MCDB 310 Lecture 19
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
How are lipids stored in adipocytes mobilized for energy?
Hormones Trigger Mobilization of Stored Triacylglycerols
- Cytosolic acylglycerol lipases are activated by hormones glucagon and epinephrine
What happens to the fatty acids?
Fatty acids are released into the blood and are taken by serum albumin protein (10 Fatty Acids per albumin) to heart, skeletal muscles, and other tissues that need energy
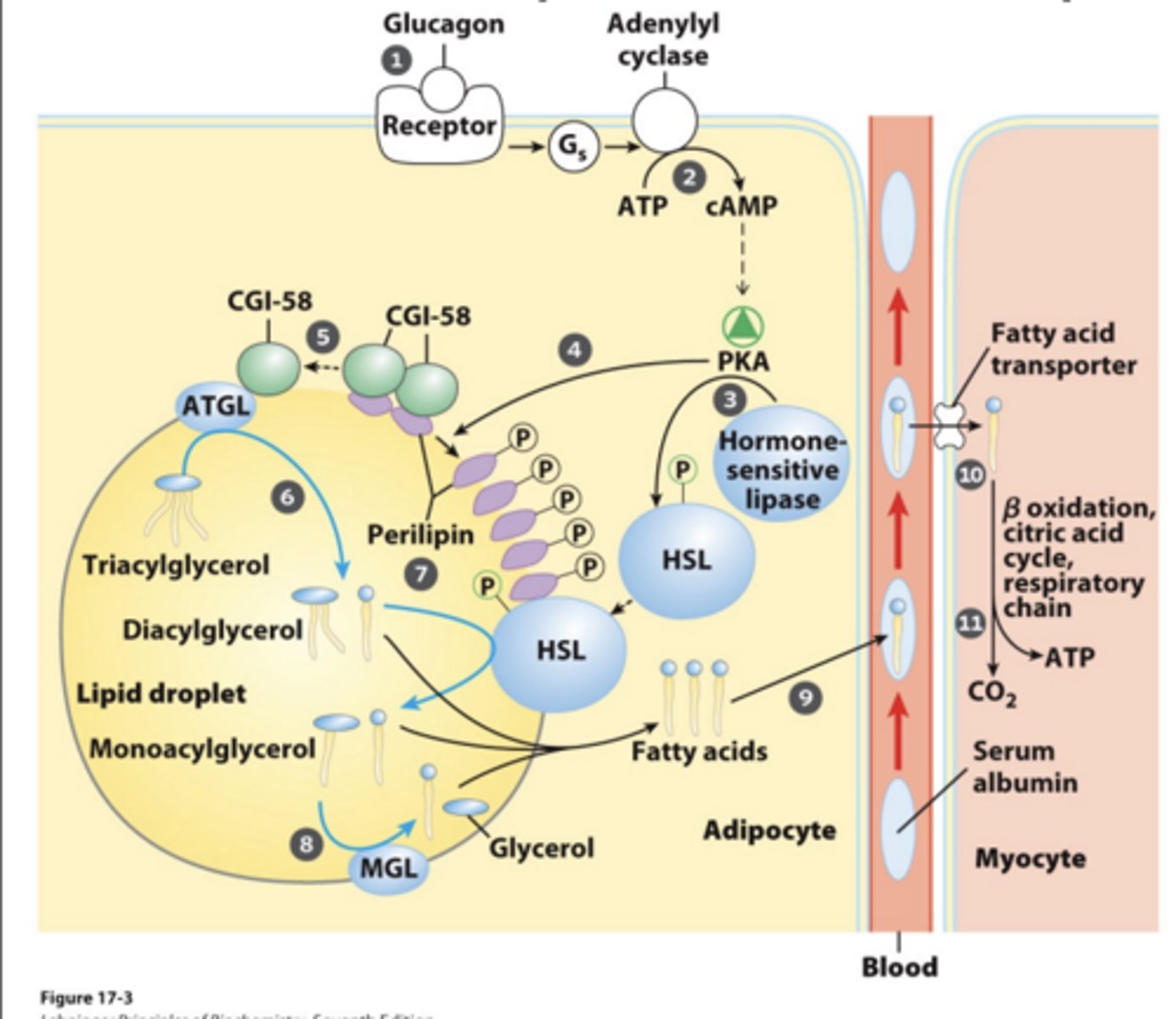
Lipases role with fatty acids
Cleave Fatty Acids from Glycerol Backbone of Triacylglycerides
What is the molecule that actually enters glycolysis
Glycerol from Fats Enters Glycolysis
What activates glycerol in this mechanism
Glycerol kinase activates glycerol at the expense of ATP.
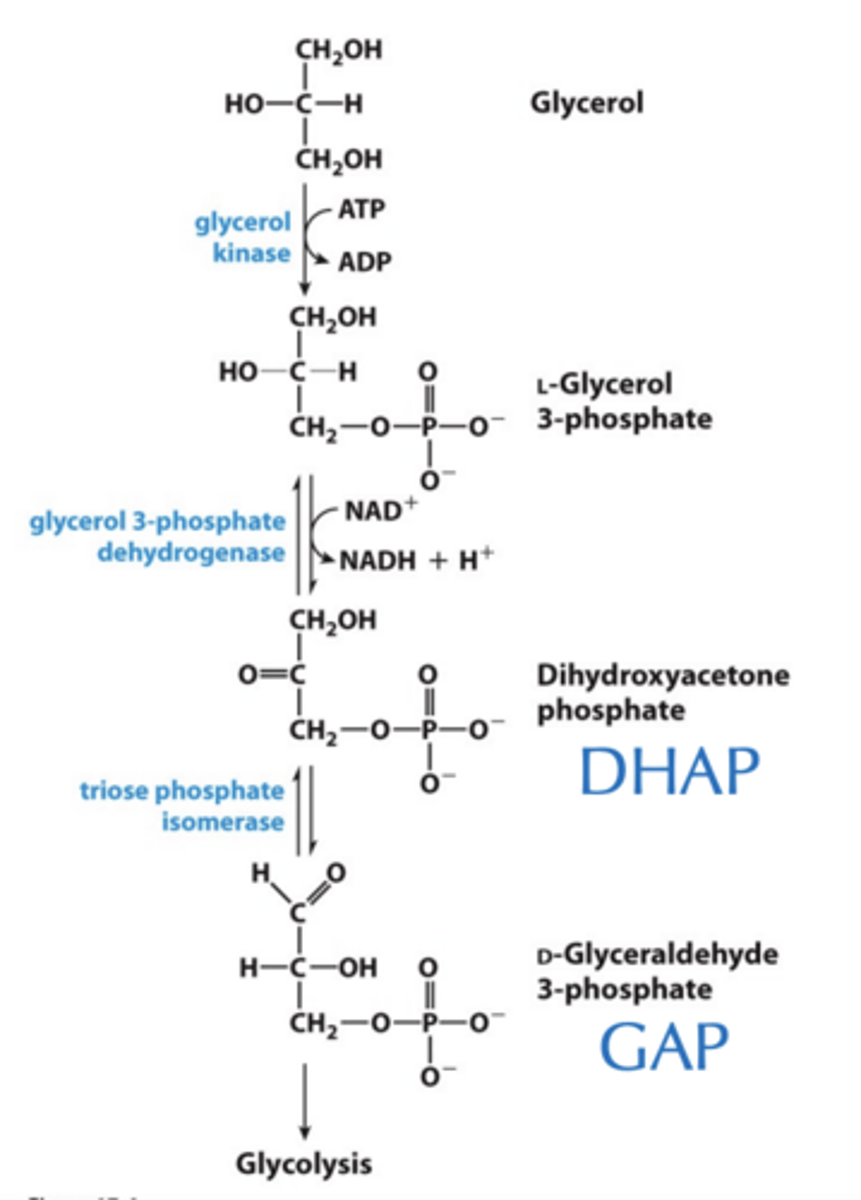
Result of subsequent reactions and what this process allows for?
•Subsequent reactions recover more than enough ATP to cover this cost.
•Allows limited anaerobic catabolism of fats
How are Fatty Acid Transported into Mitochondria? Small vs larger?
• Small (< 12 carbons) fatty acids diffuse freely across mitochondrial membrane.
• Larger fatty acids (most free fatty acids) are transported to the mitochondrial matrix via acyl-carnitine through the carnitine transporter/shuttle.
Where does b oxidation of fatty acyl CoA occurs ? What demonstrates this?
in mitochondria
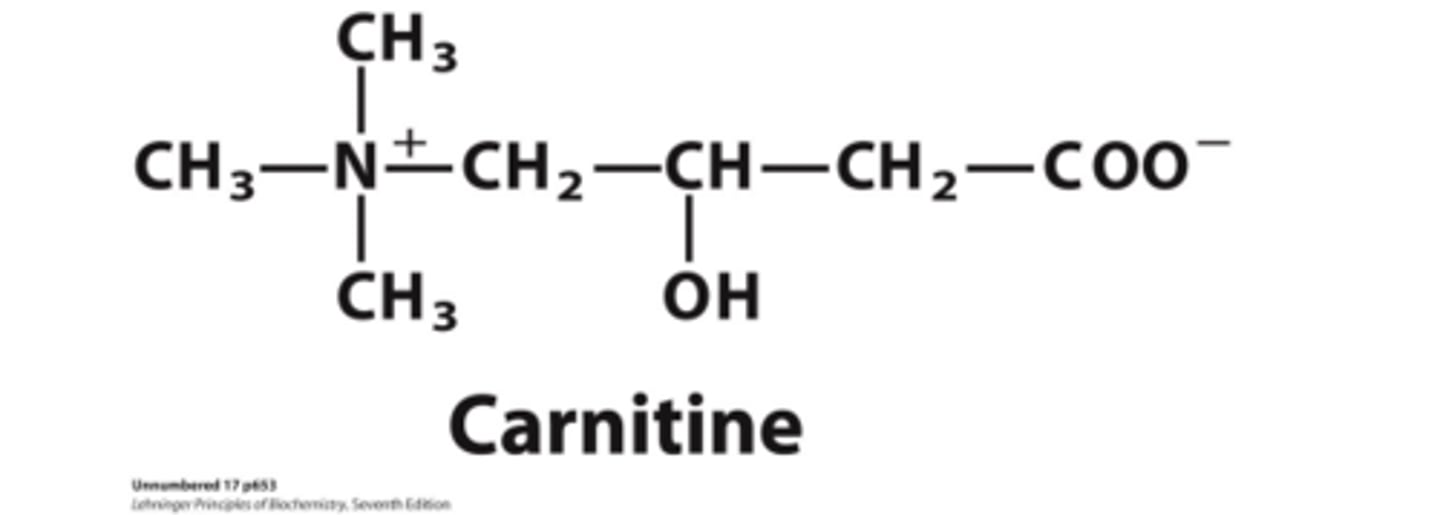
Acyl-Carnitine/Carnitine Transport process? Number of steps, rate, and point of regulation?
• Four-step process
• Rate-limiting for entire fatty acid oxidation
• Point of regulation for fatty acid oxidation
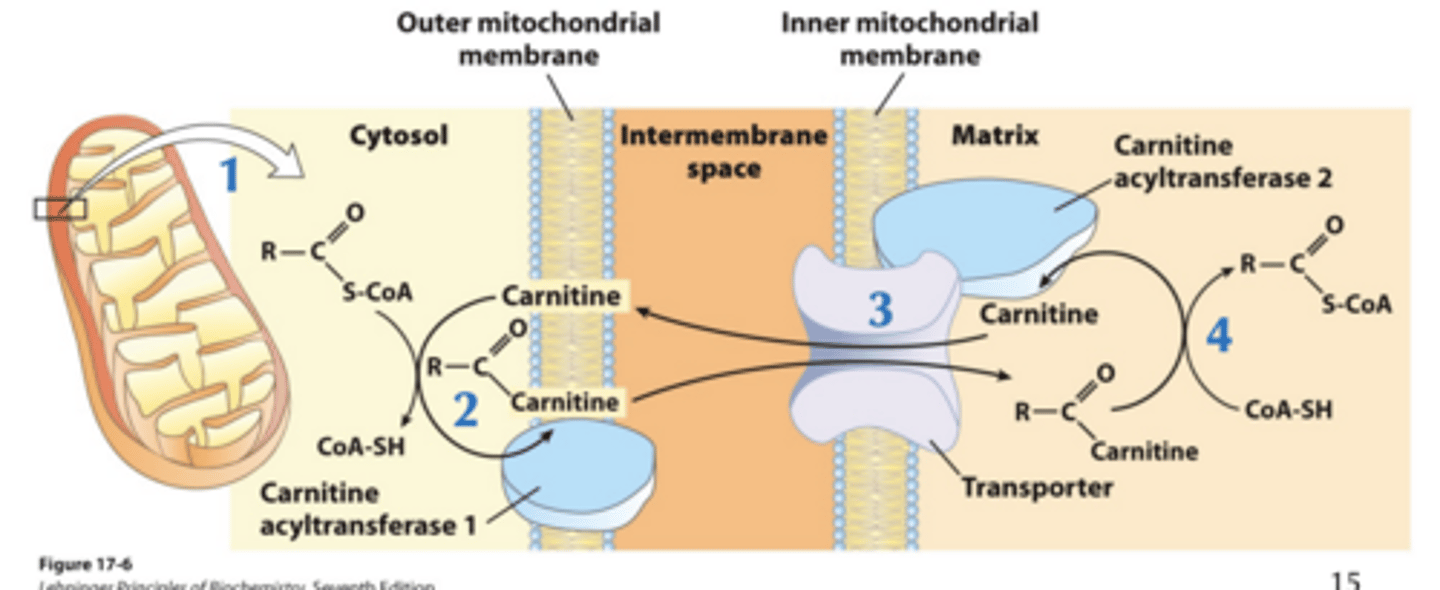
Acyl-Carnitine/Carnitine Transport step 1 , enzyme and location?
fatty acid + CoA + ATP -> fatty acyl CoA + AMP + 2Pi
Enzyme: Acyl CoA synthetase
Location: Outer mitochondrial membrane
Acyl-Carnitine/Carnitine Transport step 2 , enzyme and location?
fatty acyl CoA + carnitine -> Fatty acyl-carnitine.
Enzyme: carnitine acyl transferase 1 (CPT1) Location: Outer mitochondrial membrane
Acyl-Carnitine/Carnitine Transport step 3?
Fatty acyl-carnitine goes into the mitochondrial matrix through the acyl-carnitine/carnitine co-transporter (or antiporter) on the inner mitochondrial membrane. Carnitine is transported in the opposite direction
Acyl-Carnitine/Carnitine Transport step 4 , enzyme and location?
Fatty acyl-carnitine -> fatty acyl CoA + carnitine
Enzyme: carnitine acyl transferase 2 (CPT2) Location: Mitochondrial matrix
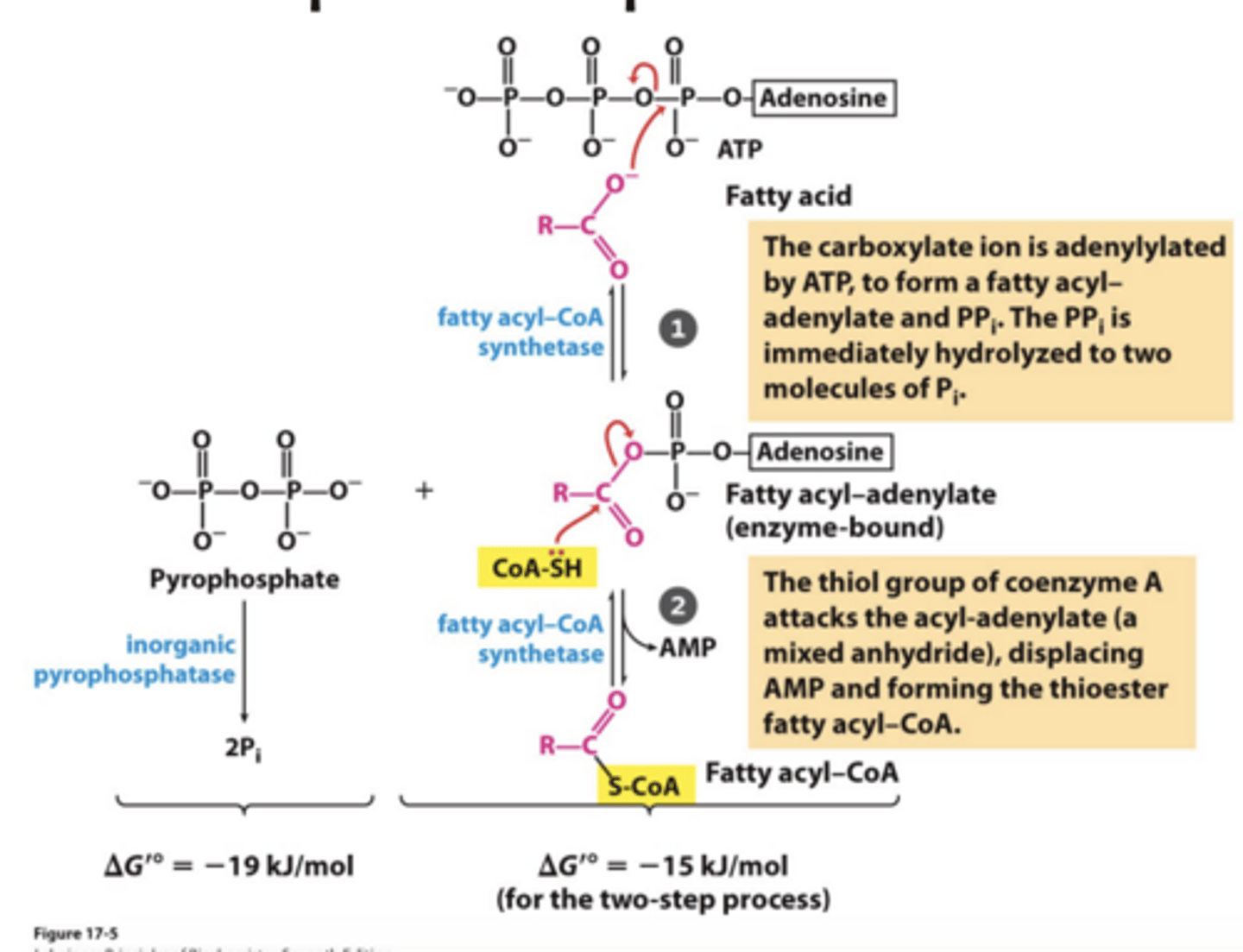
What Conversion does this Transport Require?
Conversion to Fatty Acyl-CoA
- 2 high-energy phosphates invested/used (ATP -> AMP and PPi -> 2Pi) equivalent to 2ATP -> 2ADP + 2Pi to activate fatty acid into fatty acyl CoA
Fatty Acid Oxidation in the Mitochondria occurs in how many stages?
3, but 3rd in on exam 4
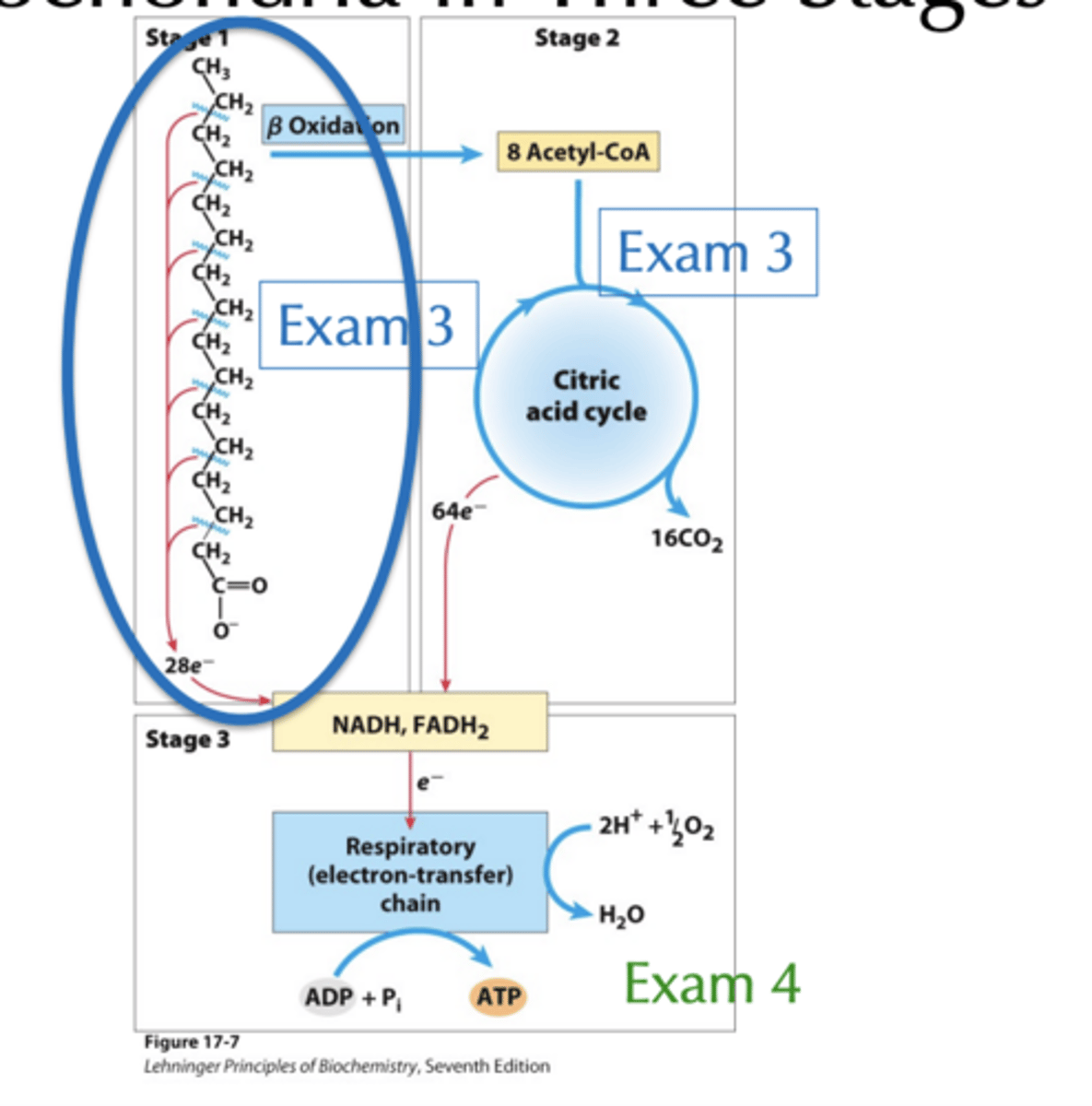
Fatty Acid Oxidation in the Mitochondria stage 1
Stage 1 consists of oxidative conversion of two carbon units into acetyl-CoA via b oxidation with concomitant generation of NADH and FADH2. This chapter!
Fatty Acid Oxidation in the Mitochondria stage 2
Stage 2 involves oxidation of acetyl-CoA into CO2 via citric acid cycle with concomitant generation NADH and FADH2. Chapter 16 (in Exam 3!)
The b-Oxidation Pathway
We first consider fully saturated, even-carbon containing fatty acids. Each pass (4 steps) removes one acetyl moiety in the form of acetyl-CoA and NADH and FADH2 - all of which are good sources of energy (ATP)!
What is steps 1-3 strategy?
The strategy of steps 1-3 is to oxidize the b -carbon to a carbonyl so that attacking it can release an acetyl CoA
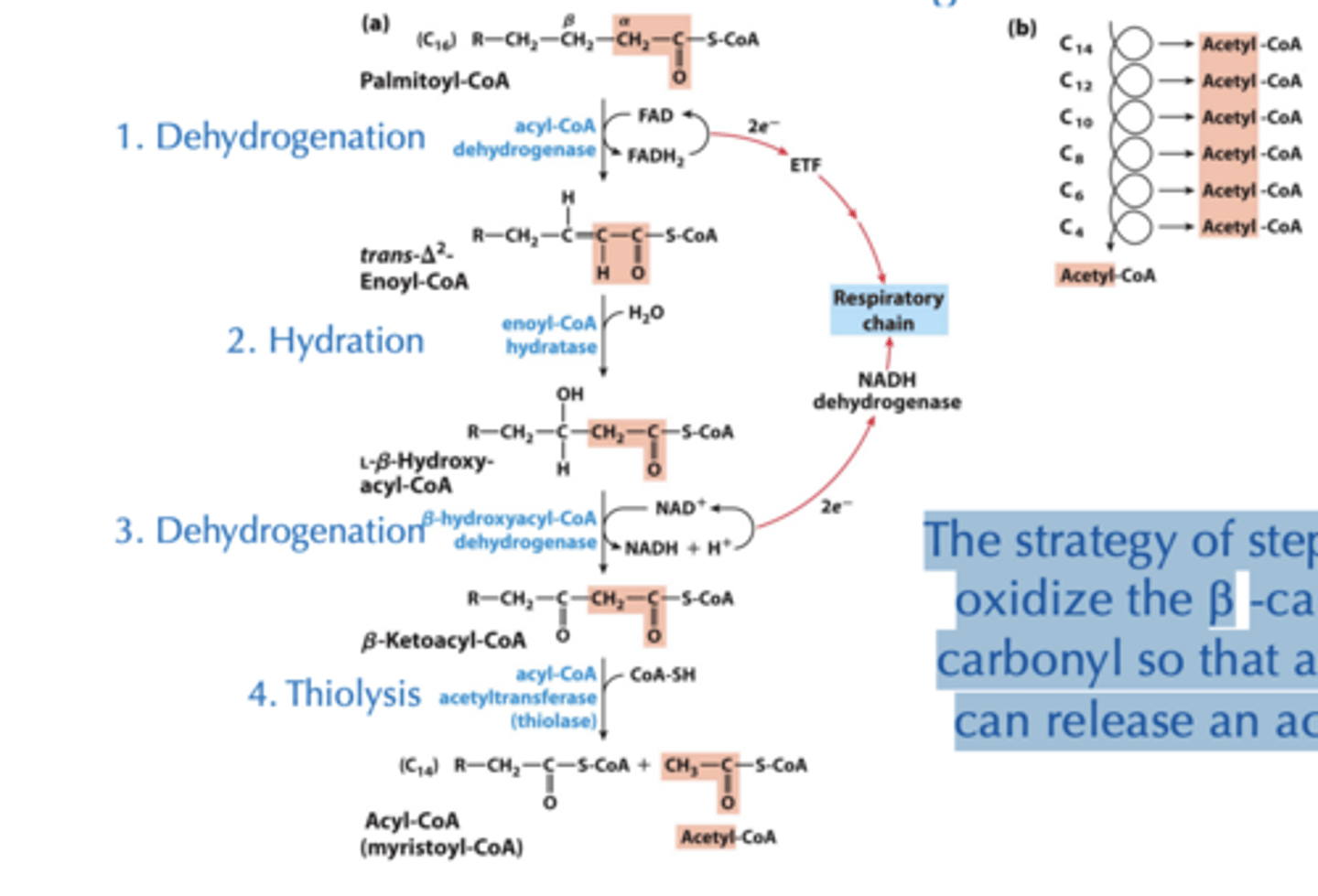
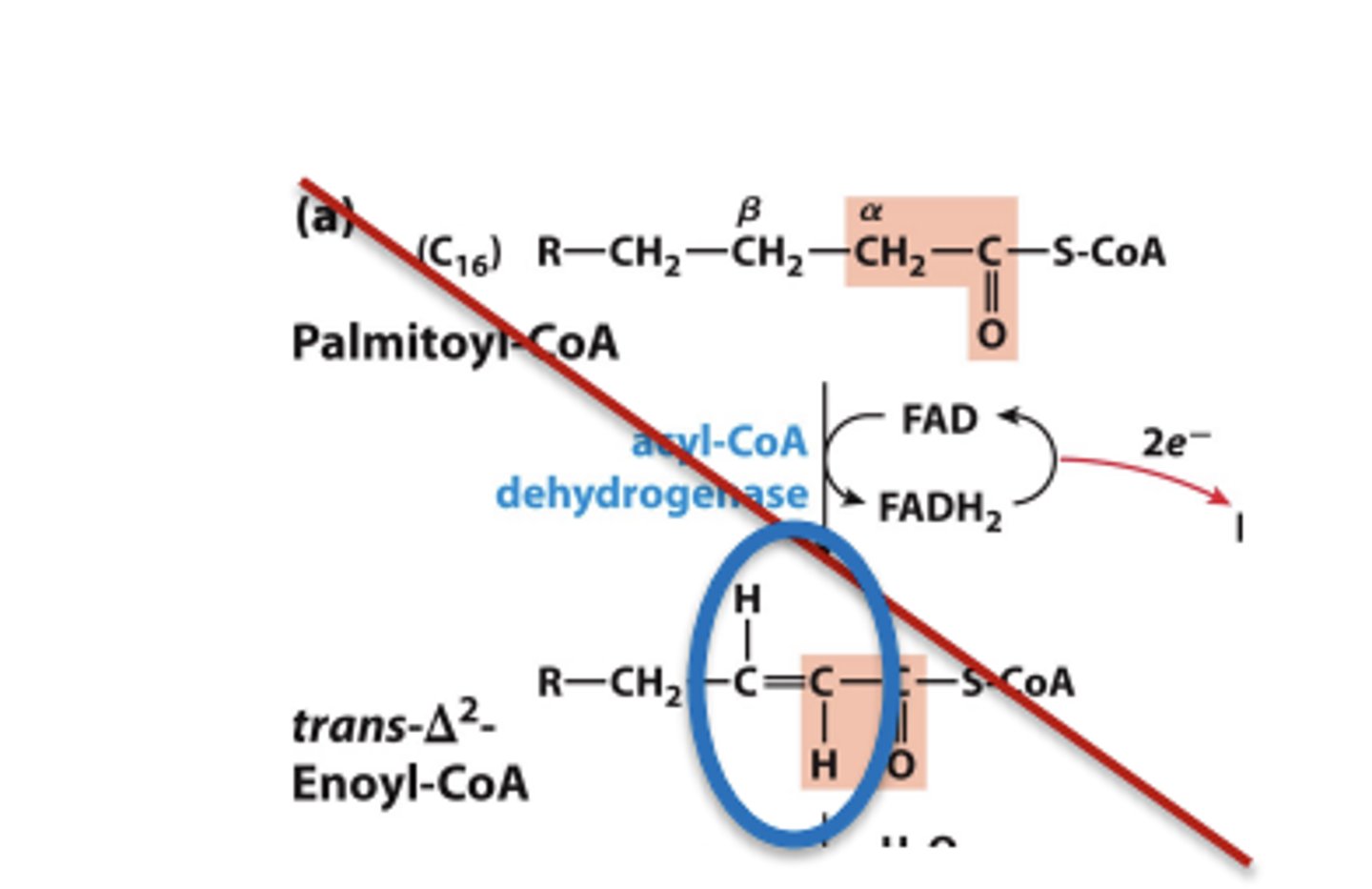
Step 1: Dehydrogenation of Alkane to Alkene
• Catalyzed by isoforms of acyl-CoA dehydrogenase (AD) on the inner-mitochondrial membrane
- long-chain AD (C12-C18)
- medium-chain AD (C4-C14)
- short-chain AD (C4-C8 carbons)
Step 1: Dehydrogenation of Alkane to Alkene results
• Results in trans double bond, different from naturally occurring unsaturated fatty acids
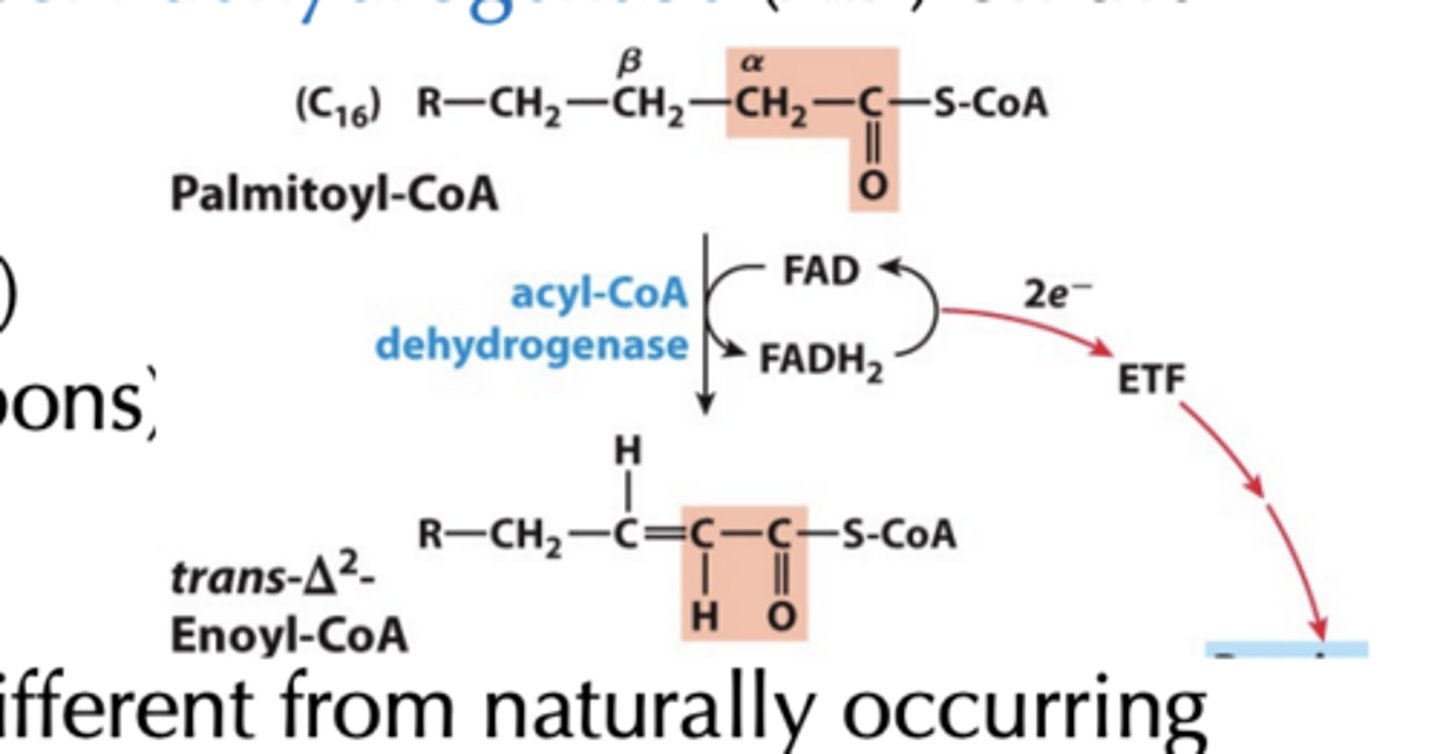
What is step 1 Analogous to
succinate dehydrogenase reaction in the citric acid cycle
- electrons from bound FAD transferred directly to the electrontransport chain via electron-transferring flavoprotein (ETF)
Step 2: Hydration of Alkene
• Catalyzed by enoyl-CoA hydratase
• Water adds across the double bond yielding alcohol on b carbon
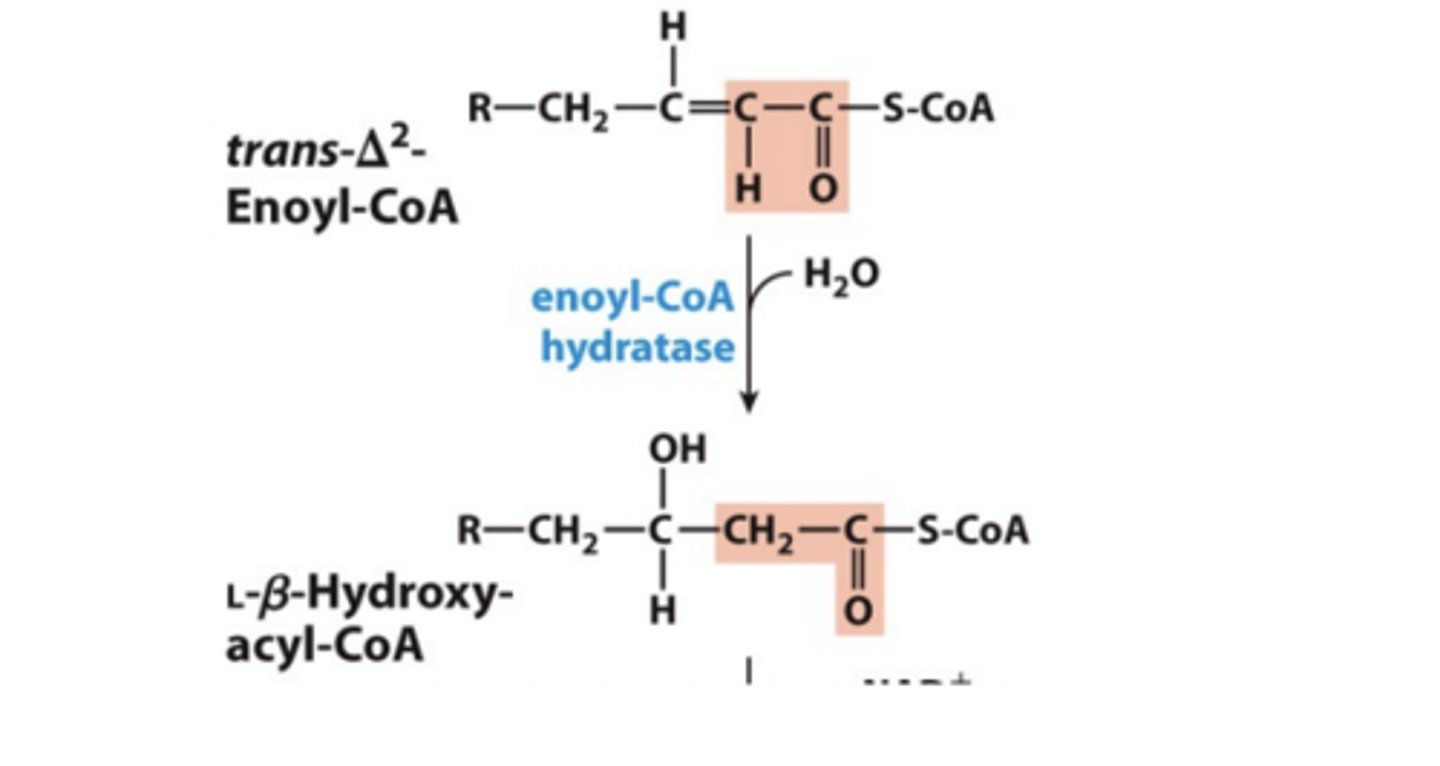
What is step 2 Analogous to
Analogous to fumarase reaction in the citric acid cycle
- same stereospecificity
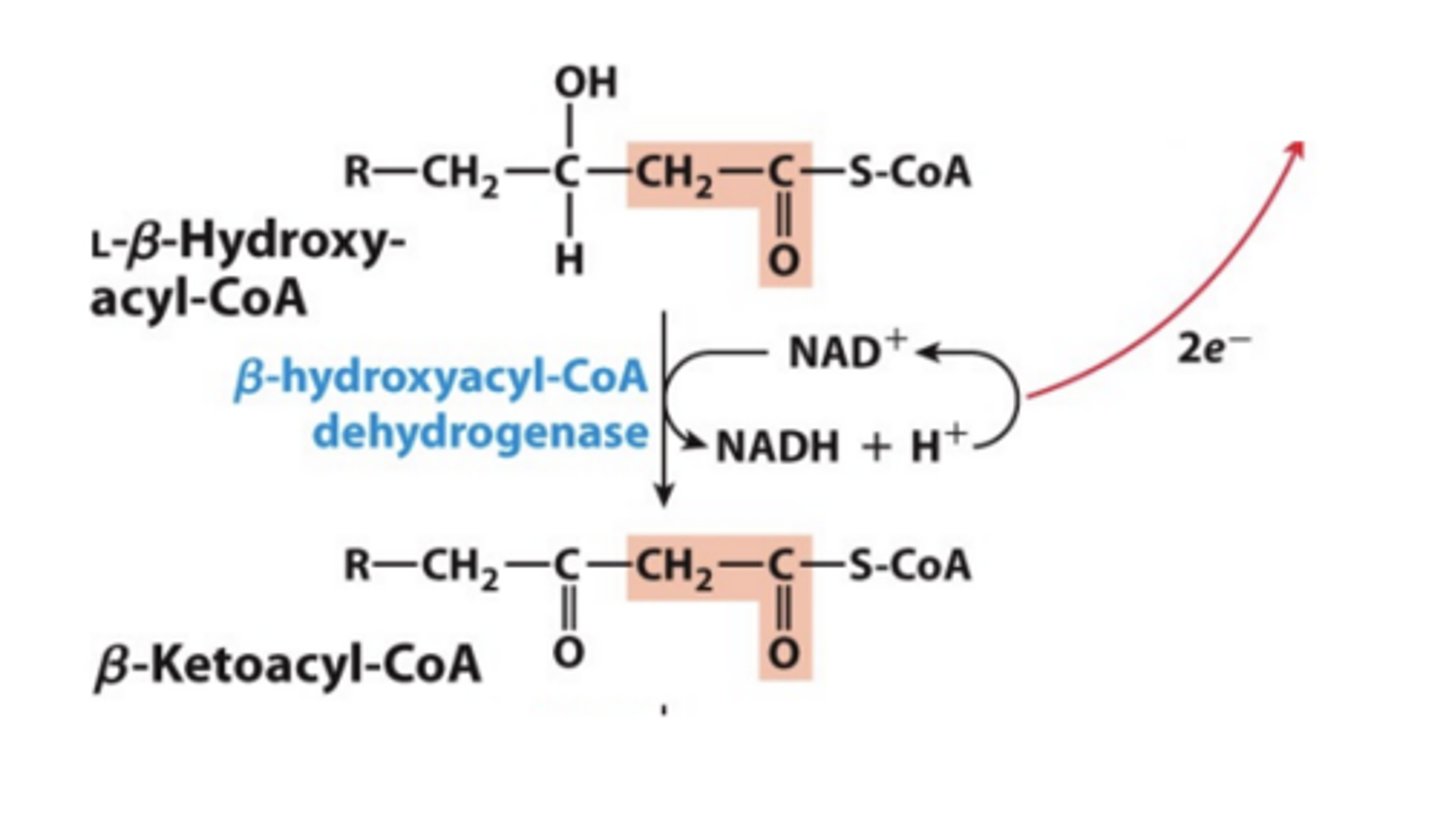
Step 3: Dehydrogenation of Alcohol
• Catalyzed by b-hydroxyacyl-CoA dehydrogenase
• The enzyme uses NAD+ cofactor as the hydride acceptor.
• Analogous to malate dehydrogenase reaction in the citric acid cycle
Similar Mechanisms Introduce Carbonyl on the β carbon
not tested on 3rd column
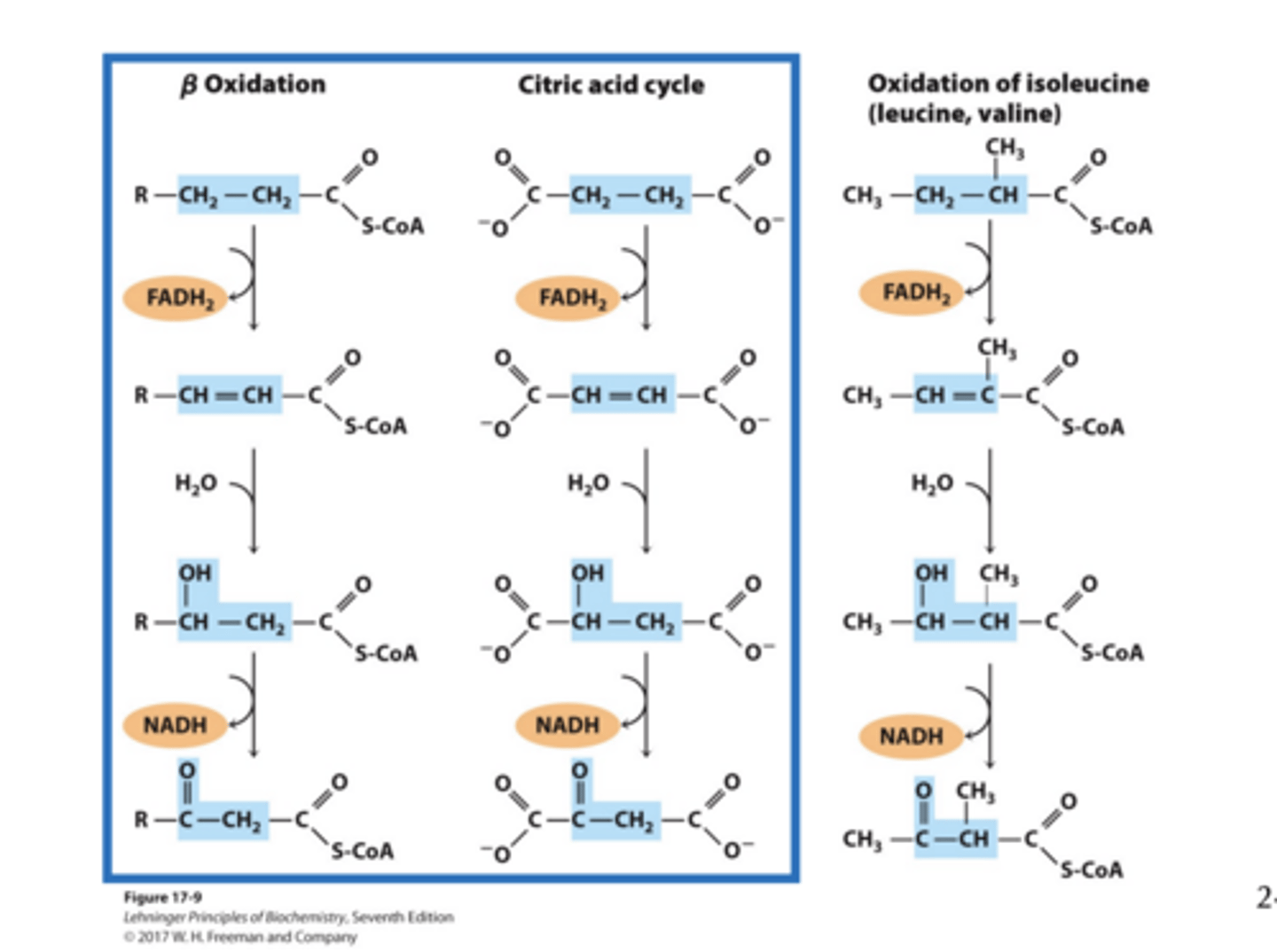
Step 4: Transfer of Fatty Acid Chain and Release of Acetyl-CoA
• Catalyzed by acyl-CoA acetyltransferase (thiolase)
- Thiol sulfur in CoA-SH acts as a nucleophile and picks up the fatty acid chain from the enzyme for next round of oxidation.
- The leaving group produces the acetyl CoA
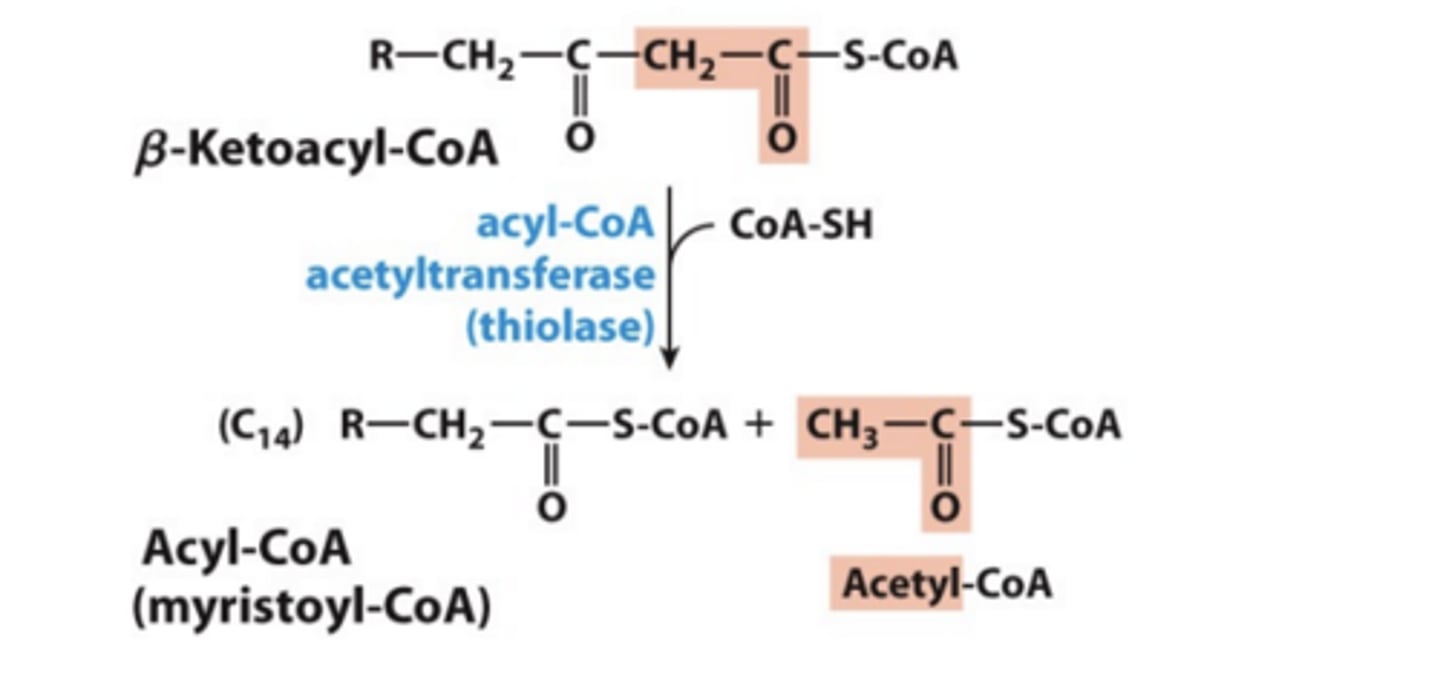
Step 4 net reaction?
thiolysis of the carbon-carbon bond
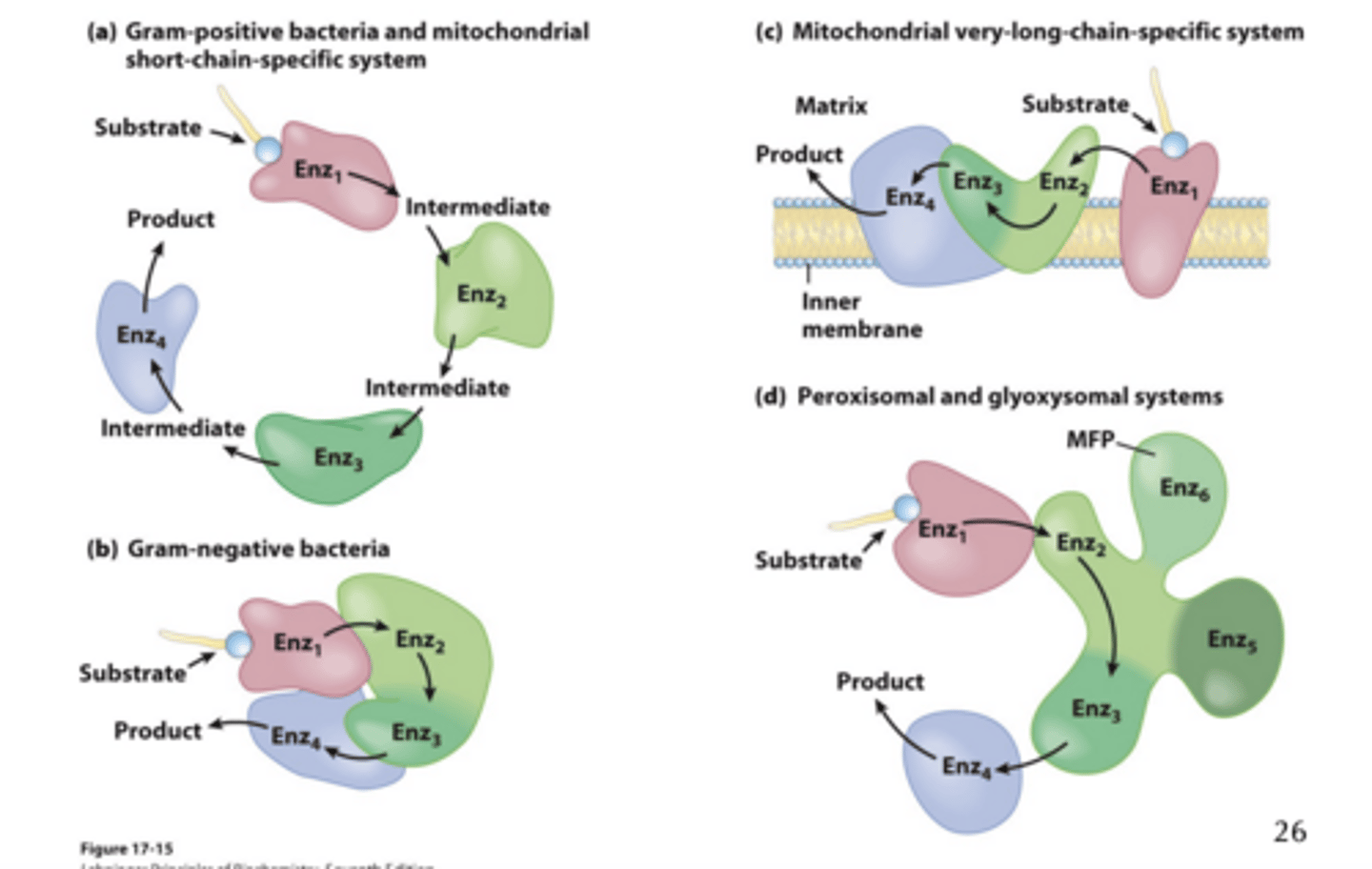
Fatty Acid Oxidation Is Performed by what?
a Single multifunctional Protein
* general dont memorize
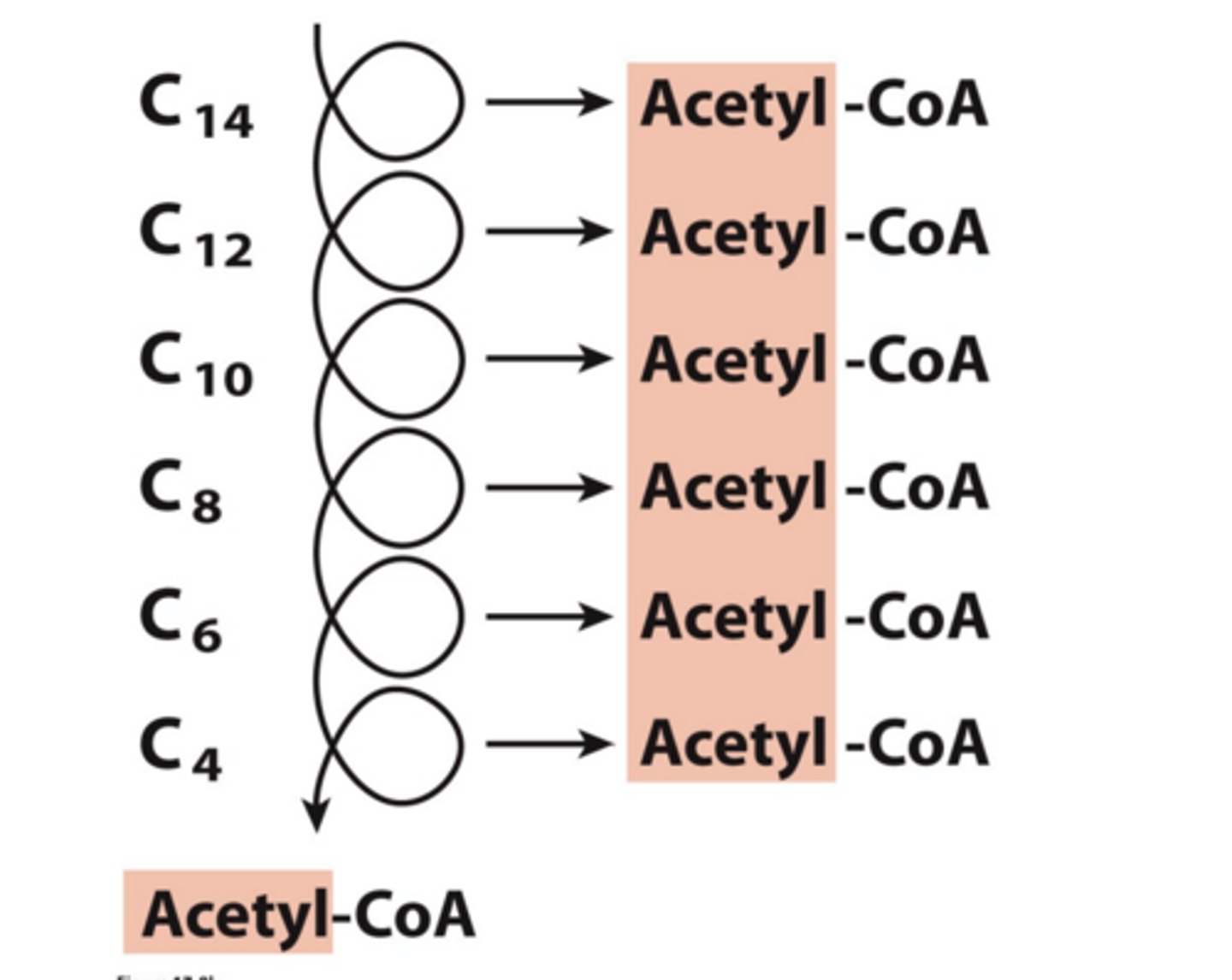
What does each Round Produce?
an Acetyl-CoA and Shortens the Chain by Two Carbons
Iclicker
E

Balance Sheet for b-oxidation of Palmitoyl-CoA (C16)? How many rounds for almitoyl-CoA ?
Palmitoyl-CoA (C16) + 7 CoA + 7 FAD + 7 NAD+ + 7 H20 -> 8 Acetyl-CoA + 7 FADH2 + 7 NADH + 7 H+

With added Oxidative phosphorylation how much energy is produce from the Balance Sheet for b-oxidation
28 ATP
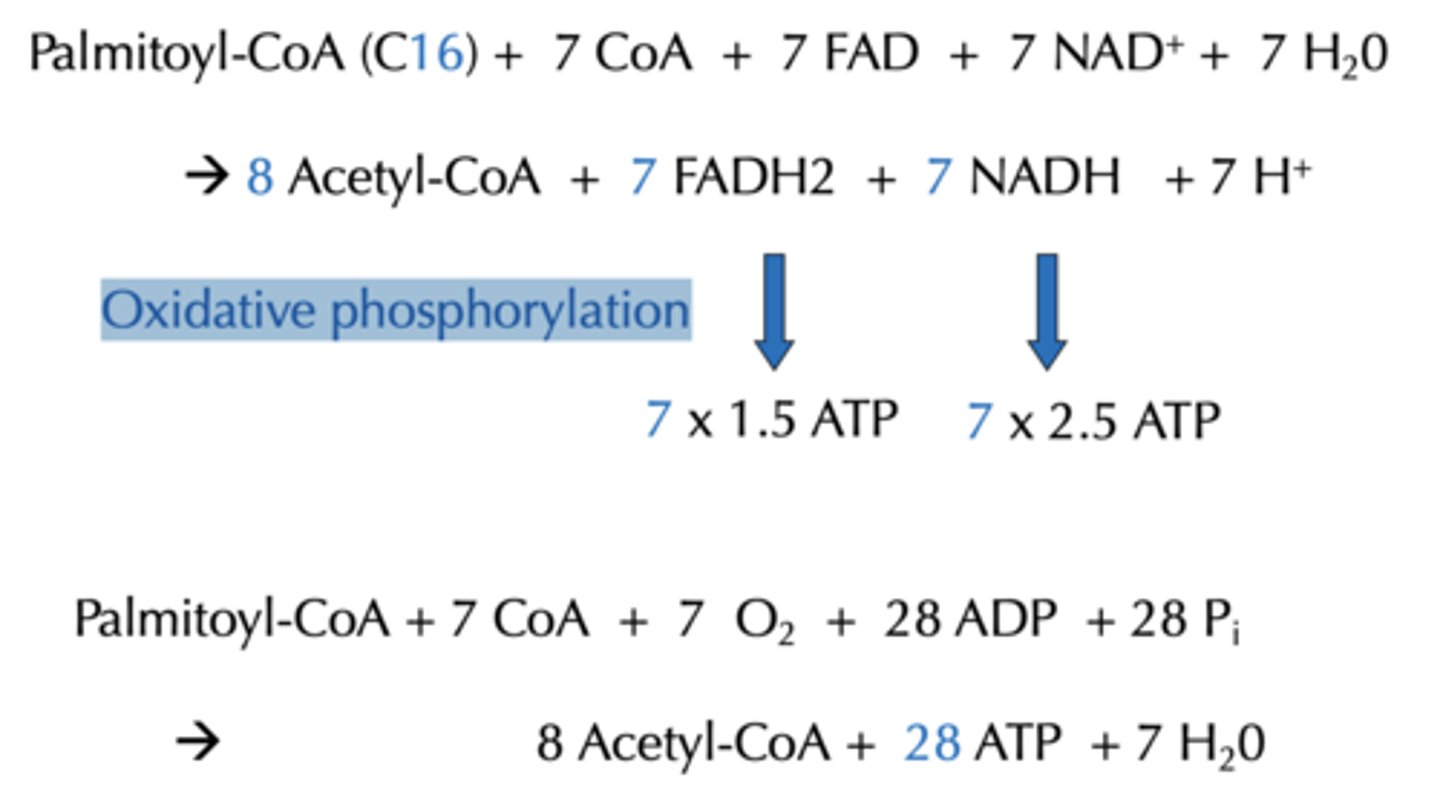
With added Oxidative phosphorylation and citric acid cycle, how much energy is produce from the Balance Sheet for b-oxidation from Acetyl-CoA?
80 ATP
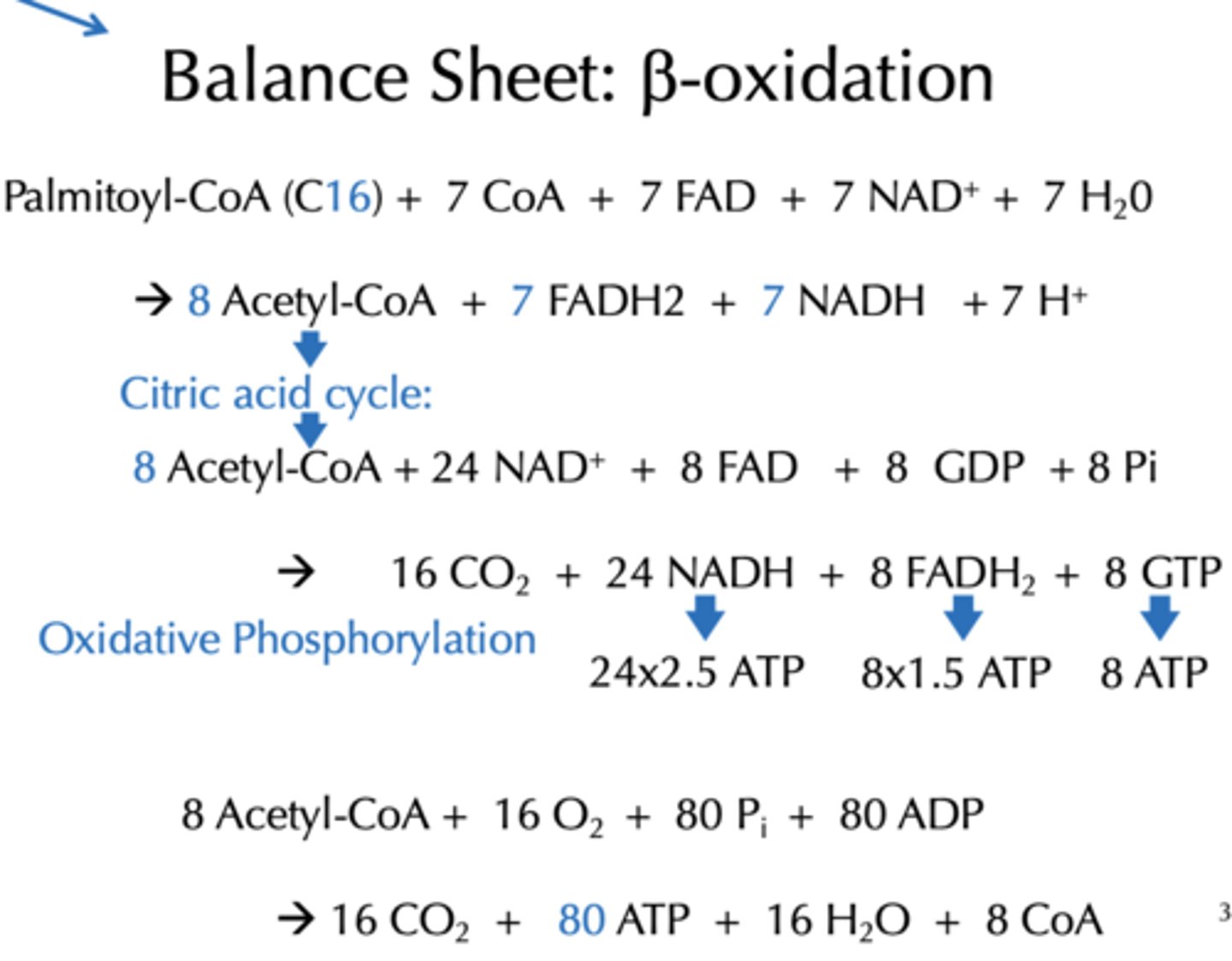
With added Oxidative phosphorylation and citric acid cycle, how much energy is produce from the Balance Sheet for b-oxidation from Palmitoyl-CoA (C16)?
108 ATP
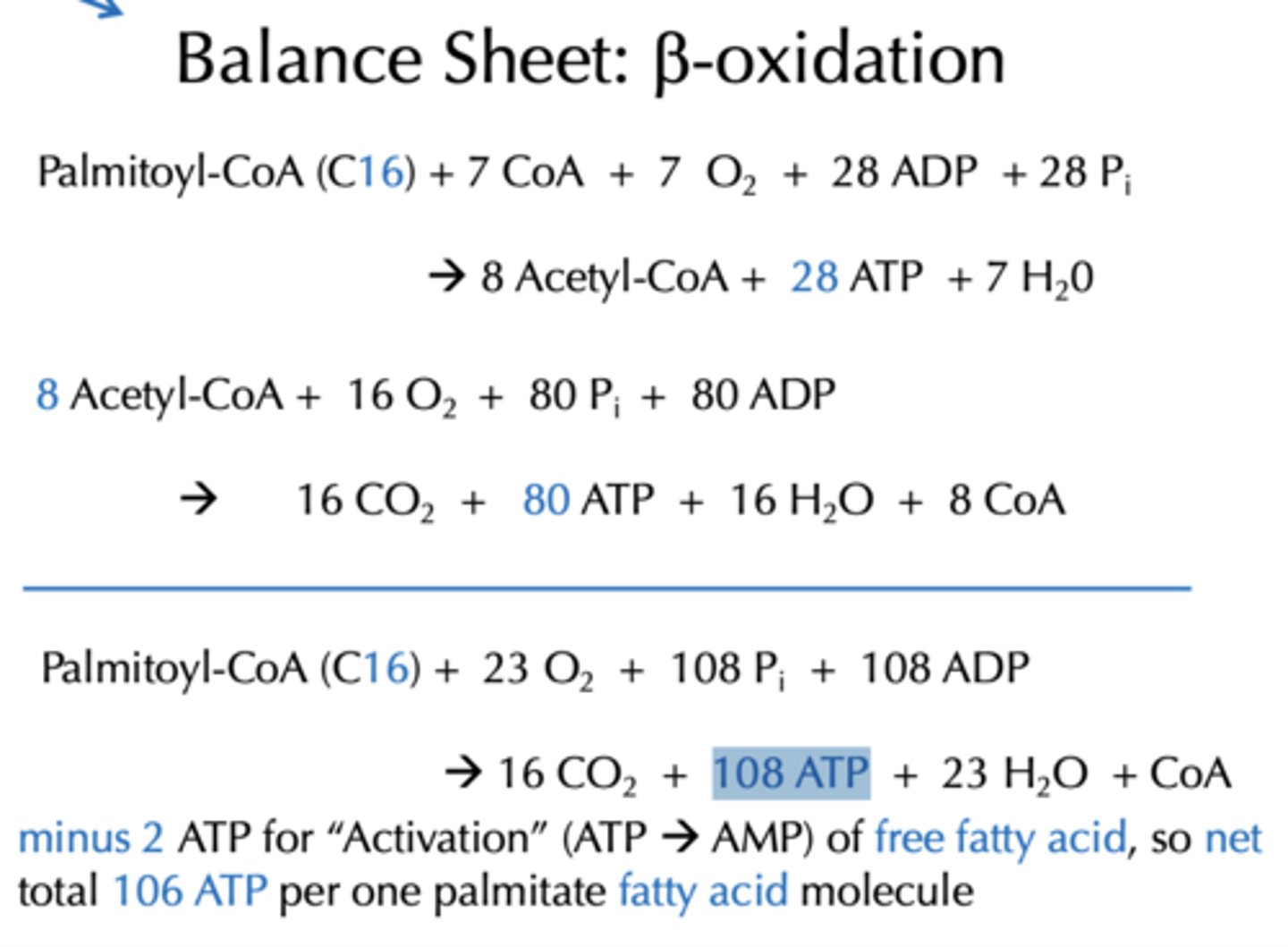
If the question is just concerning ATP, which more simple method can be used?

Example: Myristate = 14 carbons -> 7 Acetyl-CoA, 6 NADH and 6 FADH2, what is Total ATP potential/yield upon full oxidation?
94 ATP from fatty acyl CoA (92 ATP if starting with free fatty acid)

What do Naturally occurring unsaturated fatty acids contain?
cis double bonds.
- are NOT a substrate for enoyl-CoA hydratase
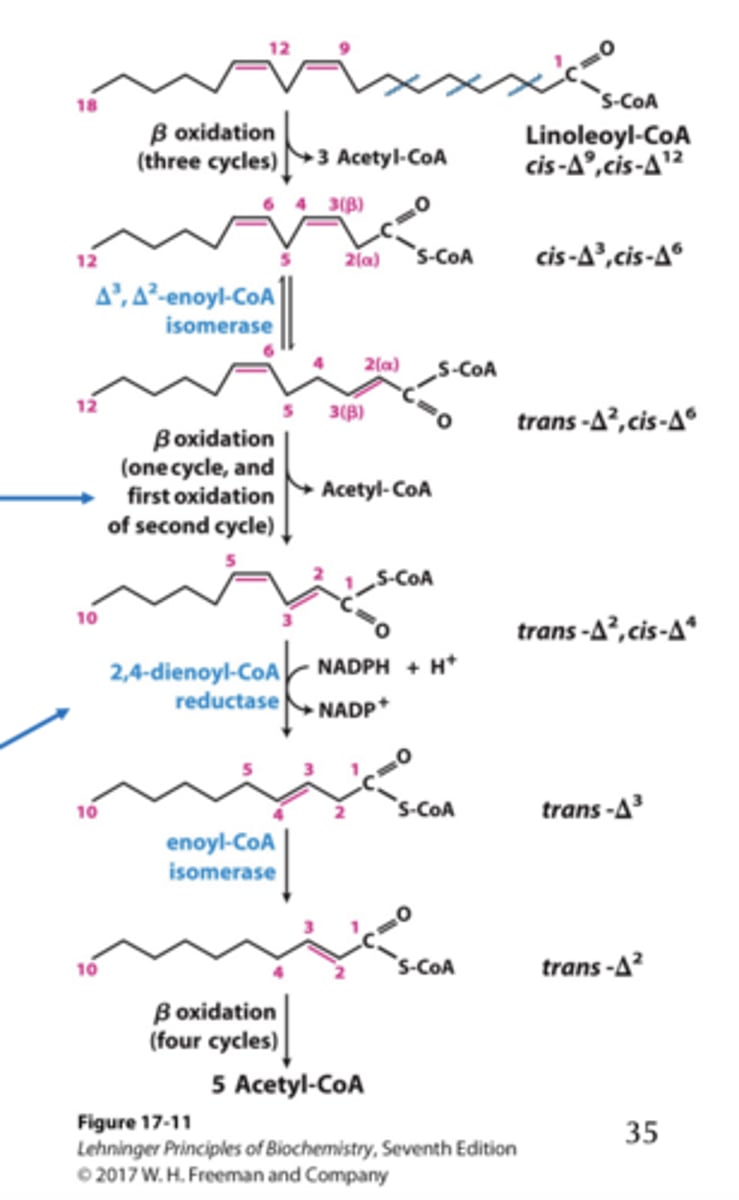
Two additional enzymes required for Oxidation of Unsaturated Fatty Acids
isomerase: converts cis double bonds starting at C3 to trans double bonds reductase: reduces cis double bonds not at C3
What does Monounsaturated fatty acids use compared to Polyunsaturated
• Monounsaturated fatty acids require only isomerase.
• Polyunsaturated fatty acids require both enzymes.
Oxidation of Monounsaturated Fatty Acids mechanism
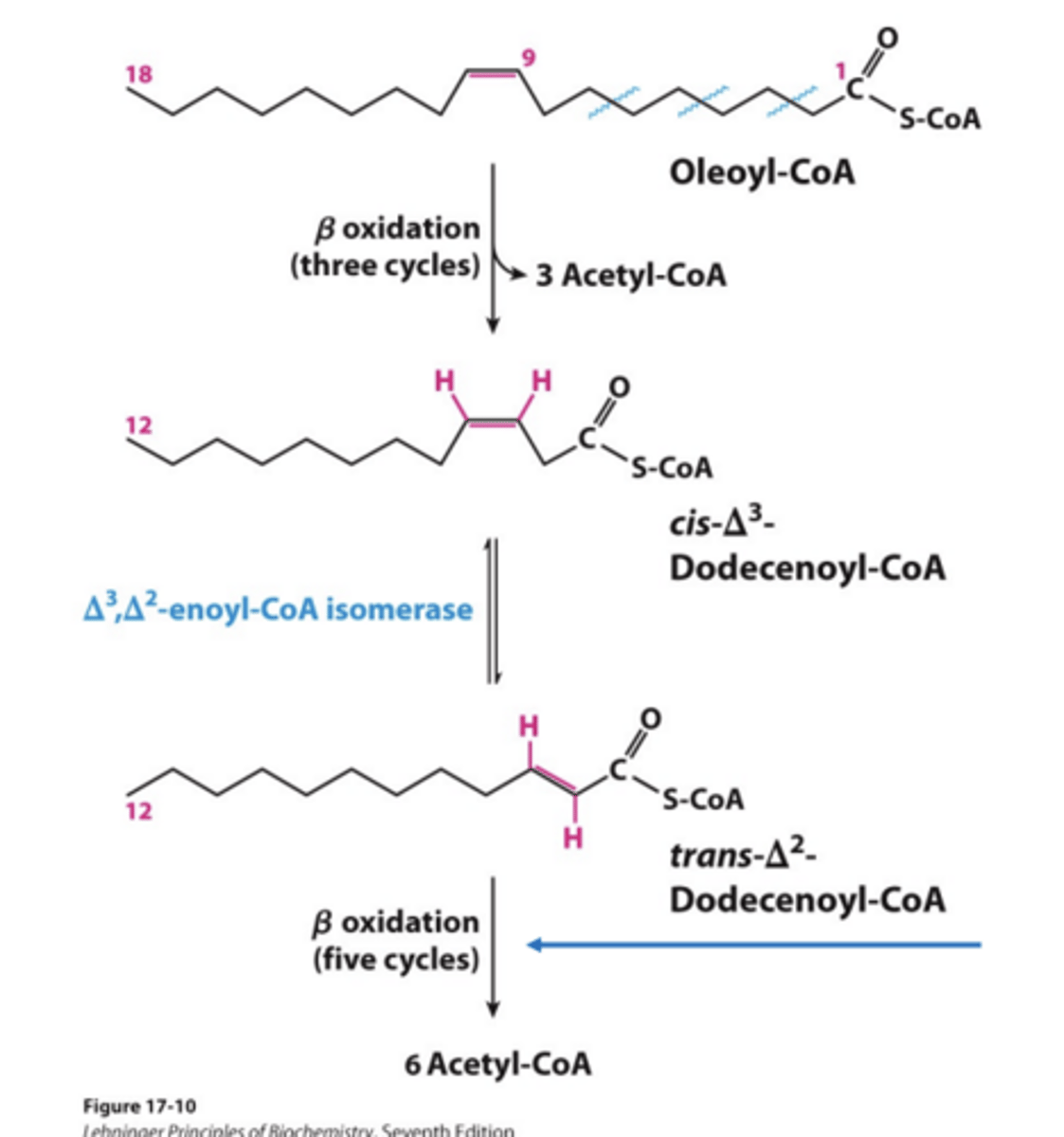
During first of the remaining cycles, what is skipped, what does this result in?
acylCoA dehydrogenase step is skipped, resulting in 1 fewer FADH2.
Oxidation of Polyunsaturated Fatty Acids results in what change for FADH2 ?
Results in 1 fewer FADH 2 after isomerization (4th cycle; C12 -C10), but 1 FADH 2 is produced during the first step of the next cycle (5th cycle; C10 -C8).
(top arrow)
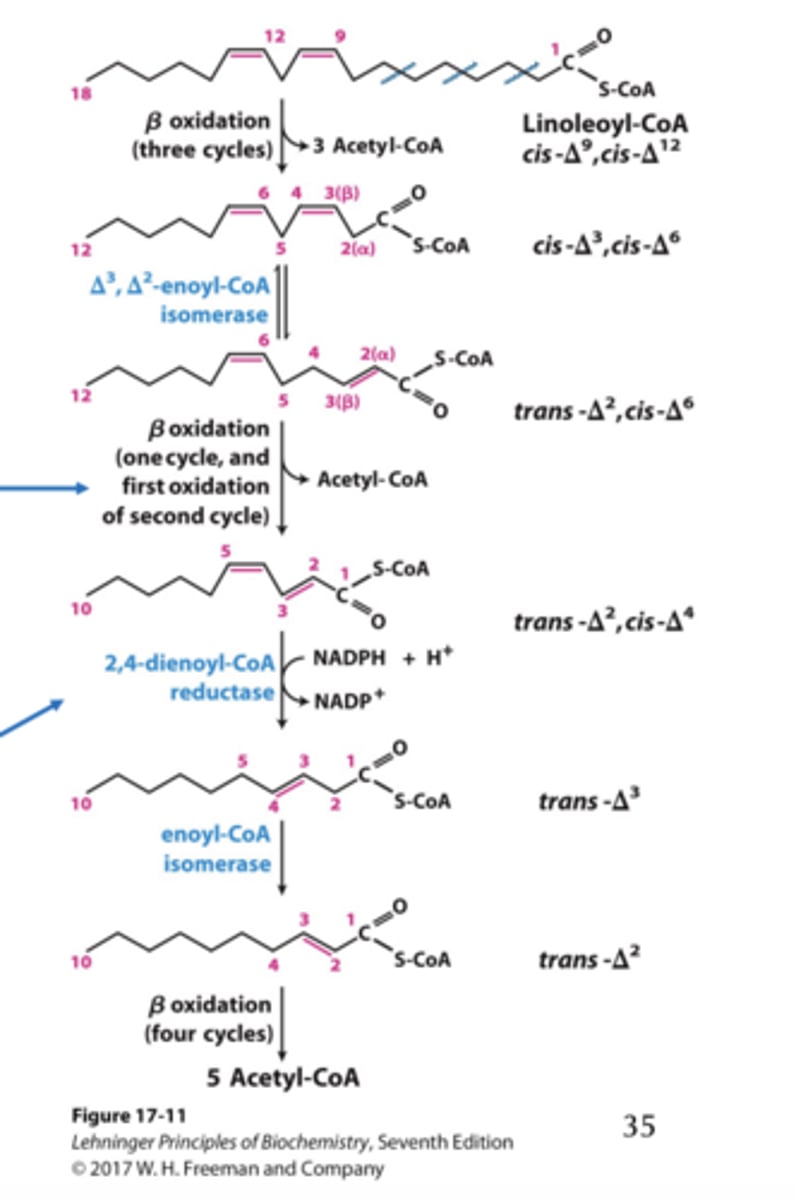
Oxidation of Polyunsaturated Fatty Acids results in what change for NADPH?
NADPH reduces the remaining unsaturated bond, resulting in no further loss of FADH 2 . (bottom arrow)
- ATP calculation is the same as monounsaturated
Oxidation of Odd-Numbered Fatty Acids in dietary fatty acids.
Most dietary fatty acids are even-numbered.
- Many plants and some marine organisms also synthesize odd-numbered fatty acids.
When does Propionyl-CoA (3-carbon compound) form?
- during final cycle of b oxidation of odd-numbered fatty acids.
- Bacterial metabolism in the rumen of ruminants also produces propionyl-CoA.
Iclicker
A

Regulation of fatty acid metabolism
• Acetyl CoA carboxylase (ACC) phosphorylation/dephosphorylation is critical
• Follows the phosphorylation rule of Chapter 15
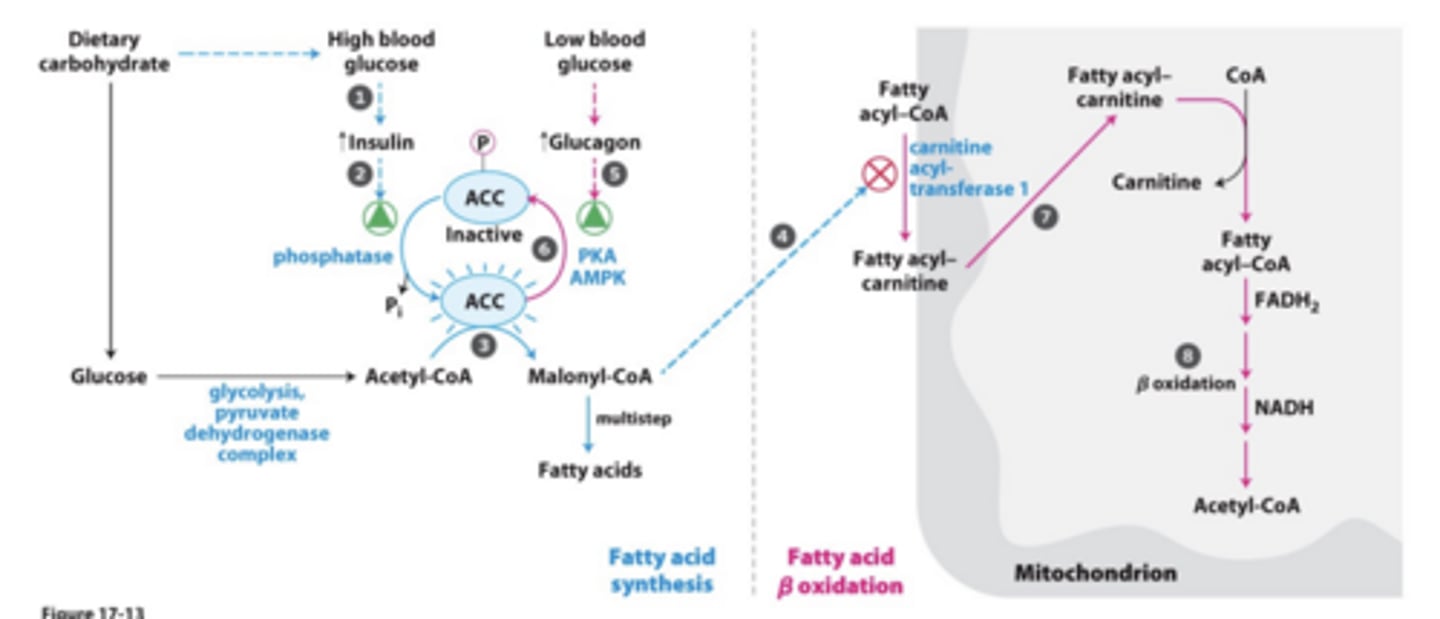
Malonyl CoA role in regulation?
Reciprocal regulation of fatty acid synthesis and breakdown
- if your are making fatty acids in the cytosol you don't also want to oxidize them
b Oxidation in Mitochondria versus Peroxisomes or Glyoxysomes?
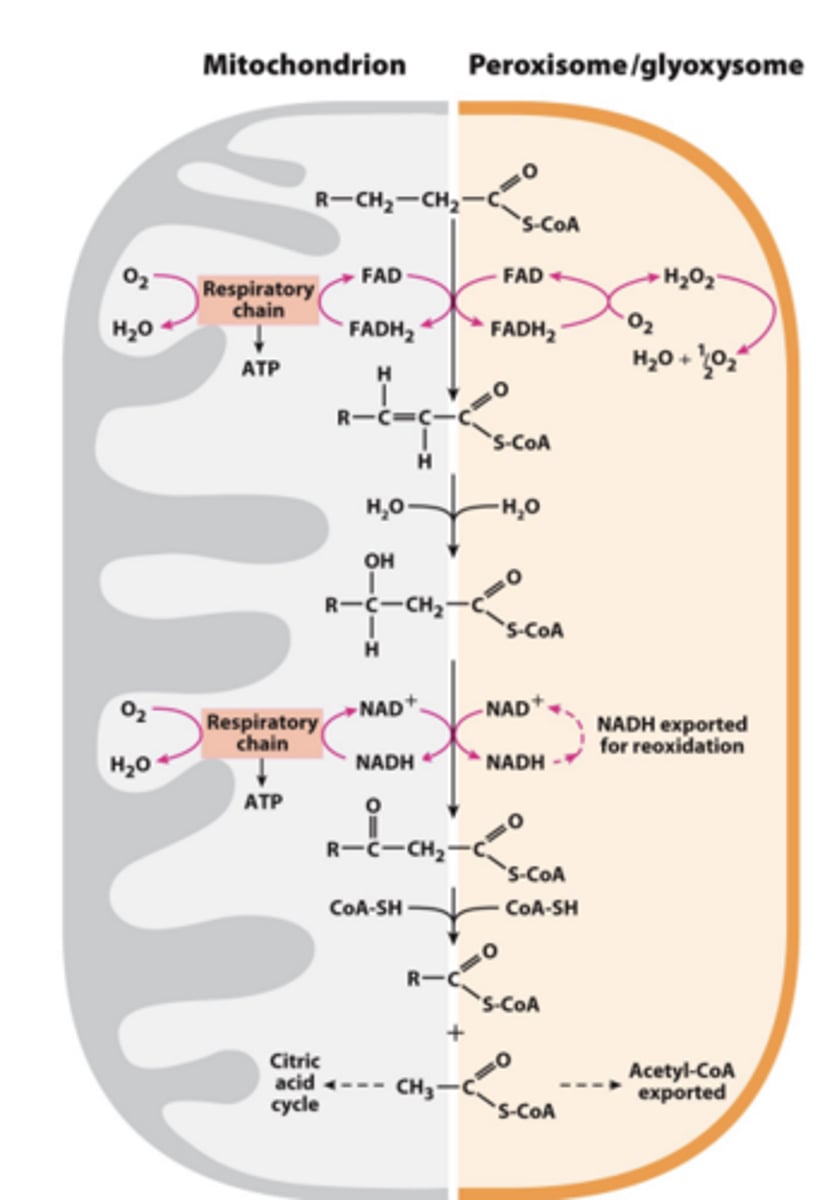
What is a peroxisome when enzymes for glyoxylate cycle are present?
also a glyoxysome
What is Acetyl -CoA released from peroxisomal b oxidation, be used for?
Acetyl -CoA released from peroxisomal b oxidation can be used in glyoxylate cycle or exported to cytosol and then imported to mitochondria
Entry of acetyl-CoA into citric acid cycle requires what?
oxaloacetate
When oxaloacetate is depleted (excessive gluconeogenesis), what is excess acetyl-CoA converted into
ketone bodies
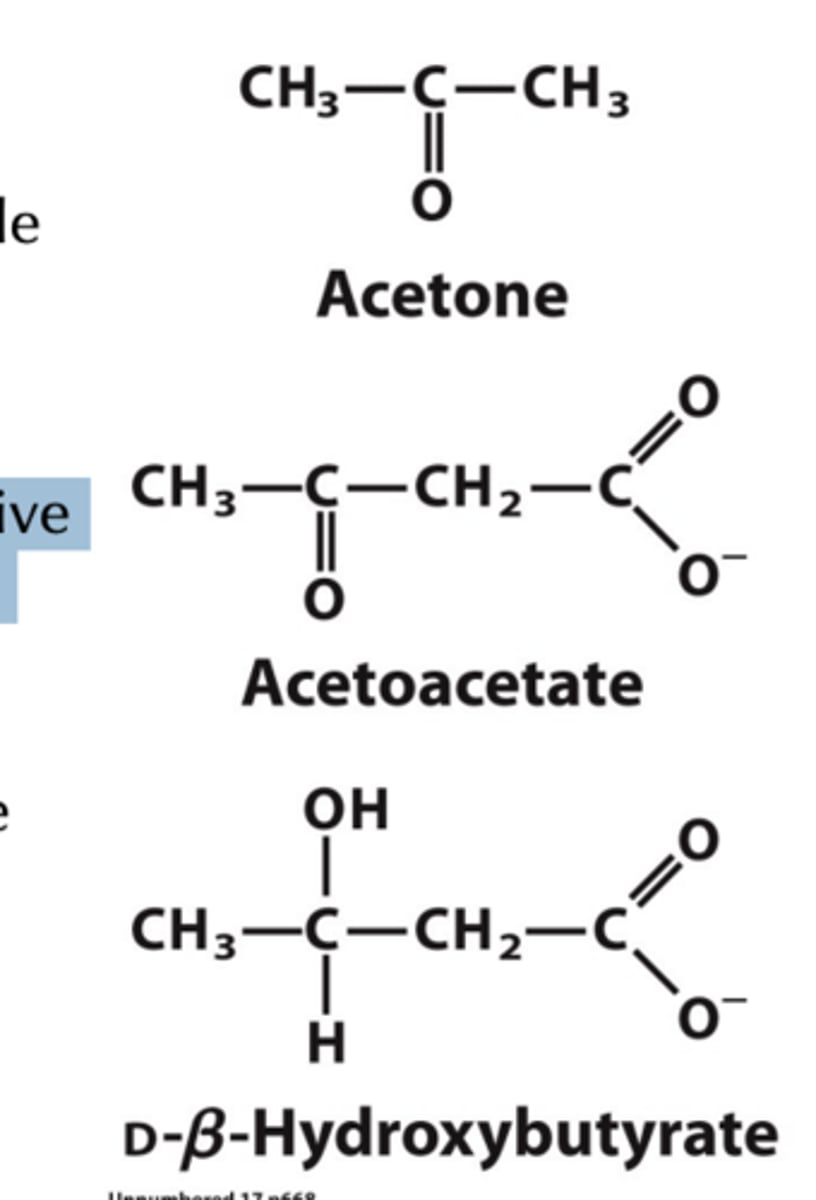
Three forms of ketone bodies can leave the liver
acetone
acetoacetate
bhydroxybutyrate
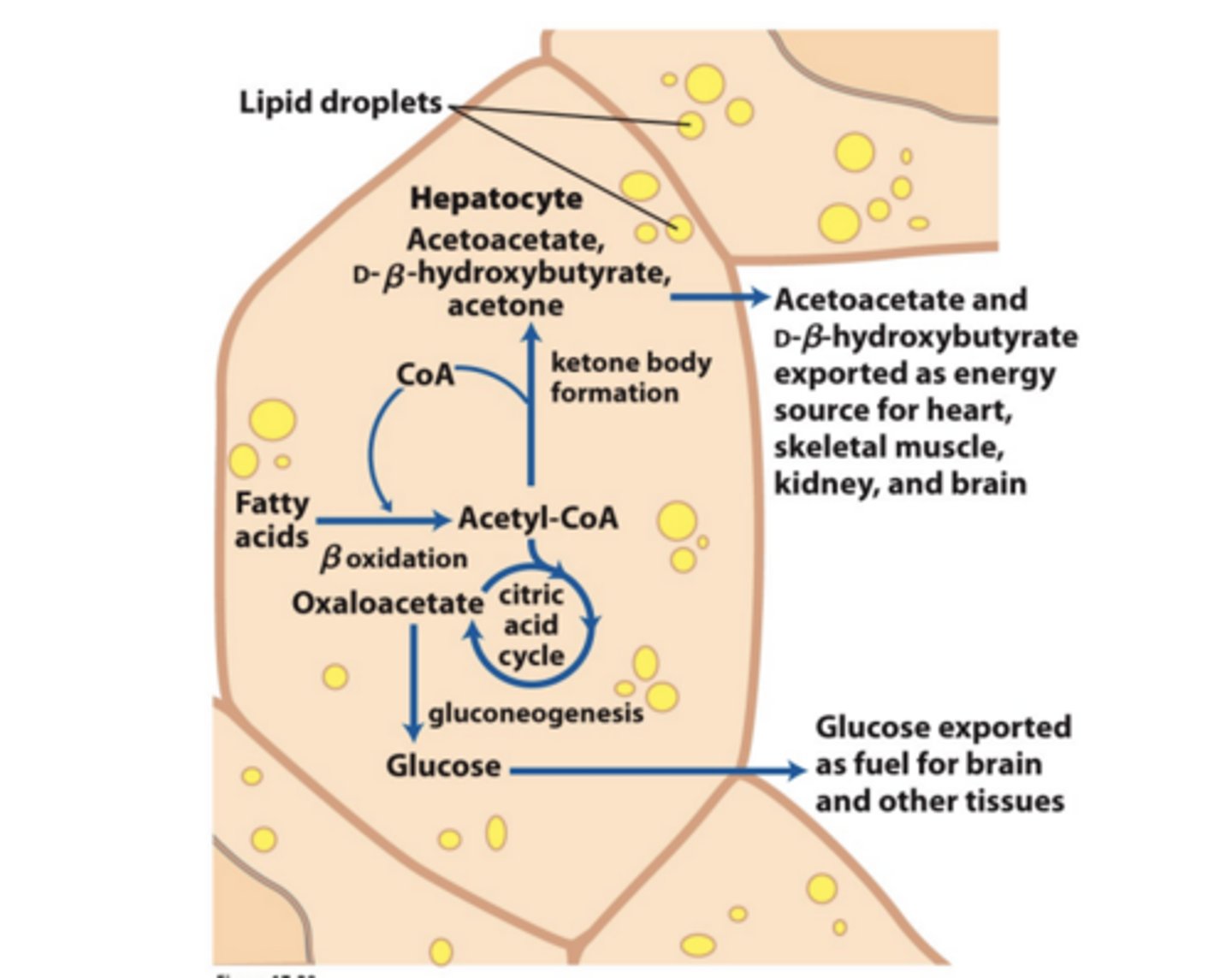
What is the source of ketone bodies? What is this process?
The Liver
Formation of Ketone Bodies through Generating Free CoA first and second step?
• The first step is reverse of the last step in the b oxidation: thiolase reaction joins two acetate units.
• A third acetyl-CoA is incorporated in the second step.
overal reaction of the Formation of Ketone Bodies via Generating Free CoA
Together, two CoA are freed from three acetyl-CoA.
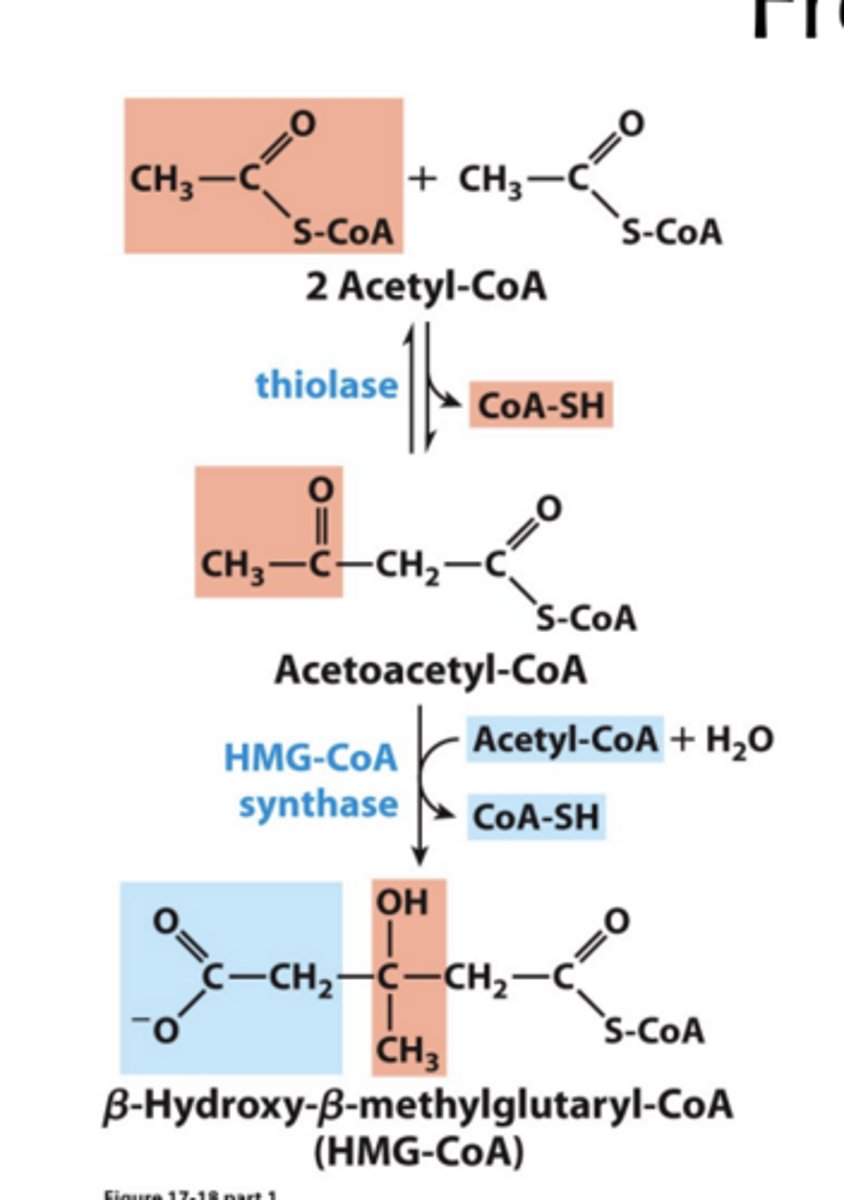
Formation of Ketone Bodies via Degradation of HMG-CoA
In order to traffic to other tissues, CoA must be removed. Acetone, acetoacetate, and b-hydroxybutyrate can then travel through the blood.
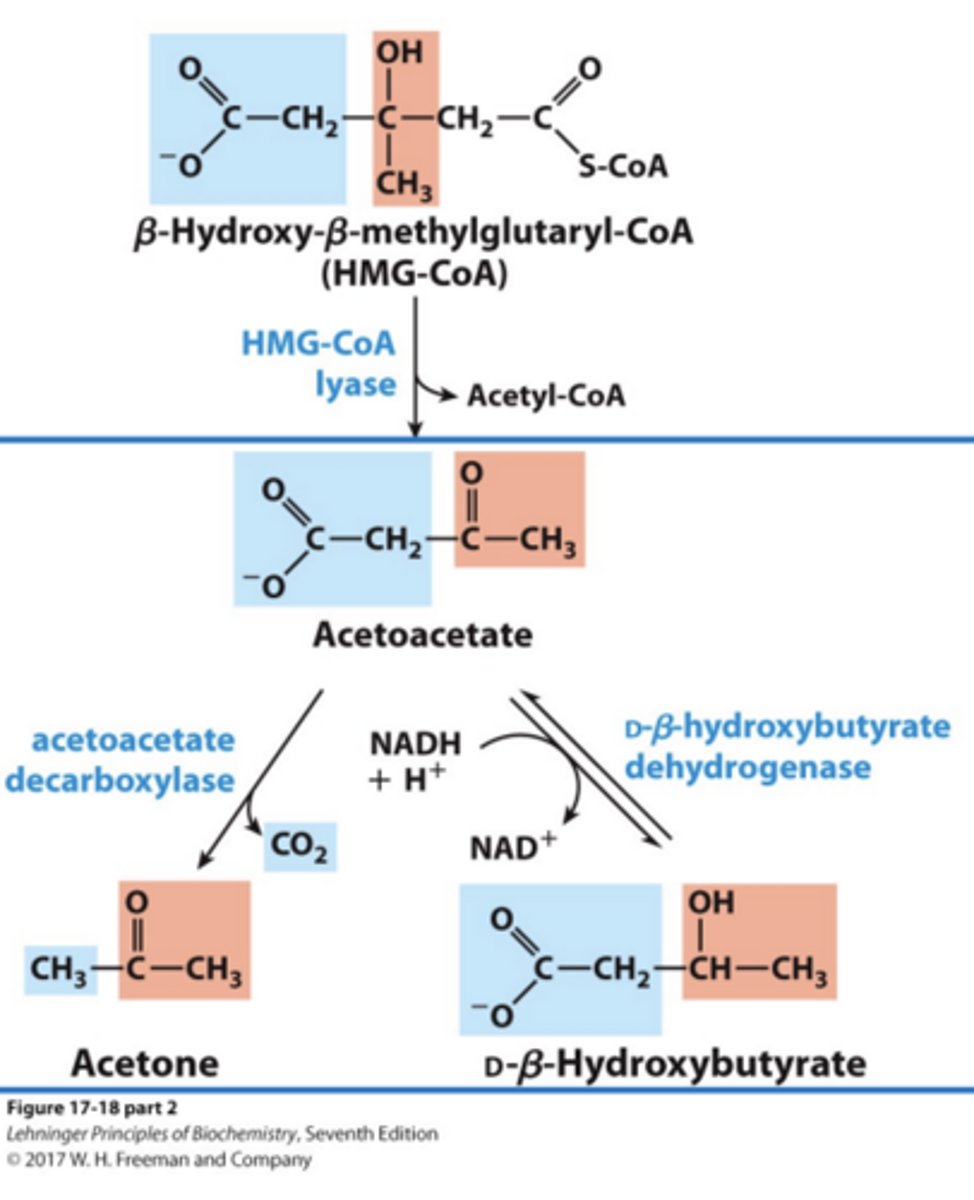
Within Formation of Ketone Bodies via Degradation of HMG-CoA what occurs to Acetone, acetoacetate, and d b-hydroxybutyrate?
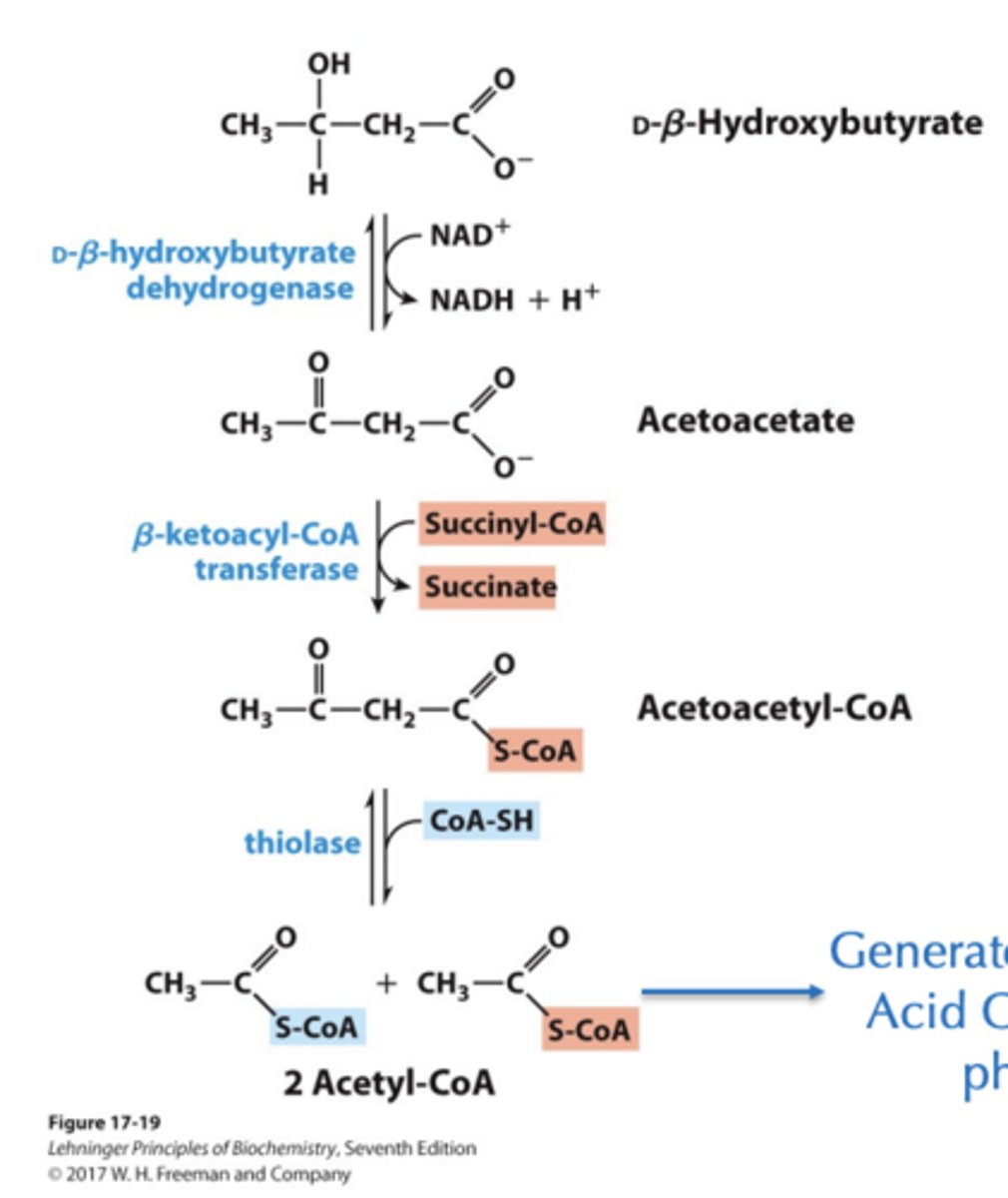
Acetone is removed as a gas and exhaled, but acetoacetate and b-hydroxybutyrate can traffic to the brain for use in energy production
Ketone Bodies as Fuel? Where does the energy come from?
Generate energy using Citric Acid Cycle and oxidative phosphorylation
Type 1 vs type 2 diabetes
Type I diabetes Insulin absent
Type II diabetes Insulin resistant → insulin produced but cells don't respond to it
Consequences of Diabetes
• High blood glucose level → Low glucose inside cells
• Excess glucose circulating in blood could result in excessive protein glycosylation → damage blood vessels and various tissues and organs (e.g., eye, kidney, nerves)
• Dehydration (as kidneys try to flush out excess glucose)
• Fatigue (as cells are starved of glucose - fuel for instant energy)
• Risk of infection (high glucose is food for microbes!)
Diabetes Diagnosis Fasting Plasma Glucose (FPG) Test
Normal: <100 mg/dL (5.6 mM) Measures ability to maintain normal glucose level in short-term
Diabetes Diagnosis Hemoglobin A1c (HbA1c) Tes
Normal: <5.7%
• High blood glucose leads to excessive glycation of proteins, including Hb
• RBCs lifespan 2-3 months. Measures long-term glucose levels
Diabetes Therapy Diet & exercise
Less calories in, less glucose in blood Exercise burns calories directly but also activates AMPK à increases catabolism
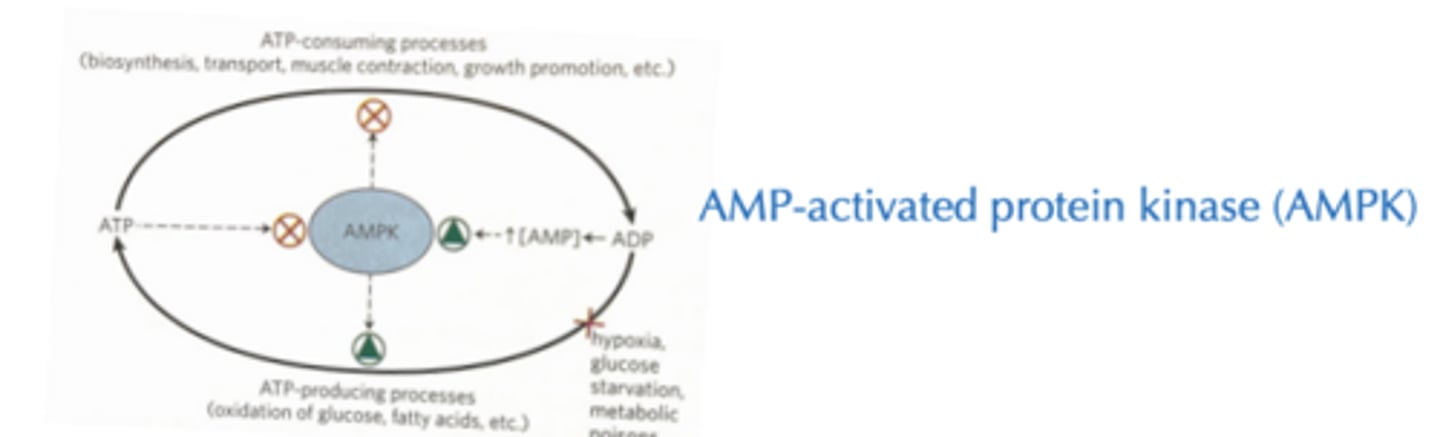
Diabetes Therapy Metformin and Insulin therapy
Metformin
- increases AMP to activate AMPK
Insulin therapy (effective for type I, less so for type II)
- Insulin injection → returns glucose levels in blood (and cells) to norma
Diabetes Therapy GLP-1 Receptor Agonist and SGLT-2 Inhibitors
GLP-1 Receptor Agonists (e.g., Semaglutide, Tirzepatide)
- Increase insulin secretion and reduce appetite
SGLT-2 Inhibitors Promote glucose - excretion in urine
What is the major Energy Source in Many Organisms? Evidence?
Oxidation of Fatty Acids
• About one-third of our energy needs comes from dietary triacylglycerols.
• About 80% of energy needs of mammalian heart and liver are met by oxidation of fatty acids.
• Many hibernating animals, such as grizzly bears, rely almost exclusively on fats as their source of energy.
Many hibernating animals, such as grizzly bears, rely almost exclusively on fats as their source of energy.
• Needs 6000 kcal/mol per day (avg. active human needs 2000 kcal/mol)
• Does not eat (or drink, urinate, defecate, etc)
• Fat stores provide all of the energy to keep the cells "running" and maintain the body temperature for months!
The advantage of fats over polysaccharides
- Fatty acids carry more energy per carbon because they are more reduced.
- Fatty acids are less hydrated because they are nonpolar - more compact, efficient storage.
What do Fats Provide
Efficient Storage and Energy Production
So why was glucose/carbohydrates retained in evolution as the "go to" energy molecule/s in the cell?
Glucose/Glycogen are for immediate (today! now!) energy needs
- quick storage, transport, and breakdown.
Good balance of solubility and reduced nature
What is fat storage good for?
Fats are for long-term (months) energy needs
- slow storage, transport, and breakdown. Highly reduced but poorly soluble
Where are Dietary Fatty Acids absorbed
Dietary Fatty Acids Are Absorbed in the Vertebrate Small Intestine
How are lipids in diet taken up by cells for storage OR energy?
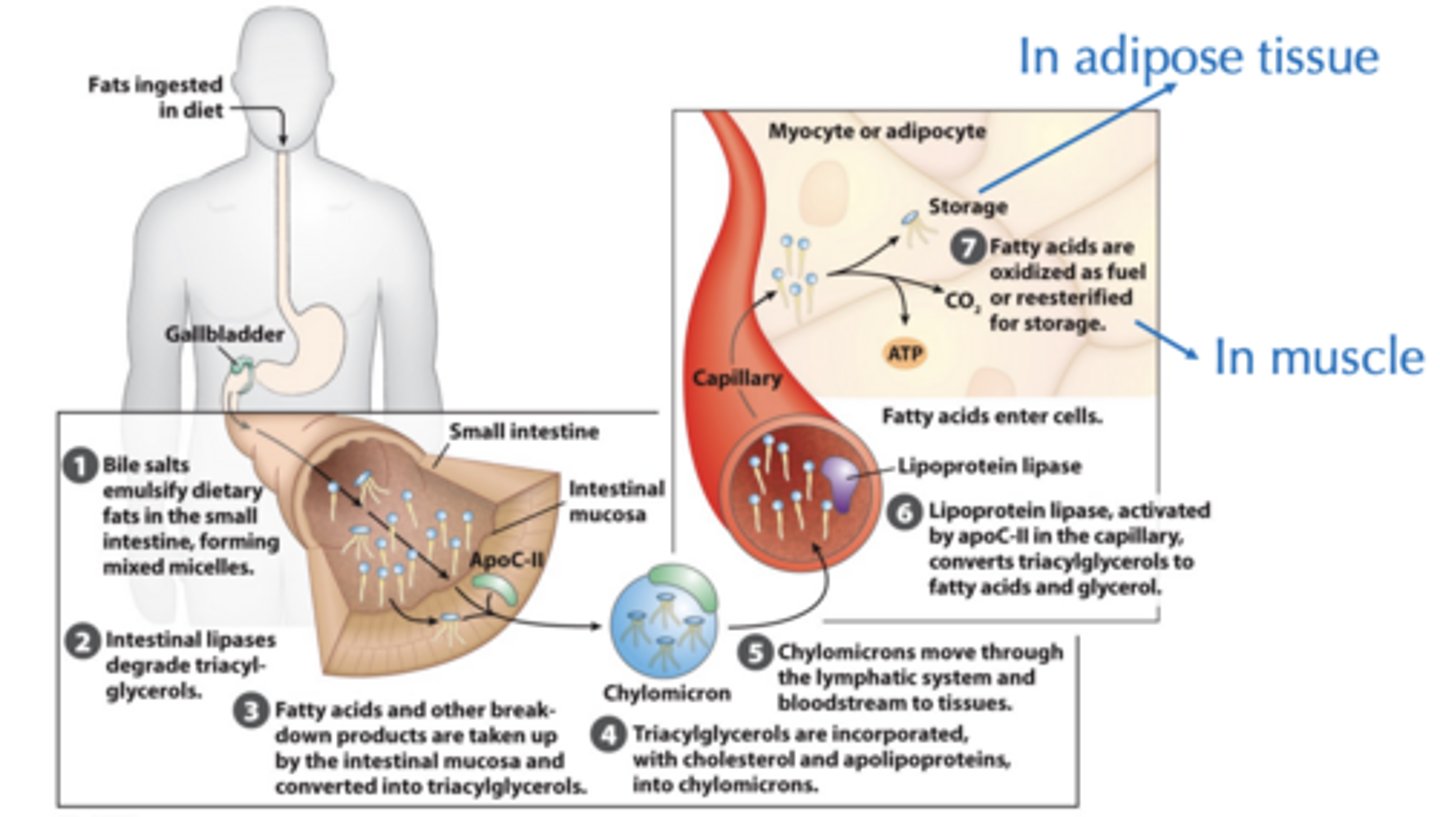
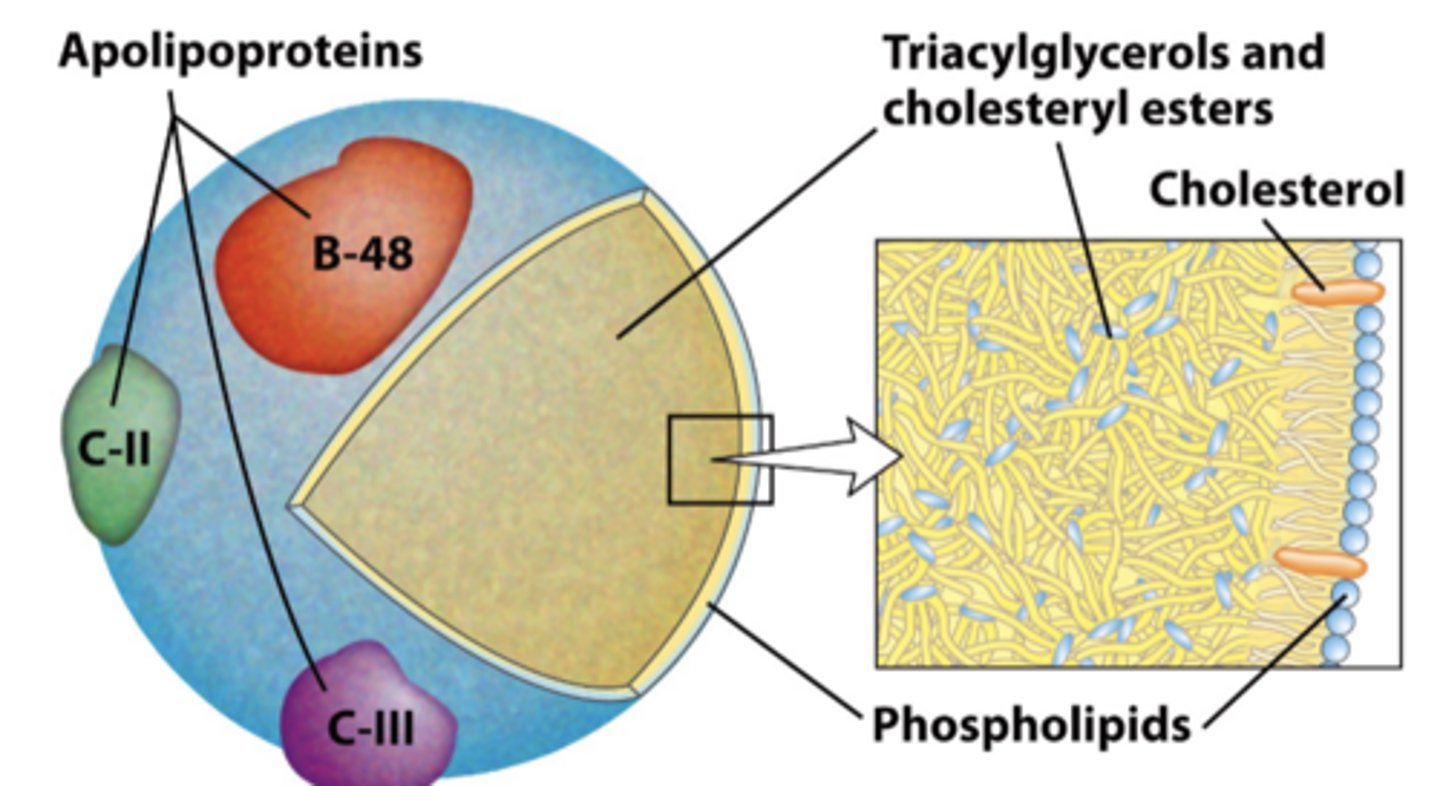
What are Lipids Are Transported in the Blood as? Types present?
Chylomicrons