Chemistry Quiz 8
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
If solid is more dense than liquid,
there is a positive slope.
If liquid is more dense than solid,
there is a negative slope.
Dissolving an involatile solute in a solvent, what does it do to the vapor pressure of the solvent?
lowers the vapor pressure of the pure solvent
Posolvent>Psolution
Dissolving an involatile solute in a solvent, what does it do to the freezing point of the solvent?
Lowers the freezing point of the pure solvent.
Tf (solution) < Tf (solvent)
dTf= Tf (solvent) - Tf (solution)
dTf = i * Kf * m
Dissolving an involatile solute in a solvent, the boiling point of the solution is
greater than the boiling point of the pure solvent.
dTf= Tf (solution) - Tf (solvent)
dTf = i * Kb * m
i = Van’t Hoff factor
= number of solute particles in solution
NaCl → Na + Cl-, i=2
The lower the boiling point.. . .
The more volatile the subtance.
The greater the vapor pressure,
the more volatile the substance.
The lower the moral mass,
the more volatile the substance
Mole fraction
= nA / (nA+nB)
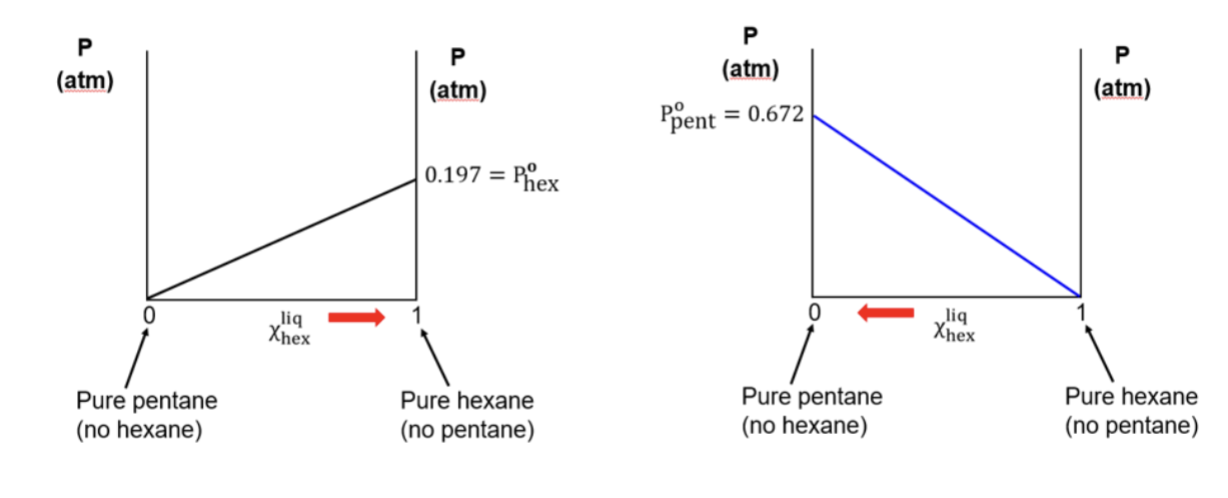
Vapor composition diagram
draw based on amount of gas A vs gas B
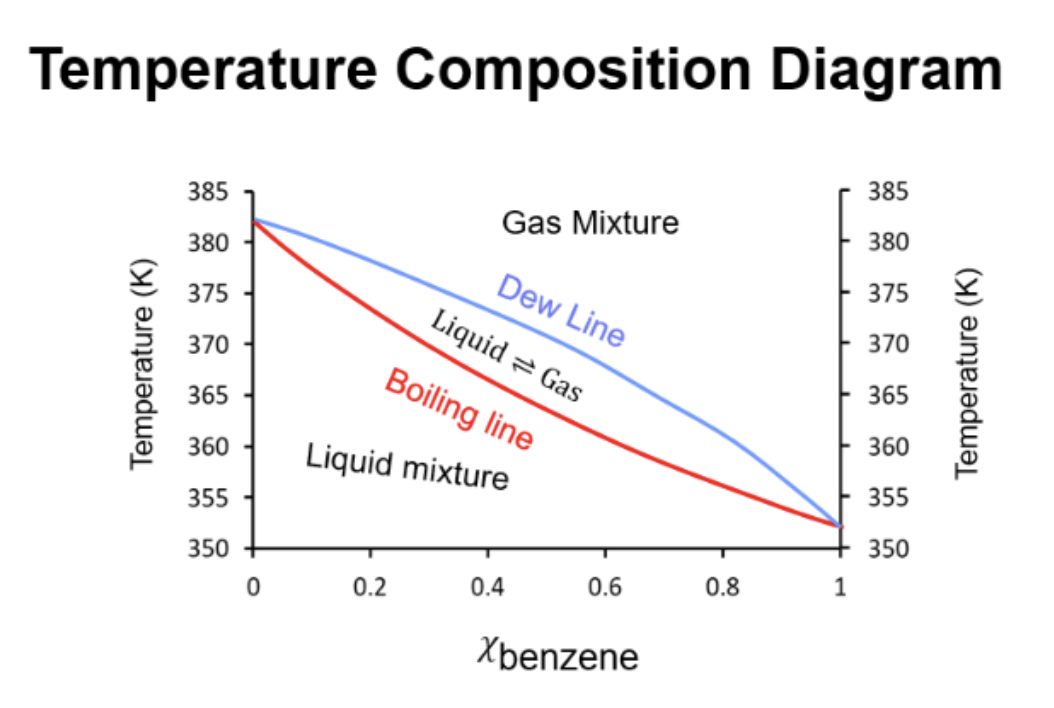
Temperature composition diagram
Dew Line is the top. Boiling Line is the bottom.
How to find the boiling range in a temp. composition diagram
boiling range is T of the boiling to T of the dew life for any mole fraction.
Finding mole fraction liq and mole fraction vap of a substance on a composition diagram
The liquid composition is read from the boiling line. The vapor composition from the dew line.
Fractional distillation
A separation process that relies on differences in boiling points of components in a mixture to achieve separation. It involves heating a liquid to create vapor and then condensing the vapor back into liquid to collect the separated components.