(P1) Amino Acid Degradation and Urea Cycle
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms








Degradation of amino acids
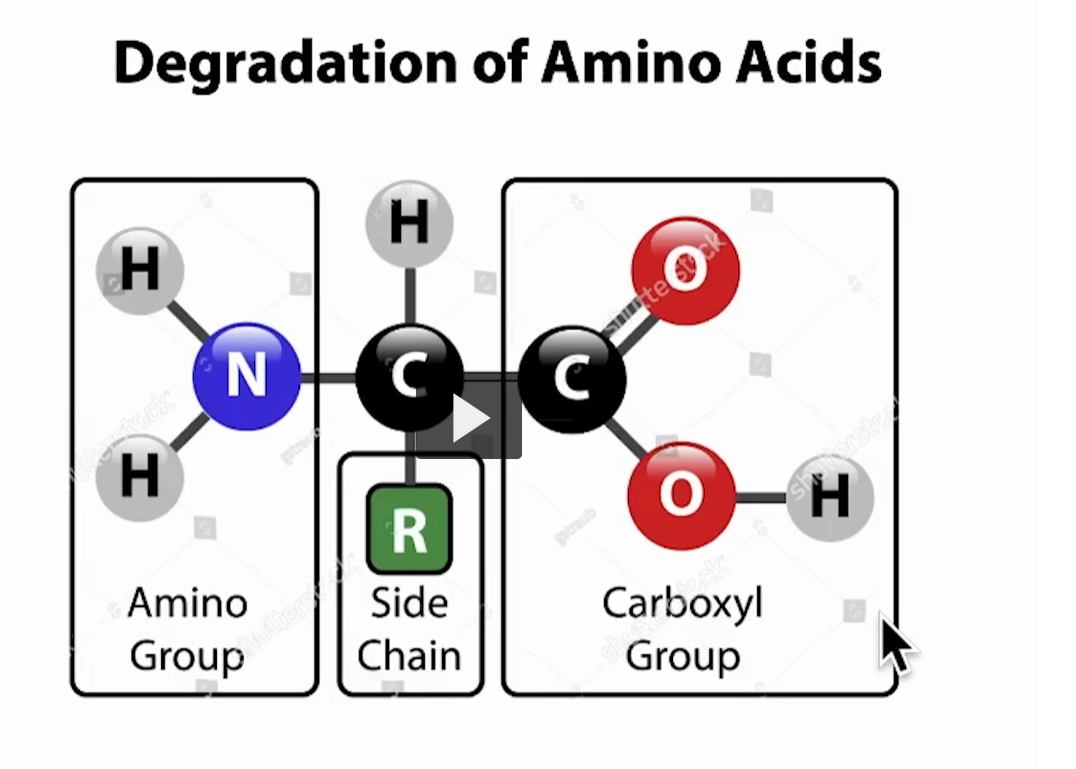
This slide is breaking an amino acid into its three key parts and showing how they are handled during degradation (breakdown).
1. Amino Group (–NH₂) → Nitrogen handling
This part contains nitrogen (N)
During degradation, it is removed (called deamination or transamination)
The nitrogen is toxic if it accumulates → gets converted into urea in the liver → excreted
👉 Key idea:
Amino group = where nitrogen is removed and detoxified
2. Side Chain (R group) → What makes each amino acid unique
The “R” group is different for every amino acid
During degradation, this part determines what the amino acid becomes
It can turn into:
Glucose precursors (glucogenic amino acids)
Ketone bodies (ketogenic amino acids)
Intermediates like pyruvate, acetyl-CoA, or TCA cycle molecules
👉 Key idea:
R group = determines metabolic fate and energy use
3. Carboxyl Group (–COOH) → Carbon backbone
This part, along with the central carbon, becomes part of the carbon skeleton
After nitrogen is removed, what remains is used for:
Energy (ATP production)
Glucose synthesis (gluconeogenesis)
Fat/ketone production
👉 Key idea:
Carboxyl group contributes to the carbon skeleton used for energy
Putting it all together (big picture)
When your body breaks down amino acids:
Remove nitrogen (amino group) → send to urea cycle
Keep carbon skeleton (carboxyl + R) → convert into usable energy molecules
R group decides pathway → glucose vs ketones vs TCA intermediates
Simple way to remember
Amino group = Nitrogen → waste (urea)
R group = Identity → determines pathway
Carboxyl group = Carbon → energy production
Amino Acid Degradation Strategy
• The degradation of amino acids and synthesis of urea may be divided for discussion into four stages:
• Removal of amino groups mostly by transamination
This slide is outlining the overall strategy your body uses to break down amino acids and safely deal with their nitrogen.
Step 1 (shown on your slide): Removal of amino groups
“Removal of amino groups mostly by transamination”
What is happening?
The amino group (–NH₂) is removed from the amino acid
BUT instead of being released directly, it is transferred to another molecule
This process is called: Transamination
How it works (simple):
Amino acid + α-ketoglutarate → new amino acid (glutamate) + new keto acid
So instead of free ammonia (toxic), nitrogen is safely carried as glutamate
Why transamination is important
Prevents buildup of toxic ammonia
Collects nitrogen in one place (glutamate)
Prepares nitrogen for the next step → urea cycle


This slide is expanding the full pathway of amino acid breakdown—basically showing how the body handles nitrogen and then uses what’s left for energy. Let’s go step by step in a clean, intuitive way.
1. Dehydration of serine and threonine
👉 Special case for certain amino acids
Serine and threonine can lose water (dehydration)
This creates intermediates that can directly release ammonia (NH₃)
Why it matters:
This is an alternative way to remove nitrogen (not just transamination)
Produces:
Pyruvate (serine) → can go to glucose
α-ketobutyrate (threonine) → energy pathways
👉 Think: shortcut way to remove nitrogen
2. Oxidative deamination of glutamate
👉 Main step where nitrogen is actually released
Earlier, nitrogen was collected on glutamate
Now glutamate is converted:
Glutamate → α-ketoglutarate + NH₃
Enzyme: glutamate dehydrogenase
Occurs mainly in the liver
Why it matters:
This is where free ammonia is produced
Links amino acid metabolism to the TCA cycle
👉 Think: this is the “release nitrogen” step
3. Ammonia transport
👉 Moving toxic nitrogen safely through the body
Ammonia (NH₃) is toxic, so it’s not transported freely.
Instead, the body uses carriers:
Main carriers:
Glutamine
Transports ammonia from tissues → liver
Alanine (glucose-alanine cycle)
Moves nitrogen from muscle → liver
Why it matters:
Prevents ammonia toxicity (especially in brain)
👉 Think: package nitrogen safely for delivery
4. Reactions of the urea cycle
👉 Detox step in the liver
Ammonia → converted into urea
Happens in liver (mitochondria + cytosol)
Urea cycle goal:
Take 2 nitrogen atoms
Turn them into urea (safe, water-soluble)
Then:
➡ Urea → blood → kidneys → urine
👉 Think: final disposal of nitrogen
5. Fate of the carbon skeleton
👉 What happens after nitrogen is removed
Once NH₃ is gone, you’re left with the carbon backbone
This can become:
Glucose (gluconeogenesis) → glucogenic amino acids
Ketone bodies or fat → ketogenic amino acids
TCA intermediates → energy (ATP)
Examples:
Alanine → pyruvate → glucose
Leucine → acetyl-CoA → ketones
👉 Think: use the leftover carbon for fuel
Big picture (connect everything)
Remove nitrogen (transamination or dehydration)
Release ammonia (oxidative deamination)
Transport it safely (glutamine/alanine)
Detox it (urea cycle)
Use carbon skeleton (energy, glucose, or fat)
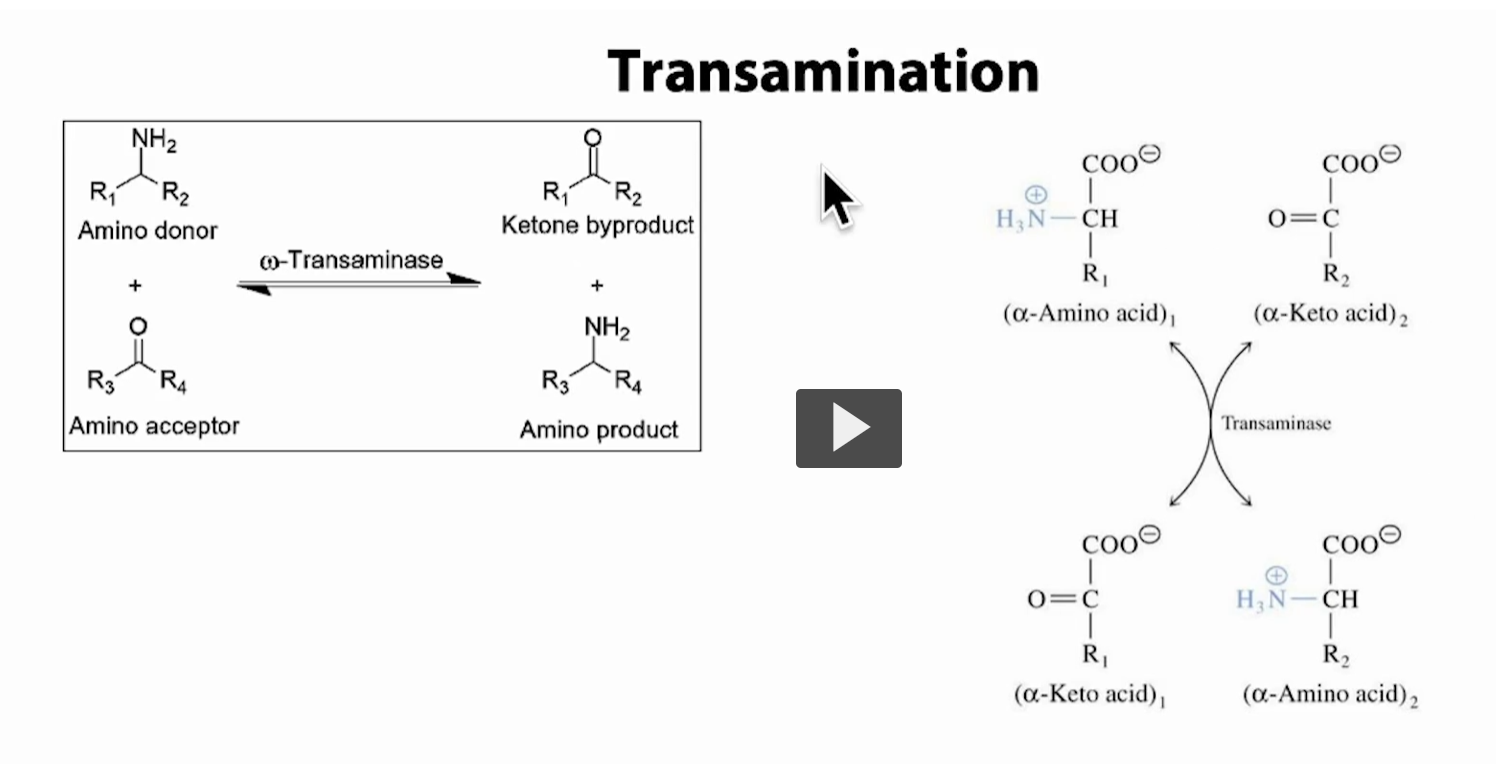
Transamination: transferring “trans” an amino group (–NH₂) “amination” from one molecule to another
Instead of releasing toxic ammonia directly, the body moves nitrogen safely between molecules.
What your slide is showing Left side (generic reaction)
Amino donor = an amino acid (has –NH₂)
Amino acceptor = a keto acid (no –NH₂)
After the reaction:
Donor loses NH₂ → becomes a keto acid (ketone byproduct)
Acceptor gains NH₂ → becomes a new amino acid
So: Amino acid₁ + Keto acid₂ ⇄ Keto acid₁ + Amino acid₂
Right side (same idea, more specific)
(α-amino acid)₁ → becomes (α-keto acid)₁
(α-keto acid)₂ → becomes (α-amino acid)₂
It’s reversible (can go both directions)
Key enzyme for transamination: transaminase
Called transaminase (aminotransferase)
Examples:
ALT (alanine aminotransferase)
AST (aspartate aminotransferase)
Requires vitamin B6 (PLP) as a cofactor
Why this process is important
1. Prevents ammonia toxicity
No free NH₃ released immediately
Nitrogen is safely transferred instead
2. Collects nitrogen onto glutamate
Most reactions funnel nitrogen to:
α-ketoglutarate → becomes glutamate
Glutamate = central “nitrogen collector”
3. Links amino acids to energy metabolism
When amino acid loses NH₂ → becomes keto acid
Keto acids enter:
TCA cycle
Gluconeogenesis
Ketone production
Example (high-yield)
Alanine + α-ketoglutarate ⇄ Pyruvate + Glutamate
Alanine loses NH₂ → becomes pyruvate
α-ketoglutarate gains NH₂ → becomes glutamate
This is one of the most important reactions (ALT)
Big picture (connect to previous slides)
Transamination → move nitrogen
Glutamate holds nitrogen
Later → deamination releases NH₃
NH₃ → urea cycle
Simple way to remember
“Swap the NH₂
One molecule gives NH₂
Another takes it
No free ammonia yet
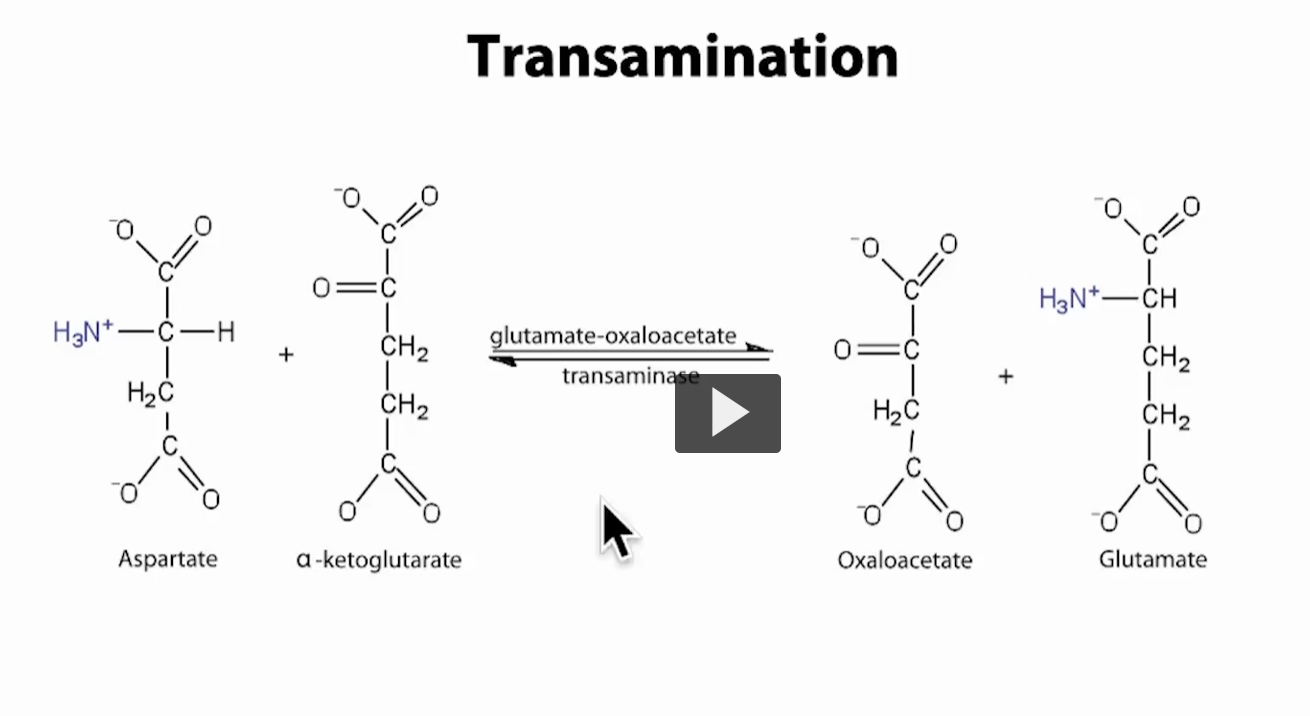
What this specific transamination reaction shows
This slide is a real example of transamination—not just the general idea.
The reaction (what’s happening)
Aspartate + α-ketoglutarate ⇄ Oxaloacetate + Glutamate
Aspartate = amino acid (has NH₃⁺ group)
α-ketoglutarate = keto acid
After the reaction:
Aspartate loses NH₂ → becomes oxaloacetate
α-ketoglutarate gains NH₂ → becomes glutamate
The enzyme for transamination: Glutamate-oxaloacetate transaminase (GOT)
Also called: AST (Aspartate Aminotransferase)
Step-by-step (simple logic)
Aspartate donates its amino group
α-ketoglutarate accepts that amino group
Products form:
Oxaloacetate (carbon skeleton of aspartate)
Glutamate (now carrying the nitrogen)
Why this reaction is important
1. Moves nitrogen safely
No free ammonia released yet
Nitrogen is now stored in glutamate
Glutamate = central nitrogen carrier
2. Feeds into the urea cycle
Aspartate is actually one of the nitrogen sources for urea
This reaction helps shuttle nitrogen into the system
3. Links to energy metabolism
Oxaloacetate enters the TCA cycle
Can be used for:
ATP production
Glucose (gluconeogenesis)
So:
Nitrogen → goes to disposal
Carbon → goes to energy
High-yield connections
This is one of the two major transaminases:
AST (this reaction)
ALT (alanine ↔ pyruvate)
Clinically:
AST levels ↑ in liver damage
Simple way to remember
👉 Aspartate gives NH₂ → becomes oxaloacetate
👉 α-ketoglutarate takes NH₂ → becomes glutamate
Big picture
This is just one example of the general rule:
👉 All amino acids transfer their nitrogen to α-ketoglutarate → forming glutamate
Then later:
Glutamate → releases NH₃ → urea cycle
What this slide is showing (in plain terms)
This is a specific example of transamination—the process where one molecule hands off an amino group (–NH₂) to another.
The reaction
Aspartate + α-ketoglutarate ⇄ Oxaloacetate + Glutamate
Read it like a swap:
Aspartate loses NH₂ → becomes oxaloacetate
α-ketoglutarate gains NH₂ → becomes glutamate
Step-by-step logic
Aspartate (amino acid) has a nitrogen
It donates that nitrogen
α-ketoglutarate accepts it
Products form:
Oxaloacetate (no nitrogen now)
Glutamate (now carrying nitrogen)
The enzyme
Glutamate–oxaloacetate transaminase (GOT)
Also called: AST (Aspartate Aminotransferase)
Why this reaction matters
1. Safely moves nitrogen
No free ammonia yet (important because NH₃ is toxic)
Nitrogen is stored in glutamate
Think: glutamate = nitrogen shuttle
2. Connects to the urea cycle
Aspartate is one of the nitrogen sources for urea
This reaction helps route nitrogen into disposal pathways
3. Feeds energy metabolism
Oxaloacetate goes into:
TCA cycle (Krebs cycle) → ATP
Gluconeogenesis → glucose
So:
Nitrogen → disposal (urea cycle)
Carbon → energy
High-yield connections
One of the two major transaminases:
AST (this reaction)
ALT (alanine ↔ pyruvate)
Clinically:
↑ AST = liver damage or muscle injury
Simple way to remember
👉 “Aspartate → Oxaloacetate (loses NH₂)”
👉 “α-ketoglutarate → Glutamate (gains NH₂)”
Big picture takeaway
This reaction is part of a larger strategy:
Collect nitrogen onto glutamate → later release it → convert to urea
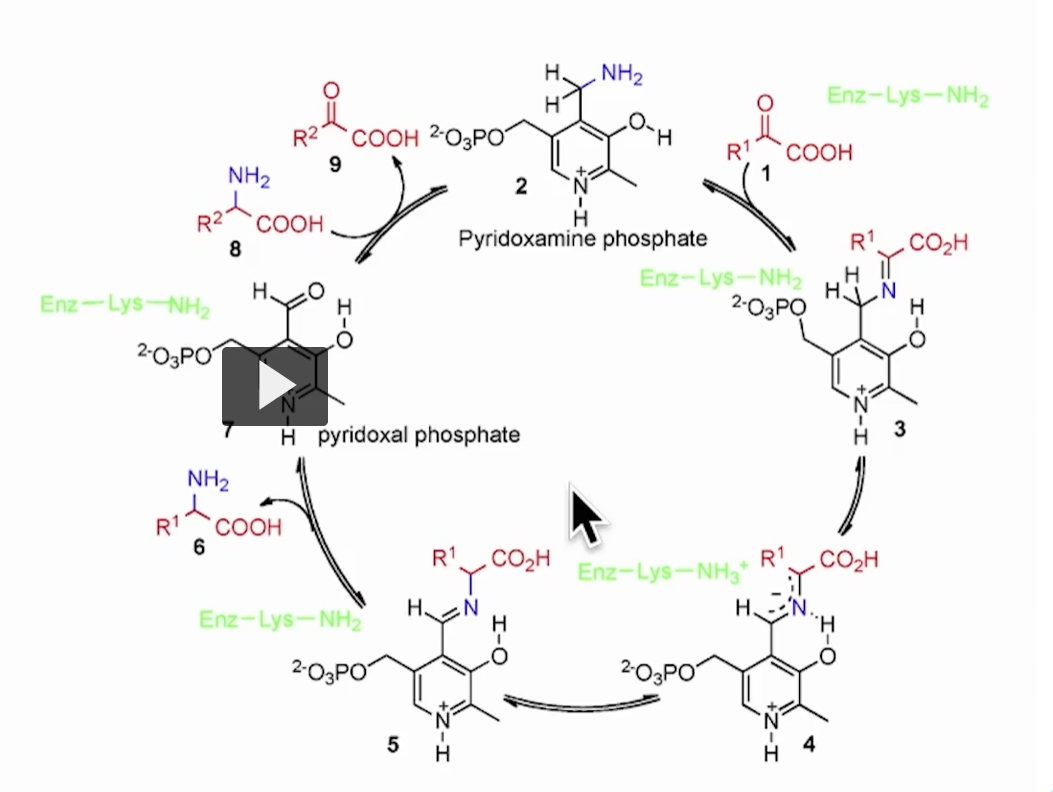
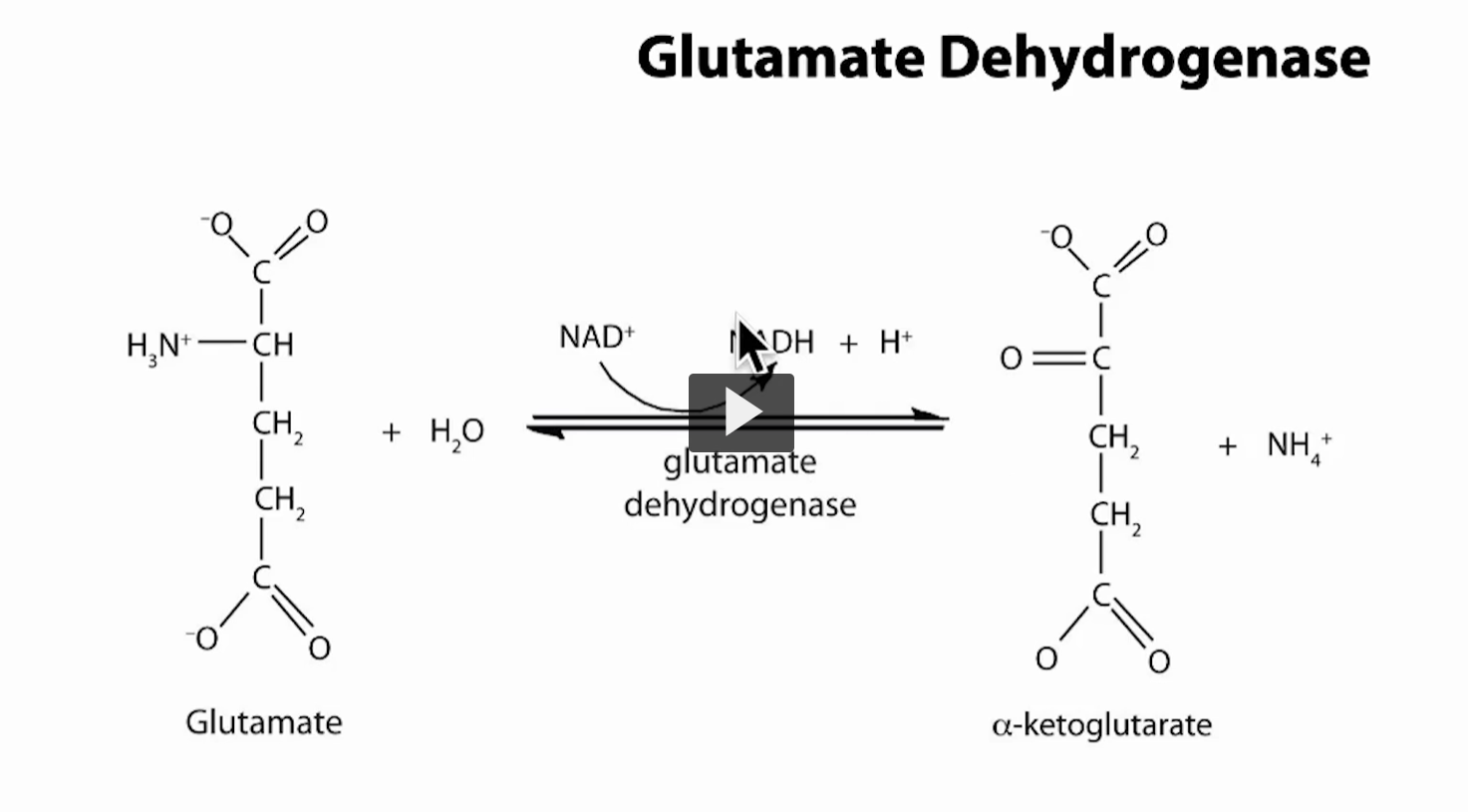
This is the key step where nitrogen is finally released as ammonia.
in the previous reaction of transamination (where aspartate becomes OAA, and alpha ketoglutarate becomes glutamate)
in this step, nitrogen, from glutamate, is being released as ammonia (NH3).
The reaction: Glutamate + H₂O + NAD⁺ ⇄ α-ketoglutarate + NH₄⁺ + NADH + H⁺
In words:
Glutamate loses its amino group
That nitrogen becomes ammonium (NH₄⁺)
The carbon skeleton becomes α-ketoglutarate
NAD⁺ is reduced → NADH
What kind of reaction is this? Oxidative deamination
Deamination = removing NH₂
Oxidative = electrons transferred to NAD⁺ → NADH (this is reduction, however, we are naming it based on the glutamate losing molecules, which is oxidation).
Step-by-step logic
Glutamate (holding nitrogen) enters
Enzyme removes NH₂
Nitrogen → released as NH₄⁺ (ammonia form)
Remaining molecule → α-ketoglutarate
NAD⁺ → becomes NADH (energy carrier)
The enzyme: Glutamate dehydrogenase (GDH)
Located in mitochondria in the liver.
Why this step is VERY important
1. This is where ammonia is actually released
All earlier steps (transamination) just moved nitrogen
This step frees it
First time NH₃/NH₄⁺ appears
2. NH4+ feeds directly into the urea cycle
NH₄⁺ → enters urea cycle → becomes urea → excreted
3. Links amino acids to energy metabolism
Product = α-ketoglutarate
This enters the TCA cycle
So:
Nitrogen → waste
Carbon → energy
4. Produces NADH
NADH → goes to electron transport chain
Generates ATP
Big picture connection
You can now see the flow:
Transamination → collect nitrogen on glutamate
Glutamate dehydrogenase (this step) → release NH₄⁺
Urea cycle → detoxify NH₄⁺
Carbon skeleton (α-ketoglutarate) → energy
This slide is showing how nitrogen is safely moved FROM muscle TO liver and disposed of, while also recycling carbon for energy. It combines three key ideas:
1. The big picture (what’s the goal?)
When muscle breaks down amino acids:
You get toxic nitrogen (NH₃ / NH₄⁺)
You also get carbon skeletons for energy
Problem: ammonia is toxic
Solution: package it (ammonia) as alanine → send to liver → convert to urea
2. Glucose–Alanine Cycle (top diagram)
In muscle:
Glucose → Pyruvate (glycolysis)
Amino acids lose nitrogen → becomes NH₄⁺
That nitrogen is transferred to pyruvate
via alanine aminotransferase
Pyruvate + NH₃ → Alanine
Alanine = safe nitrogen carrier
In blood: Alanine travels to the liver
In liver:
Alanine → Pyruvate + NH₃
Pyruvate → Glucose (gluconeogenesis)
NH₃ → Urea (detoxified)
Glucose (from gluconeogenesis) goes back to muscle → cycle repeats
3. The nitrogen flow (bottom diagram)
This is the core chemistry behind it all:
Step 1: Transamination : Moves nitrogen between molecules (no free ammonia yet)
Key reaction:
Amino acid + α-ketoglutarate ⇌ α-keto acid + glutamate
Glutamate = nitrogen collector
Step 2: Oxidative deamination
Now nitrogen is actually released
Glutamate → α-ketoglutarate + NH₃
This is where your earlier question comes in:
NAD⁺ → NADH
So:
Glutamate is oxidized
NAD⁺ is reduced
✔ That’s why it’s called oxidative deamination
Step 3: Urea cycle
NH₃ + CO₂ → Urea
Urea is excreted safely
4. How everything connects
Think of it like a logistics system:
Muscle
Packs nitrogen → alanine (safe transport)
Blood
Delivers alanine
Liver
Unpacks nitrogen → ammonia
Converts → urea (safe disposal)
Sends glucose back
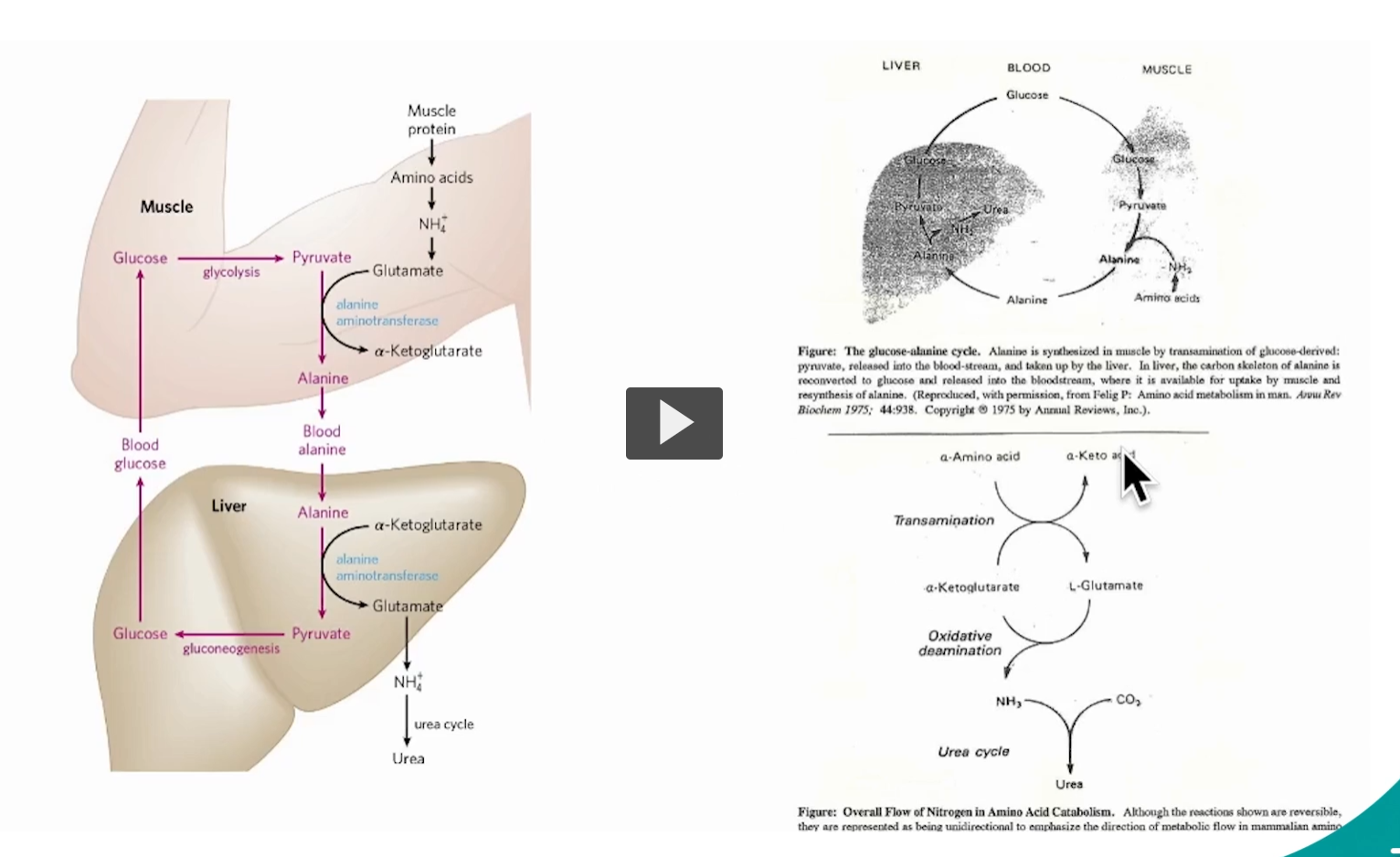

Non-oxidative deamination = removing NH₃ without using NAD⁺/NADH
No redox
No electron transfer
Just rearranging + breaking bonds
What’s special here?
Most amino acids: Transfer Nitrogen → glutamate → oxidative deamination
BUT
Serine & threonine can skip that (Serine and threonine can skip transamination because their side chains contain an –OH group that enables a dehydration reaction.) → they directly release NH₃
Mechanism (this is the key)
Step 1: PLP grabs the amino acid
Enzyme: serine dehydratase
Cofactor: PLP (vitamin B6)
PLP stabilizes the amino group and makes the molecule reactive
Step 2: Dehydration (this is the weird part)
H₂O is removed
OH from side chain
H from adjacent carbon
👉 This creates a double bond intermediate
Step 3: Rearrangement → unstable intermediate
Forms an imine-like structure (C=NH)
This is key: now the nitrogen is easier to remove
Step 4: NH₃ leaves: The amino group is released as NH₃
Step 5: Final product forms
Remaining carbon skeleton becomes: Pyruvate
Net reaction (super important)
Serine → Pyruvate + NH₃
⚖ Compare to oxidative deamination
Feature | Oxidative | Non-oxidative |
|---|---|---|
Uses NAD⁺? | ✅ Yes | ❌ No |
Uses glutamate? | ✅ Yes | ❌ No |
Direct NH₃ release? | ❌ No | ✅ Yes |
Amino acids | Most | Serine, Threonine |
Intuition (easy way to remember)
Serine has –OH group
→ That allows dehydration (loss of H₂O)Once water leaves:
Structure becomes unstable
NH₃ can leave easily
Big idea
When amino acids are degraded:
Nitrogen → urea (waste)
Carbon skeleton → used for energy or glucose
This slide focuses on the carbon part
🔥 What “gluconeogenic” means
Gluconeogenic amino acids = can be converted into glucose (amino acids that are able to make glucose “gluconeogenic)
👉 The carbon FROM AMINO ACIDS ends up as molecules that can:
enter the TCA cycle
then become oxaloacetate → glucose
The 5 key entry points
These are the molecules your slide lists — they are all TCA cycle intermediates or closely related
1. Pyruvate
Direct precursor to glucose
Can become: Oxaloacetate → glucose
👉 Example amino acids:
Alanine
Serine
2. Oxaloacetate (OAA)
Already a gluconeogenesis starting point
👉 Goes straight to: PEP → glucose
👉 Example: Aspartate
3. α-Ketoglutarate
Enters TCA cycle
Eventually becomes oxaloacetate
👉 Example: Glutamate
4. Succinyl-CoA
TCA intermediate → oxaloacetate
Example:
Methionine
Valine
5. Fumarate
TCA intermediate → oxaloacetate
Example:
Phenylalanine
Tyrosine
The unifying logic
All of these:
👉 Feed into the TCA cycle
👉 Become oxaloacetate (Goes straight to: PEP → glucose)
👉 Then → glucose (via gluconeogenesis)
KEY EXAM TRAP
Not all amino acids can make glucose.
👉 Only gluconeogenic ones do this
Contrast:
Glucogenic → glucose
Ketogenic → ketone bodies (NOT glucose)
Ketogenic (important!):
Leucine
Lysine
Leucine and lysine are ketogenic because their carbon skeletons become acetyl-CoA (or acetoacetate), NOT TCA intermediates that can make glucose.
Why that matters
To make glucose (gluconeogenesis), you need:
👉 Oxaloacetate (OAA)
But:
👉 Acetyl-CoA cannot be converted into oxaloacetate in humans (Glycolysis → pyruvate, Pyruvate → acetyl-CoA (via pyruvate dehydrogenase) This is called the link reaction (not a cycle)
Clean mental model
Think: Amino acids → remove nitrogen → carbon skeleton → plug into TCA → become glucose

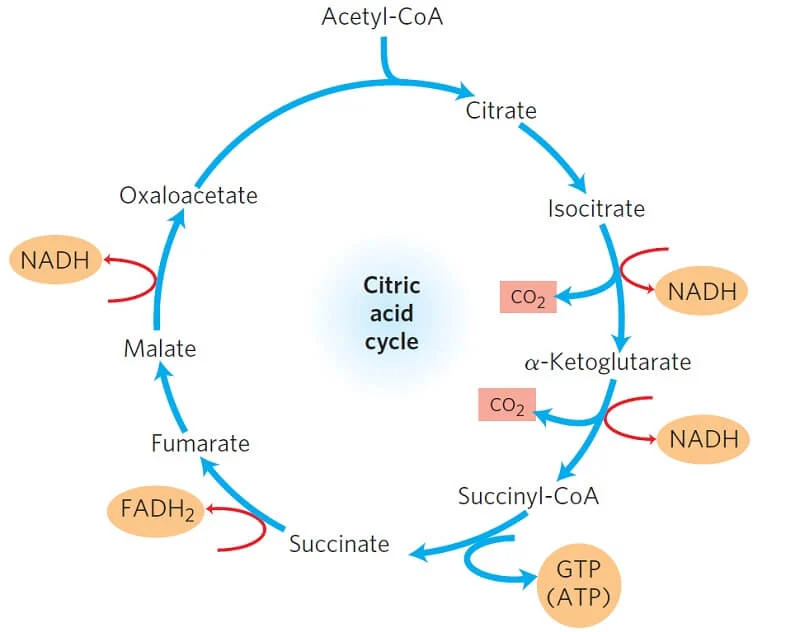
TCA cycle for reference of oxaloacetate
Position of oxaloacetate in the TCA cycle
Oxaloacetate (OAA) is both:
the last product of the cycle
and the starting molecule for the next turn
🔄 Order at the end of the TCA cycle
The final steps are:
Malate → Oxaloacetate
Oxaloacetate + Acetyl-CoA → Citrate (cycle restarts)
So:
Malate = second-to-last
Oxaloacetate = last
🔁 Why it’s confusing
Because OAA is:
immediately used up to form citrate
👉 it feels like it’s not the end
But chemically:
👉 it is the final product regenerated each cycle

This slide is about what the carbon skeletons of amino acids become after you remove nitrogen (via transamination/deamination).
Big idea
Once the amino group (NH₃) is removed, the leftover carbon skeleton:
either helps make glucose → “gluconeogenic”
or helps make ketone bodies / fat → “ketogenic”
1. Gluconeogenic vs Ketogenic (core distinction) Gluconeogenic amino acids
Become intermediates like:
pyruvate
oxaloacetate
α-ketoglutarate
succinyl-CoA
These can go → gluconeogenesis → glucose
Ketogenic amino acids
Become:
acetyl-CoA
acetoacetate
These go → ketone bodies or fatty acids, NOT glucose
Why not glucose?
Because acetyl-CoA carbons are lost as CO₂ in the TCA cycle, so there’s no net glucose production.
2. Aromatic amino acids (Phe, Tyr, Trp)
These are both gluconeogenic AND ketogenic.
👉 Why?
Because when they break down, they produce two types of products:
Some carbons → TCA intermediates → glucose
Some carbons → acetyl-CoA / acetoacetate → ketones
So they “split” into both pathways.
3. Lysine & Leucine (special case)
Purely ketogenic
They break down ONLY into:
acetyl-CoA
acetoacetate
❗ They cannot form glucose at all.
Easy way to remember
“LL = only fat”
👉 Lysine & Leucine = strictly ketogenicAromatic trio (Phe, Tyr, Trp)
👉 both (they’re versatile)
🧬 Why this matters (clinically + MCAT)
During fasting/starvation:
gluconeogenic AAs → maintain blood glucose
ketogenic AAs → fuel brain (via ketones)
Test favorite:
“Which amino acids are purely ketogenic?”
✔ Leucine & Lysine


Big idea
After deamination, the carbon skeleton enters metabolism at specific points:
All of these products can make glucose → gluconeogenic
The “carbon-count rule” 3-carbon amino acids → Pyruvate
End up as pyruvate (3C) (because pyruvate has 3 carbons)
Pyruvate can:
go → glucose (gluconeogenesis)
go → acetyl-CoA (energy, enters into the Krebs cycle)
Think: small = pyruvate
4-carbon amino acids → Oxaloacetate (OAA)
End up as oxaloacetate (4C) (because OAA has 4 carbons)
Oxaloacetate is:
a direct gluconeogenesis substrate
part of the TCA cycle
Very efficient for making glucose
5-carbon amino acids → α-Ketoglutarate
End up as α-ketoglutarate (5C) (because alpha-ketoglutarate has 5 carbons)
➡ This enters the TCA cycle, then can become:
oxaloacetate → glucose
according to the figure for a-ketoglutarate, it’s already a substrate for the Kreb’s Cycle
🔄 Why this works (connect the dots)
All three products:
Pyruvate (end of glycolysis)
Oxaloacetate (end of TCA and also start of TCA)
α-Ketoglutarate (middle of TCA)
are either:
already gluconeogenesis substrates
or can become one through the TCA cycle
Contrast with ketogenic (important)
These DO NOT follow this rule:
Leucine & Lysine → acetyl-CoA only → NO glucose
Because: acetyl-CoA carbons are lost as CO₂
no net glucose production
Easy memory shortcut
3C→ Pyruvate
4C → OAA
5C → α-KG
count every carbon in the molecule.
All = gluconeogenic pathway
🧬 Why this matters (test logic)
If you see:
amino acid → pyruvate / OAA / α-KG
✔ Answer = gluconeogenic
If you see:
amino acid → acetyl-CoA
✔ Answer = ketogenic
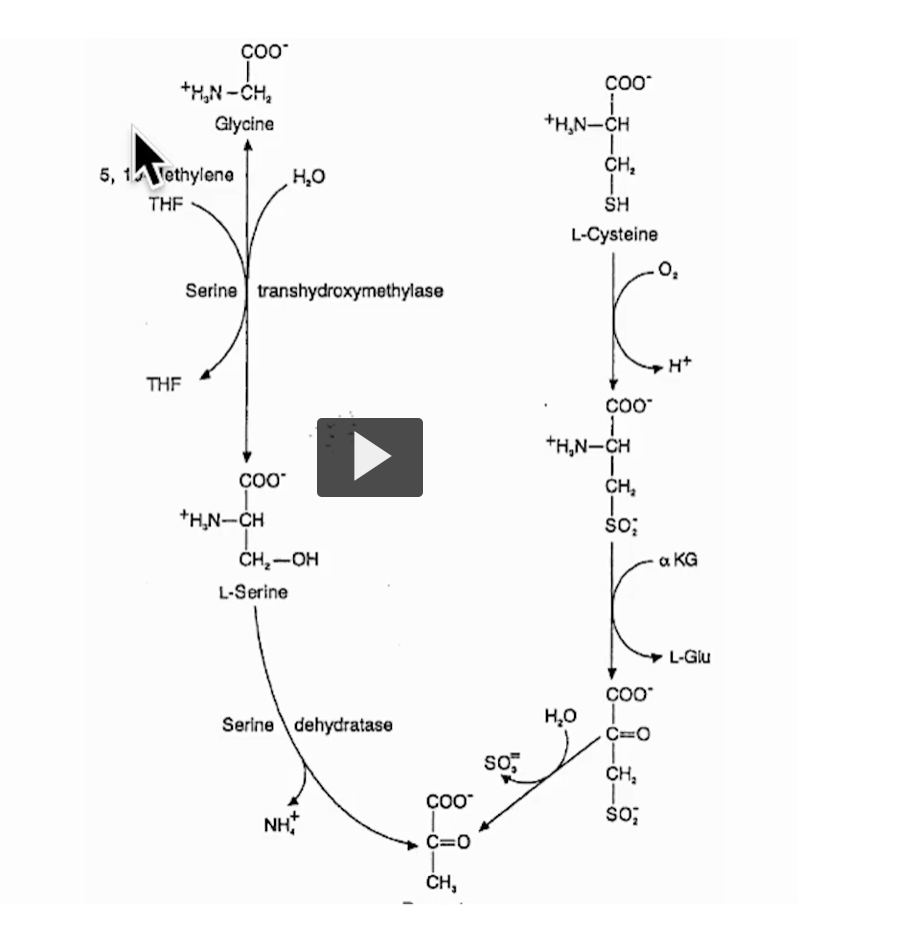
This slide is showing how serine, glycine, and cysteine are metabolized, and how their carbons end up as pyruvate (→ gluconeogenesis) while also linking to nitrogen metabolism and one-carbon metabolism.
Let’s break it cleanly 👇
LEFT SIDE: Serine ↔ Glycine (One-Carbon Metabolism)
Key reaction: Serine ⇄ Glycine
Enzyme: serine hydroxymethyltransferase
What’s happening?
Serine (3C) loses one carbon
That carbon is transferred to THF (tetrahydrofolate)
Forms:
Glycine (2C)
5,10-methylene-THF
🔑 Why this matters:
THF carries 1-carbon units
Used for:
DNA synthesis
nucleotide production
👉 This is part of folate metabolism (VERY high-yield)
🔥 ALSO: Serine → Pyruvate
Enzyme: serine dehydratase
Reaction:
Removes NH₄⁺ (ammonia)
Leaves behind pyruvate
👉 This shows:
✔ Serine is gluconeogenic
🧠 RIGHT SIDE: Cysteine → Pyruvate
Step-by-step: 1. Cysteine oxidation
Cysteine + O₂ → oxidized form (adds oxygen to sulfur)
2. Transamination
Uses α-ketoglutarate (α-KG)
Produces:
L-glutamate (L-Glu)
modified cysteine intermediate
3. Sulfur removal
Releases SO₃²⁻ (sulfite)
4. Final product:
👉 Pyruvate
🔑 Big picture (connect everything)
All three amino acids:
Amino Acid | Final Carbon Product |
|---|---|
Serine | Pyruvate |
Glycine | → Serine → Pyruvate |
Cysteine | Pyruvate |
👉 Therefore:
✔ All are gluconeogenic
⚠ Important insights 1. These AAs can release free NH₄⁺ directly
Unlike most AAs:
they don’t always need glutamate first
2. Link between pathways
This slide connects:
Amino acid metabolism
Folate (1-carbon) metabolism
TCA cycle (via pyruvate)
Nitrogen disposal
🔥 Easy way to remember
👉 “Serine family → pyruvate”
Serine → pyruvate
Glycine → serine → pyruvate
Cysteine → pyruvate
🧬 Why this is high-yield
Folate cycle questions (THF!!)
Which AAs → pyruvate
Direct NH₃ release (serine, threonine similar idea)
Gluconeogenesis during fasting