced equation starting with ethane nitrile.
acid hydrolysis
heat/H⁺(aq) - heat under reflux with dilute HCl(aq)
CH₃CN + 2H2O + HCl → CH₃COOH + NH₄Cl
OR
alkaline hydrolysis
heat under reflux
reactants: NaOH(aq) followed by dilute acidic work up with HCl(aq)
CH₃CN + H2O + NaOH → CH₃COO⁻Na⁺ + NH₃
CH₃COO⁻Na⁺ + HCl → CH₃COOH + NaCl
how do you convert a nitrile into a carboxylic salt? state conditions and an example word equation and balanced equation starting with ethanenitrile.
hydrolysis of nitrile under alkali conditions - alkaline hydrolysis
heat under reflux with NaOH(aq) (base)
e.g. ethanenitrile + water + sodium hydroxide → sodium ethanoate + ammonia
CH₃CN + H₂O + NaOH → CH₃COO⁻Na⁺ + NH₃
how do you reduce a carboxylic acid and give an example using ethanoic acid (word equation)
dry lithium aluminium hydride (LiAlH₄), aka lithal or dry sodium borohydride (NaBH4)
in dry ether
followed by dilute HCl(aq) to protonate the intermediate formed (acidic work up)
reduces to an alcohol + water
ethanoic acid + 4[H] → ethanol + water
how can you produce an acyl chloride? state conditions and give an example balanced equation starting from ethanoic acid including state symbols + observations. what type of reaction is this?
by reacting a carboxylic acid with anhydrous phosphorous (V) chloride at room temperature (298K) as reaction is very vigorous
CH₃COOH(l) + PCl₅(s) → CH₃COOCl(l) + POCl₃(l) + HCl(g)
observations: effervesence and steamy fumes (HCl)
substitution reaction
how do you form an ester? what type of reaction is the forward reaction? what type of reaction is the reverse reaction? give an example showing the balanced equation and word equation. what should be noted about this reaction?
react a carboxylic acid with an alcohol in the presence of a concentrated sulphuric acid catalyst + heat under reflux
forward reaction - esterification
reverse reaction - ester (acid) hydrolysis
CH₃OH + CH₃COOH ⇌ CH₃COOCH₃ + H₂O
methanol + ethanoic acid ⇌ methyl ethanoate + water
this reaction is reversible and reaches a dynamic equilibrium
what is acylation?
replacing a hydrogen with an acyl group
acyl chloride + water → ?
e.g. ethanoyl chloride + water → ?
what type of reaction is this?
acyl chloride + water → carboxylic acid + HCl
ethanoyl chloride + water → ethanoic acid + HCl
substitution reaction
acyl chloride + alcohol → ?
e.g. ethanoyl chloride + methanol → ?
what type of reaction is this and what are the conditions?
acyl chloride + alcohol → ester + HCl
ethanoyl chloride + methanol → methyl ethanoate + HCl
esterification reaction
this reaction is irreversible and goes to completion
reaction does not require catalyst and occurs at room temperature (as acyl chlorides are highly reactive electrophiles)
acyl chloride + concentrated ammonia → ?
e.g. ethanoyl chloride + concentrated ammonia → ?
acyl chloride + concentrated ammonia → an amide + ammonium chloride
ethanoyl chloride + concentrated ammonia → ethanamide + ammonium chloride
ethanoyl chloride + R-NH₂ (amine) → ?
example equation using
N-Rylethanamide + R-N⁺H₃ + Cl⁻
write a general word equation for the acid hydrolysis of an ester (reverse of esterification). then write a balanced equation for the acid hydrolysis of ethyl ethanoate and state the required conditions and reactants. what must be noted about this reaction?
ester + water → carboxylic acid + alcohol
reactant: H2O
catalyst: concentrated sulphuric acid, H2SO4
heat under reflux
CH3COOCH2CH3 + H2O ⇌ CH3COOH + CH3CH2OH
reaction is reversible and reaches a dynamic equilibrium
write a general word equation for the alkaline hydrolysis of an ester. then write a word equation and balanced equation for the alkaline hydrolysis of methyl ethanoate and state the required conditions and reactants. what must be noted about this reaction?
ester + base (NaOH(aq)) → carboxylate salt + alcohol
methyl ethanoate + sodium hydroxide → sodium ethanoate + methanol
CH3COOCH3 + NaOH → CH3CO(-)Na(+) + CH3OH
reactant: concentrated NaOH(aq)
heat under reflux
reaction is irreversible and goes to completion
how are condensation poylmers formed?
condensation polymers are formed when a water molecule is removed from the species of a reaction (so the reaction produces water therefore condensation reaction)
how is a polyamide formed?
when a dicarboxylic acid reacts with a diamine (also forms water).
how is a polyester formed? what type of reaction is this?
when a dicarboxylic acid reacts with a diol (also forms water)
condensation polymerisation reaction
what do reductions of aldehydes and ketones form?
alcohols
opposite of oxidation
give the steps to show how 2,4-DNPH could be used to distinguish between propanone and propanal.
formation of yellow precipitate
filter (each precipitate/to collect precipitate) then recrystallise products
determination of melting points - measure melting points of derivatives
compare melting points to data base values
what is an acyl group + examples?
examples include acyl chlorides, esters, ketones, aldehydes and amides
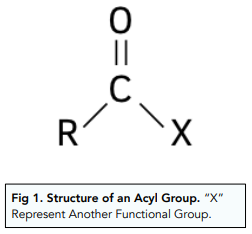
under what conditions do reactions with acyl chlorides take place?
room temperature - 298K
describe how a crystalline solid is separated and purified
filter/decant
dissolve in minimum volume
of hot solvent
cool/leave (to crystallise) AND filter (under reduced pressure)
wash with cold solvent/water AND dry (with method)
draw the transition state (bonds partially broken + partially formed) of 2-bromobutane undergoing a nucleophilic substitution reaction with an OH⁻ nucleophile. draw bond angles and state the type of mechanism this is.
SN2
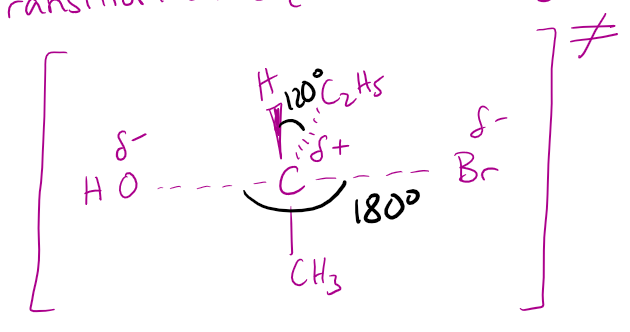
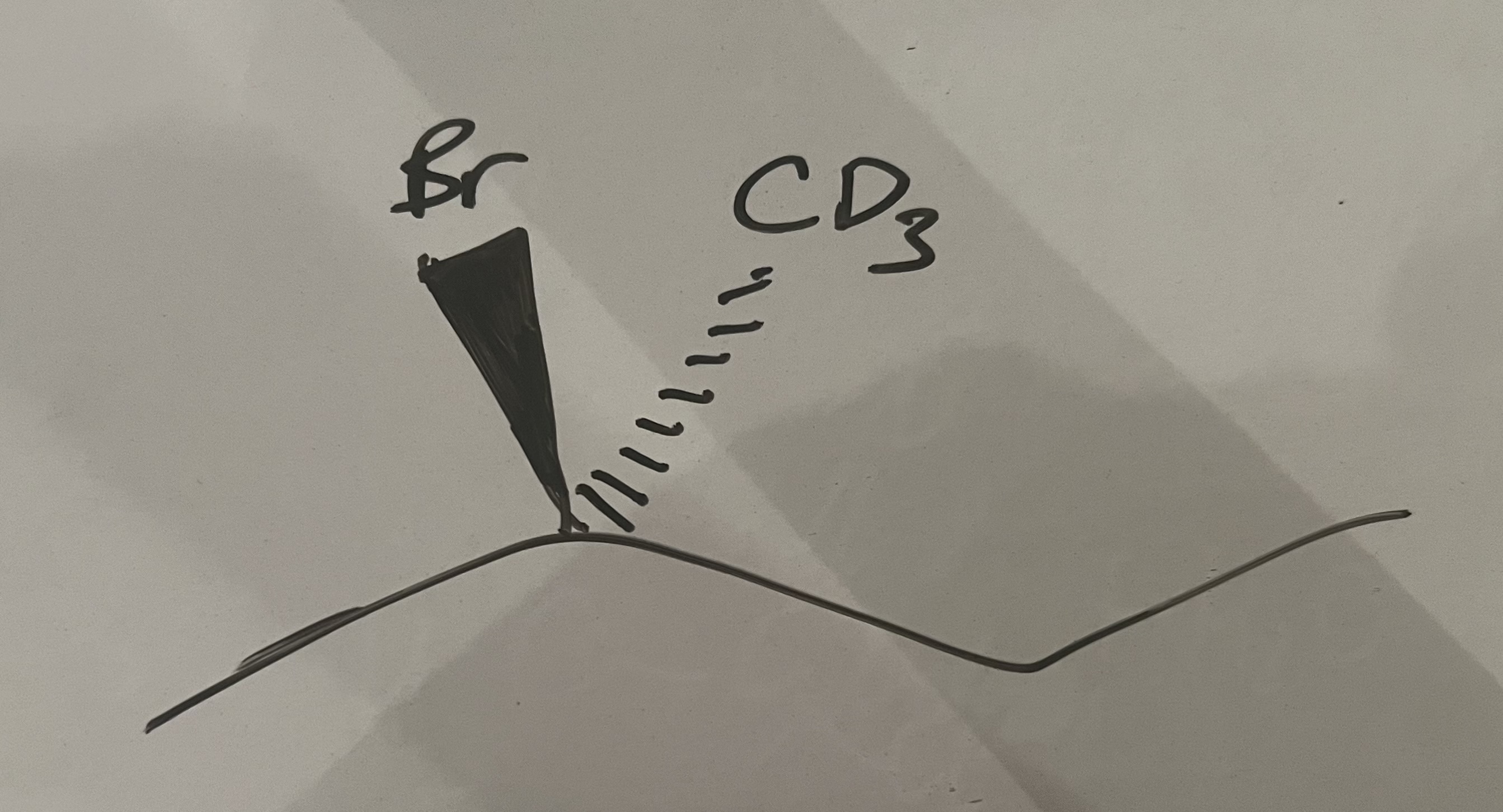
draw the transition state (bonds partially broken + partially formed) of this molecule undergoing a nucleophilic substitution reaction with an OH⁻ nucleophile. state the type of mechanism this is.
SN1
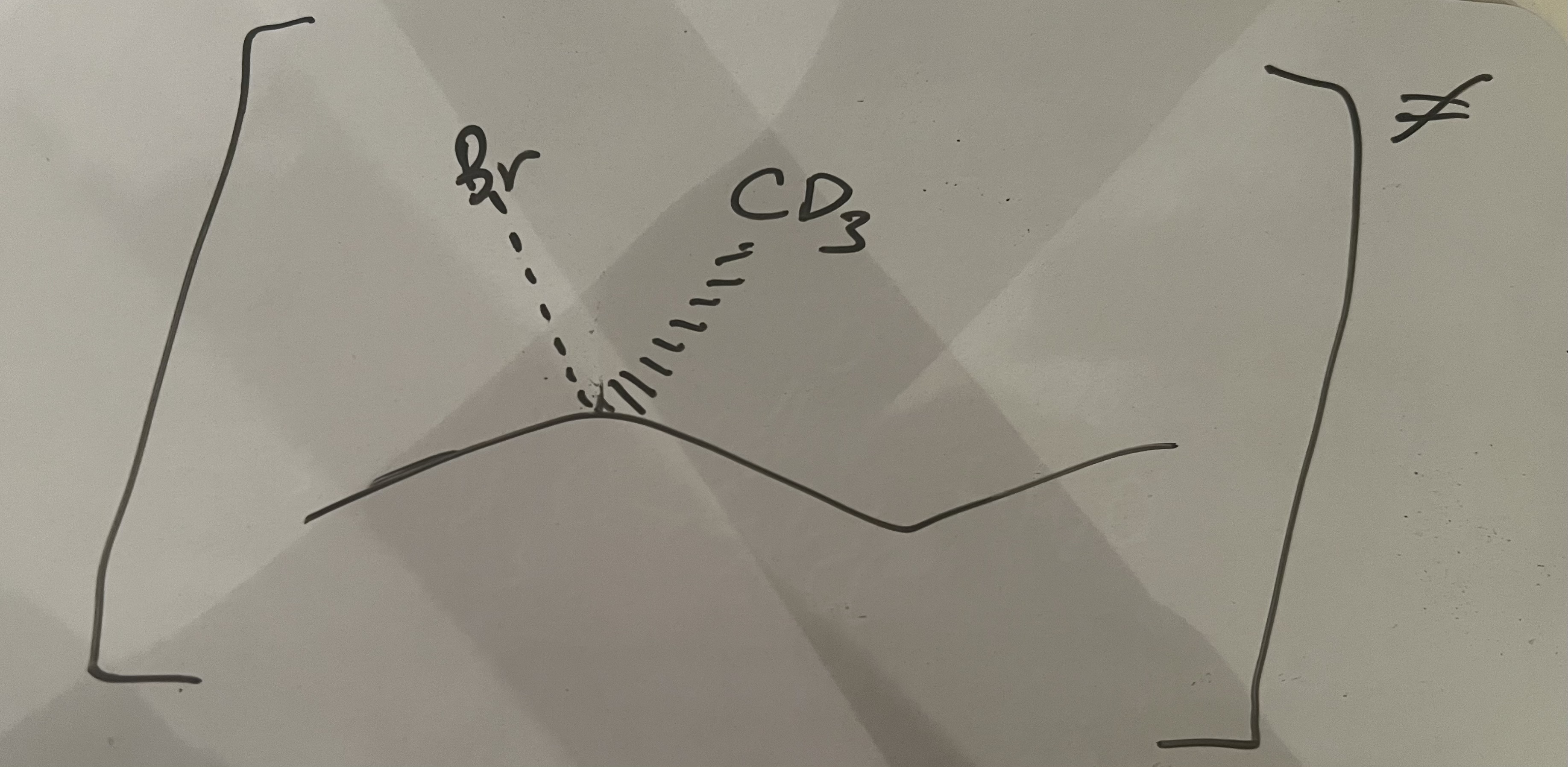
how do you produce a carboxylic acid from an acyl chloride?
react the acyl chloride with water at room temperature
how do you convert a carboxylic acid into a carboxylate salt? what type of reaction is this? give an example balanced equation using ethanoic acid.
reactant: dilute alkali, e.g. NaOH
acid-base reaction
CH3COOH + NaOH → CH3COONa + H2O
OR
reactant: metal, e.g. Mg
redox reaction
2CH3COOH + Mg → (CH3COO)2Mg + H2
how do you convert a carboxylate salt into a carboxylic acid? what type of reaction is this? give an example balanced equation using sodium ethanoate.
reactant: dilute acid e.g. HCl
acid-base reaction
CH3COONa + HCl → CH3COOH + NaCl
how do you convert an alcohol/ketone (containing CH3CH(OH)- or CH3CO- respectively) carboxylic acid (with 1 fewer C atom in chain)
reactants: iodine and aqueous sodium hydroxide (alkali)
positive test → yellow precipitate (CHI₃ - triiodomethane, also known as iodoform) + antiseptic smell
followed by dilute acidic work up to protonate the carboxylate salt to carboxylic acid
how do you convert an acyl chloride into an amide (primary, secondary and tertiary)? name the type of mechanism and reaction, and the reactants required. give example balanced equations starting from ethanoyl chloride to form ethanamide, N-ethylethanamide and N,N-dimethylethanamide
reactants: ammonia or an amine
nucleophilic (acyl) addition-elimination reaction
CH3COCl + 2NH3 → CH3CONH2 + NH4Cl (primary amide)
CH3COCl + 2CH3CH2NH2 → CH3CONHCH2CH3 + CH3CH2NH3Cl (secondary amide)
CH3COCl + 2(CH3)2NH → CH3CON(CH3)2 + (CH3)2NH2Cl (tertiary amide)
what reducing agent and conditions can be used to reduce a carboxylic acid? what type of reaction is this? what is formed?
dry lithium aluminium hydride (LiAlH₄), aka lithal or dry sodium borohydride (NaBH4)
in dry ether
followed by dilute HCl(aq) to protonate the intermediate formed (acidic work up)
if a carboxylic acid is reacting then this is just a reduction
an alcohol is formed
how do you convert an aldehyde/ketone into a hydroxynitrile?
react the aldehyde/ketone with HCN and KCN catalyst
KCN acts as a homogenous catalyst
what are the hazards associated with HCl gas?
HCl gas is toxic and corrosive
how do you convert an ester into a carboxylic acid?
acid hydrolysis
how do you convert an ester into a carboxylate salt?
alkaline hydrolysis
how do you convert a halogenoalkane to a Grignard? what type of reagent is a Grignard? give an example equation starting from 1-bromopropane.
reactant: magnesium metal
conditions: in dry ether
the Grignard reagent is a nucleophile - it donates electrons from the C—Mg σ bond
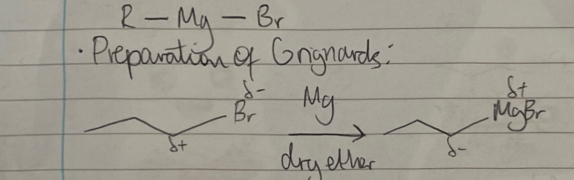
what are the conditions for reactions with Grignards
react with Grignard reagent in dry ether
followed by a dilute acidic work up with HCl(aq)
how do you convert a Grignard into an alcohol (with increased carbon chain length - possible for primary, secondary and tertiary)? what type of mechanism and reaction is this? what are the conditions? give an example balanced equation(s) starting with CH3CH2MgBr being converted to CH3CH2CH2OH.
reactants: carbonyl compound (aldehyde or ketone), e.g. ethanal, followed by dilute acidic work up with HCl(aq)
nucleophilic addition reaction
CH3CH2MgBr + HCHO (methanal) → CH3CH2CH2OMgBr
CH3CH2CH2OMgBr + HCl(aq) → CH3CH2CH2OH + MgBrCla primary alcohol is formed with an extended carbon chain
write a balanced equation for the reaction between ethane-1,2-diol and ethanedioic acid using skeletal formula and then structural formula
n CH2OHCH2OH + n HOOCCOOH → n[OCH2CH2OCOCO] + 2n H2O
![<p><span style="font-family: "Segoe UI", sans-serif">n CH<sub>2</sub>OHCH<sub>2</sub>OH + n HOOCCOOH →</span><span> </span><span style="font-family: "Segoe UI", sans-serif">n[OCH<sub>2</sub>CH<sub>2</sub>OCOCO] + 2n H<sub>2</sub>O</span></p>](https://knowt-user-attachments.s3.amazonaws.com/c5416646-21c6-4004-a2a0-0fd659701f5a.png)
give advantages and disadvantages of using reaction (b), compared to reaction (a) when making methyl butanoate.
(a) reaction of butanoic acid with methanol, using a concentrated sulphuric acid catalyst and heating under reflux
(b) reacting butanoyl chloride with methanol
advantages:
higher yield/not reversible/goes to completion
no heat/no refluxing/less energy needed
no catalyst needed/faster
by-product is a gas (so easier to separate)
disadvantages:
acyl chloride is more expensive/corrosive
toxic HCl gas evolved
has lower atom economy
X is a compound with molecular formula C4H8O2, and you are told X is a neutral organic compound. what does this piece of information suggest about the nature of X?
X is neutral so not a carboxylic acid (neither acidic nor basic)