Freezing Point Depression
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Purpose of the Freezing Point Depression Lab
To construct cooling curves of pure and mixed lauric acid to determine the freezing-point depression and the molar mass of an unknown organic substance.
Colligative Properties
Properties that only depend on the number of solute particles in
solution and not, the identity of the solute.
What happens to vapor pressure when solute is added?
Vapor pressure lowers when solute is added
The solute particles stop the solvent
particles in transitioning to the vapor phase
What happens to boiling point when solute is added?
Boiling point increases when solute is added
What happens to freezing point depression when solute is added
Freezing point depression lowers when solute is added
List the 4 colligative properties
Vapor pressure
Boiling point
Freezing point
Osmotic pressure
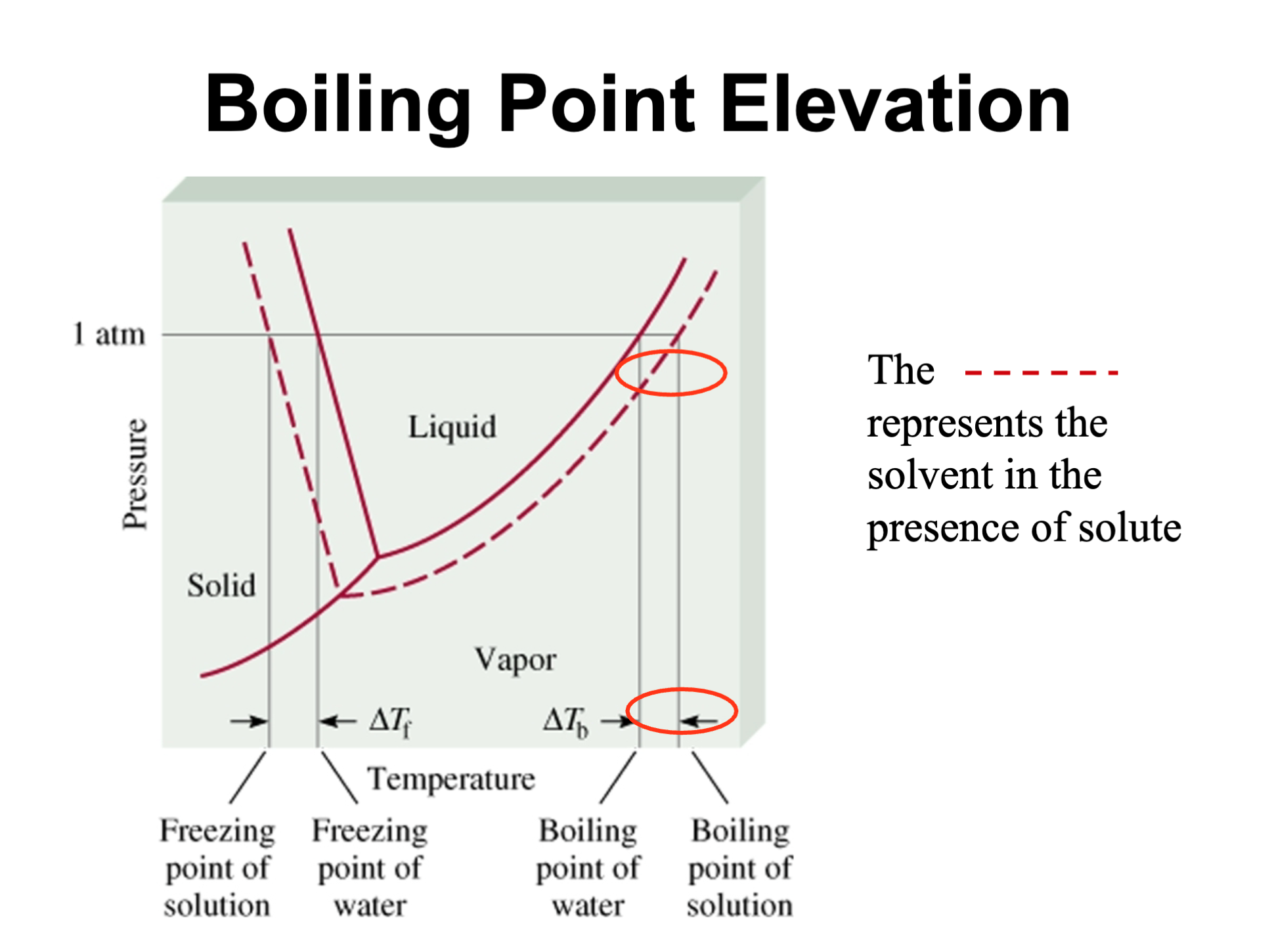
Boiling Point Elevation Graph
Solid to the left
Liquid in the Y
Vapor to the right
Dashed Line = presence of solute
Freezing-Point Depression Equation
ΔTf = iKfm
What is ΔTf in the freezing point depression equation?
ΔTf = Tºf – T f
Tºf = freezing point of pure solvent
T f = freezing point of the solution
What is Kf in the freezing point depression equation?
The molal freezing point depression constant (0C/m) that is specific to the solvent
what does m stand for in the freezing point depression equation?
m = molality, moles of solute/ kg of solvent
What data do you need to determine molar mass using freezing point depression?
• Amount of solvent
• Amount of solute added
• Freezing point (or b.p.) before and after adding solute
What is the Van’t Hoff Factor (i) the freezing point depression equation?
Number of particles a solute forms in solution
( i = 1 for nonionic compounds)
Whats the purpose of acetone in this lab?
To remove lauric acid residue
What are the hazards for this lab?
Lauric acid, and the unknown solids are all Flammable.
They are all eye, skin and respiratory irritants.
Calculation Example
A solution containing 0.050 g of an unknown nonelectrolyte in 2.50 g of Cyclohexane was found to freeze at 5.1ﹾC . What is the molar mass of the Unknown substance ?
Kf of cyclohexane = 20.4 ﹾC/m
Freezing point of pure cyclohexane = 6.6 ﹾC
find change of temp: ΔT = 6.6 – 5.1 = 1.5 ﹾC
Substitute in thethe equation to find molality: ΔT = Kf m
1.5 ﹾC = 20.4 ﹾC/m x m
m = 1.5 / 20.4 = 0.0735 = moles of solute
Divide g of solute by 1000 kg 2.50 g /1000 = 0.0025 kg
Divide moles of solute by kg = 0.0735 x 0.0025 = 0.0001837 mol
Divide g by moles of unknown to get molar mass unknown = g /mol = 0.050 g / 0.0001837 mol = 272.18 g/mol
What are the x and y coordinates of the cooling graphs?
x = time(seconds), y = temperature(celsius)
How to find freezing point depression in cooling graphs:
where the graph starts to flatten
Materials (7)
1 large test tube
Hotplate
Beaker
Ring Stand
Utility Clamp
Rubber Stopper
Digital thermometer through the rubber stopper
What is the i value for Nacl?
i = 2 because NaCl (aq) = Na+ (aq)+ Cl- (aq) (2 particles)
How do you calculate molality from ΔTf? (Assume i = 1)
m = ΔTf / Kf
Solvent used in the experiment
Lauric Acid
Kf of lauric acid
3.90 °C/m
Why do we make cooling curves?
To determine the freezing point from the plateau region.
What does the flat part of the cooling curve represent?
The freezing point of the substance.
General steps to solve the calculations
Find ΔTf
Solve for molality with freezing point depression equation ΔTf=iKfm
Find Moles of Solute: mol solute=(molality) ×(kg solvent)
Calculate Molar Mass: g of unknown/ mol of solute
why do nonelectrolytes have i = 1
nonelectrolytes do not dissociate into ions when dissolved in a solvent so it produces 1 particle in the solution