Chemistry all of reactivity
1/165
Earn XP
Description and Tags
IB chemistry 2025 spec R1, R2, and R3
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
166 Terms
What is a Bronsted-Lowry acid?
A substance that donates protons (H+) in a chemical reaction.
What is a Bronsted-Lowry base?
A substance that accepts protons (H+) in a chemical reaction.
What is a Lewis acid?
A substance that can except an electron pair.
What is a Lewis base?
A substance that can donate an electron pair.
What is a nucleophile?
A chemical species that donates an electron pair to form a chemical bond in a reaction.
What is an electrophile?
A chemical species that accepts an electron pair to form a chemical bond in a reaction.
What is electrophilic substitution?
A chemical reaction in which an electrophile replaces a substituent in an aromatic compound.
What is electrophilic addition?
A reaction between an electrophile and nucleophile, adding to double or triple bonds
What is a conjugate base?
A Bronsted-Lowry acid after it has donated a proton.
What is a conjugate acid?
A Bronsted-Lowry base after it has accepted a proton.
What is nucleophilic attack?
The process by which a nucleophile donates an electron pair to an electrophile.
What is electrophilic attack?
The process by which an electrophile accepts an electron pair from a nucleophile.
What is a ligand?
A molecule or ion that binds to a central metal atom to form a coordination complex.
Define enthalpy
The total heat energy of a system at constant pressure. Often used to quantify energy changes during a reaction.
Define specific heat capacity
The energy required to raise 1kg of a substance by 1K
What is the standard enthalpy of atomisation?
The enthalpy change when an element in its standard state is converted to 1 mole of gaseous atoms.
What is Hess’s Law?
the total enthalpy change during the complete course of a chemical reaction is independent of the pathways taken if the starting and ending points are the same
What is oxidation?
Increase in oxidation state (loss of electrons)
What is reduction?
Decrease in oxidation states (gain of electrons)
What is a carbocations?
Positive carbon ions
What is the difference between an open, closed and isolated system?
In an open system matter and energy can transfer across the boundary. In a closed system no matter can be transfered but energy can. In an isolated system matter and energy can neither enter nor exit the system
What is heat?
A form of energy transferre from a warmer body to a cooler body as a result of a temperature gradient.
What is bond dissociation energy?
The energy absorbed by the reaction system to break the chemical bond. It is endothermic.
Is bond making endo or exo thermic
Exothermic. When chemical bonds are made energy is released into the surroundings. (Negative enthalpy)
What is the standard enthalpy change for a reaction?
The heat transferred at constant pressurre under standaard conditions and states.
When reactants are less stable than products the reaction is …
Exothermic (and usually spontaneous)
Define bond enthalpy
The enrgy required to break one mole of bonds by homolytic fission in one mole of gaseous covalent molcules under standard conditions.
Why aren’t bond enthalpies exact.
They are average values
What is Hess’s Law
Regardless of the route by which a chemical reaction proceeds, the enthalpy change will always be the same, as long as the initial and final states of the system are the same.
Define Enthalpy of formation (DfHº):
Enthalpy change when one mole of a substance is formed from its constituent elements with all substances in their standard states
Define Enthalpy of combustion (DcHº)
Enthalpy change when one mole of a substance undergoes complete combustion in oxygen with all substances in standard states
Is ionisation enthalpy/energy endo or exo thermic?
The ionisation enthalpy is always endothermic (takes in energy from the surroundings)
explain the first ionisation energy
enthalpy change when each atom in one mole of gaseous atoms loses one electron to form one mole of gaseous 1+ ions
What is the second ionisation energy
enthalpy change when each ion in one mole of gaseous 1+ ions loses one electron to form one mole of gaseous 2+ ions
What is the first electron affinity
enthalpy change when each atom in one mole of gaseous atoms gains one electron to form one mole of gaseous 1– ions.
What is the second electron affinity
enthalpy change when each ion in one mole of gaseous 1– ions gains one electron to form one mole of gaseous 2– ions.
What is Enthalpy of atomisation (DatHº)
Enthalpy change when one mole of gaseous atoms is produced from an element in its standard state. (an endothermic process)
Enthalpy of solution (DsolHº)
Enthalpy change when one mole of an ionic solid dissolves in an amount of water large enough so that the dissolved ions are well separated and do not interact with each other.
What is crued oil?
It is a fossil fuel that is a natural mixture of hydrocarbons, organic compunds containing nitrogen, sulfur and oxygen, and a wide variety of other elements.
What happens during the complete combustion of alkanes
Alkanes undergo complete combusion in the presence of excess oxygen. The reaction is exothermic and it produces carbon dioxide and water
alkane + oxygen → carbon dioxide + water
What happens during the incomplete combustion of alkanes
alkane + oxygen → carbon monoxide + water
alkane + oxygen → carbon (soot) + water
What is the specific energy of a fuel?
The amount of heat energy released per mass of the fuel
What are biofuels ?
Renewable resources produced from organic compounds
What is biological carbon fixation?
The production of organic compounds from carbon dioxide. e.g. photosynthesis
What is the equation for photosythesis?
6 CO2 + 6 H2O → C6H12O6 + 6 O2
What is the equation for the fermentation of glucose
C6H12O6 (aq) → 2C2H5OH(aq) + 2CO2(g)
Law of conservation of energy
States that the total energy of a closed system remains constant and that energy can neither be created nor destroyed, only transferred.
What is lattice enthalpy
The enthalpy change that occurs when one mole of an ionic solid is dissociated into its gaseous ion
What is a primary (voltaic) cell
An electrochemical cell that converts energy from spontaneous redox reactions into electrical energy. These reactions are usually irreversible
What is the difference between primary and secondary cells?
In primary cells the reactions are usually irreversible. In secondary cells the chemical reactions are reversibe so the battery can be recharged.
What is a fuel cell?
A cell used to convert chemical energy of a fuel directly into electrical energy.
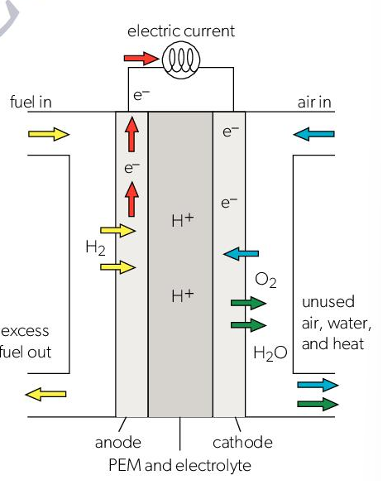
What is a proton exchange membrane (PEM) in a hydrogen fuel cell?
A membrane that selectively allows hydrogen ions (protons) to diffuse between the cathode and anode but prevents the passge of other ions, molecules and electrons between these electrodes.
Draw a hydrogen fuel cell
In a hydrogen fuel cell what is the half equation at the anode.
H2(g) → 2H+(aq) + 2e-
In a hydrogen fuel cell what is the half equation at the cathode.
O2(g) + 4e- + 4H+(aq) → 2H2O(l)
What are the benefits of using a direct-methanol fuel cell (DMFC) rather than a hydrogen fuel cell
methanol can be produced from renewable resources through fermentation
Methanol production releases less green house gases
Methanol has a greater energy density than hydrogen gas
State the half and overall equations for the electrochemical reactions in a direct-methanol fuel cell (DMFC)
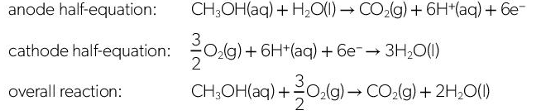
What does it mean for a reaction to be spontaneous?
It moves toward completion or equilibrium under a given set of conditions without external intervention
Define entropy S
A measure of the distribution of the total avaible enrgy or matter in a system. (Measure of disorder within a system)
What is the second law of thermodynamics?
Chemical reactions are spontaneous if they result in an overall increase in the total entropy value
What is the change is Gibbs energy dG
is the maximum amount of energy that can be obtained from a system
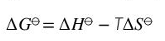
What are the units for Gibbs energy
kJ/ mol
Is a reaction with a positive change in enthalpy (exothermic) and positive change is entropy spontanious?
Gibbs free energy can be positive or negative therefore spontaneity is dependent of the temperature of the system

What is the reaction quotient Q?
The ratio of concentration of products to reactants

What is Gibbs energy change when the the equilibrium constant is 1 K=1
dG = 0
What is a limiting reactant
The reactant that is used up completely in a reaction while the other reactants are present in excess. It controls the amount of product formed and therefore the theoretical yield.
What is the law of conservation of mass?
In a chemical reaction the total mass of the reaction products is equal to the total mass of the consumed reactants
How is percentage yield calculated
experimental yield / theroretical yield x 100%
What is the instantaneous reaction rate
The concentration change over an infinitsimally small period of time.
What is the average kinetic energy (according to kinetic molecular theory)
The avergae kenetic energy of gas particles is proportional to the absolute temperature of the gas
How does concentration of a reactant affect the rate of reaction
An increase in the concentration of reactant leads to more frequenct collisions between the particles of that reactant and other species, so the reaction rate increase. (And vice versa)
What is a phase
an indivdual substance or mixture that has uniform chemica and physical prperties
What is activation energy?
The minimum kineic energy of colliding particles required for the reaction to occur
What is the transition state of a reaction
The least stable arrangement of atoms in the reacting species
What is a catalyst
A substance that increases the rate of a chemical reaction but is not consumed in the reaction. By providing an alternative pathway for a reaction, a catalyst reduces its activation energy. Usually a catalyst recats with a reactant to form an unstable compound known as an intermediate.
What is a mulitstep reaction
A reaction mechanism with more than 1 elementary step
What happens in an elementary step
1, 2 (or very rarely 3) molecular species undergo a chemical chage through a single transition state and no intermediates.
Why are termolecular reactions very rare
3 particles are very unlikely to collide with each other at the exact same time and in the correct orientation
What is the rate-determing step.
The slowest step of a reaction which limits the overall rate of reaction
How is the arrehenius equation used in a graph
ln(k) is plotted against 1/T. The gradient of the line is equal to -Ea/R and the y-intercept is ln(A)
What are the units of activation energy Ea (in the arrehenius equation)?
J/mol
What is a hetrogeneous equilibrium
An equilibrium where the species are in different phases
A saturated solution …
has the highest possible concentration of the solute under given conditions
When a system is at equilibrium
chemical and/or physical changes continue at the microscopic level
the forward and reverse reaction proceed at equal rates
the concentrations of reactants and products remains constant
macroacopic properties of the system, such as colour or density, remain unchanged
the same equilibrium state can be achieved from either direction
What is Le Chantelier’s principle

How does pressure effect the position of equibrium
If pressure is increased, the equilibrium shifts to the side of the reaction with fewer gaseous molecules to reduce pressure.
Decrease in Pressure: If pressure is decreased, the equilibrium shifts to the side with more gaseous molecules to increase pressure.
If the number of gaseous molecules is the same on both sides of the equation, pressure changes will have no effect on the position of equilibrium.
What is the only factor that effects both the position of equilibrium and the equilbirum constant
Temperature
How do catalysts affect the position of equilibrium
They don’t. Catalsyst increase the rate of both the forward and reverse reaction so equilibrium position does not shift.
What is K
The equilibrium contant. At equilbrium the reaction quotient Q = K
What happens when Q < K
The reaction contain more reactants and less product than needed for equilibrium so the formard reaction will be favoured
What happens when Q>K
there are less reactants and more products than needed for equilibrium so the reverse reaction will be favoured
What is an arrhenius acid
A substance that dissociates in water to form hydrogen ions (this has been replaced by Brosted-Lowry theory - a proton donor)
What is an arrhenius base
A substance that dissociates in water to form hydroxide ions (this has been replaced by Brosted-Lowry theory - a proton acceptor)
What are cojugate acid-base pairs
Acid-base pairs in which the species differ by exactly one proton
What is amphiprotic
species that can be both a borsted lowry acid or base ( can accept or donate a proton
What is amphoteric
A species that can react with both acids and bases
What is monoprotic
An acid that contains one exchangable hydrogen atom e.g. HCl
What is diprotic
An acid that contains two exchangable hydrogen atom e.g. H2SO4
What is does the stregth of an acid refer to
How easily the acid dissociates in aqueous solutions