L16 Renal pharmacology ADH and RAAS
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
what does the posterior pituitary do (neurohypophysis)
stores and secretes hormones synthesised in the hypothalamus (oxytocin, ADH)
what does the anterior pituitary do (adenohypophysis)
synthesises and secretes hormones in response to hypothalamic regulation (TSH, ACTH, FSH, LH, GH, PL)
what does the posterior pituitary consist of
neuronal projections (axons) extending from the supraoptic and paraventricular nuclei of the hypothalamus
these axons release peptide hormones into capillaries of the hypophyseal circulation
the posterior pituitary doesnt produce any hormones, but simply stores and secretes…
hormones produced in the hypothalamus
what hormones does the posterior pituitary secrete
Antidiuretic hormone (ADH, vasopressin)
regulation of renal function
Oxytocin
regulation of uterine contraction (both short peptides (9.a.a)
stored in neurosecretory vesicles
posterior pituitary secretes ADH in response to low blood volume/pressure (baroreceptor)
ADH binds to blood vessel smooth muscle V1 GPCR to initiate vasoconstriction (maintains blood pressure)
negative feedback loop
posterior pituitary secretes ADH in response to high osmolarity (osmoreceptors) in the hypothalamus
ADH binds to distil tubule V2 receptors on the GPCR which increase water reabsorption to maintain blood volume/pressure
negative feedback loop
anterior pituitary in the hypothalamus signals posterior pituitary to secrete ADH from
paraventricular nucleus cells
ADH along an axon past axon terminals and into the capillary and out into the veins
mechanism of action of ADH
ADH binds to V2 receptors on the basolateral of principle cells
this promotes conversion of ATP to cAMP via adenylate cyclase
PKA is activated and promotes fusion of aquaporin 2 into the apical luminal membrane, enhancing permeability to H20
This increases water permeability in the distal convoluted tubule and the collecting duct which results in concentrated urine
the ascending limb of the loop of henle, DCT and collecting duct is impermeable to…
H20
H20 absorption can occur in the late DCT and collecting duct in the presence of the peptide hormone…
ADH
ADH promotes membrane fusion of
Aquaporin 2
concentrating urine (anti-diuresis)
ADH hormone level stimulants
opioids
anti-depressants
nicotine
MDMA
ADH hormone level depressant
alcohol
syndrome of inappropriate ADH secretion (SIADH)
excessive ADH secretion leads to high urine osmolatity which increases total body water
hyponatremia
hypoosmotic blood plasma
hypervolemia
what are the causes of inappropriate ADH secretion (SIADH)
post operative trauma and stress (metabolic) 30%
head trauma (20%)
ectopic ADH production (tumours) (20%)
drugs
what is treamtent for inappropriate ADH secretion (SIADH)
ADH V2 Antagonist
Tolvaptan
describe inappropriate ADH secretion (SIADH) syndrome
increasing ADH increases water permeability of the late distal convoluted tubule and the collecting duct which increases blood volume
increased blood volume dilutes sodium in the blood so a compensatory mechanism in the kidney decreases renin which decreases angiotensin 2 which decreases mineralocorticoids and decreases sodium absorption into the blood
this means a high concentration of sodium is in the urine that makes hyponatremia worse
describe diabetes insipidus
non-functional ADH system resulting in excessive water loss
polyuria, polydipsia, hypernatremia, hypotension
describe neurogenic diabetes insipidus
failure of ADH secretion-lesion of hypothalamus or pituitary
treatment for neurogenic diabetes insipidus
synthetic ADH (Desmopressin)
describe nephrogenic diabetes insipidus
failure of principal cells to respond to ADH (V2 receptor mutation)
what is the treatment for diabetes insipidus
restricted Na+ diet
what is too little ADH treated with
V2 agonists
lypressin
desmopressin
excess ADH treatment
V2 antagonist
demeclocylcine
tolvaptan
adrenal gland adrenal cortex anatomy zona glomerulosa
mineralocorticoids (aldosterone)
adrenal gland adrenal cortex anatomy zona fasciculata
glucocorticoids (cortisol)
adrenal gland adrenal cortex anatomy zona reticularis
androgens (DHEA)
adrenal gland anatomy medulla
AD and NAD
adrenal steroidogenesis
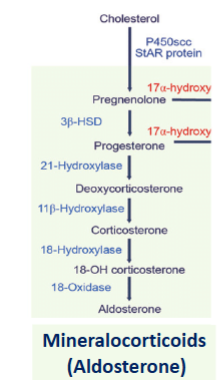
cholesterol forms pregnenolone which forms progesterone which forms deoxycorticosterone which forms corticosterone which forms 18-OH corticosterone which forms aldosterone (mineralocorticoid)
describe steroid hormone receptor signalling
steroids diffuse across the cell membrane and into the cell
binding to intracellular receptors induces translocation to the nucleus
activated receptors bind to specific target gene response elements in DNA
modulates transcription of specific genes (synthesis of mRNA)
mRNA is translated to the protein
protein exerts its effect on the cell, altering cellular activity
what does aldosterone regulate
Na+ and K+ balance (water balance)
acts on distal tubules principle cells to increase Na reabsorption (blood)
increases K+ secretion (urine)
aldosterone is a nuclear steroid hormone synthesized by
zona glomerulosa of the adrenal gland
aldosterone acts on mineralocorticoid receptors specific to the
kidney
also colon, bladder, sweat glands
nuclear steroid receptor
regulates DNA transcription
direct aldosterone regulation
stimulated by a low plasma Na+ or a high K+ (action on zona glomerulosa cells of the adrenal cortex)
indirect aldosterone regulation
stimulated by angiotensin II
what are the primary causes of hyperaldosteronism
adrenal gland
Conn’s syndrome- adrenal hyperplasia/tumour of z.glomerulosa
what are the secondary causes of hyperaldosteronism
pathology outside of the adrenals
chronic low blood pressure- congestive heart faillure= high renin =excess aldosterone
increasing aldosterone causes decreased Na+ secretion into the urine
this causes Hypernatremia (thirst, odema) that causes hypertension (headache, fatigue)
increasing aldosterone increases K+ secretion into the urine
this causes Hypokalemia (heart arrhythmia, constipation)
what is hyperaldosteronism treated with
Mineralocorticoid Receptor antagonist
spironolactone
eplerenone
primary causes of hypoaldosteronism
adrenal gland- Addison’s disease -autoimmune disorder- destruction of z.glomerulosa cells
secondary causes of hypoaldosteronism
pathology outside of the adrenals
renin deficiency -genetic predisposition (certain ethnic groups)
decreasing aldosterone increases Na+ secretion into urine
this causes hyponatremia (confusion, fatigue) which causes hypotension (vascular collapse, dizziness)
decreased aldosterone causes decreased K+ secretion into urine which causes
hyperkalemia (heart arrhythmias, constipation)
hypoaldosteronism is treated with
a mineralocorticoid agonist
fludrocortisone
what is hypersecretion of aldosterone (Conn’s syndrome) treated with
MR antagonist: spironolactone, eplerenone
what is hyposecretion of Aldosterone (Addison’s) treated with
replacement therapy
fludrocortisone (aldosterone itself isnt used due to a short half life)
mineralocorticoid summary
Endogenous Ligand: Aldosterone
Receptor: Mineralocorticoid Receptor (MR)
Regulated by: Na+ / K+ and Renin/Angiotensin
Physiological function - water & electrolyte
balance
(increases transcription of Na channels
and Na/K-ATPase)
Main Disorders
Hyperaldosterone: Conn’s disease
Hypertension – MR antagonist Spironolactone
Hypoaldosterone: Addison’s disease
Hypotension – MR agonist Fludrocortisone
glucocorticoids summary
Endogenous Ligand: Cortisol
Receptor: Glucocorticoid Receptor (GR)
Regulated by HPA axis (stress, circadian, feedback)
Physiological function – Increased metabolism
(gluconeogenis & protein catabolism)
Supraphysiological : Anti-inflammatory/
immunosuppressive
(Decrease cytokines and inflammatory mediators)
Exploited therapeutically: Glucocorticoid agonists –
Dexamethasone, Hydrocortisone, Beclomethasone
Main Disorders
Hypercortisol: Cushing’s syndrome
Metabolic dysfunction – Cortisol Synthesis
antagonist - Metyrapone
aldosterone has high affinity for MR but
low affinity for GR
cortisol has high affinity for MR but
low affinity for GR
at basal circulating plasma levels of cortisol, MR receptors are
fully saturated by cortisol
basal conditions: cortisol conc is higher than aldosterone
MR is fully saturated by Cortisol
MR will not respond to a change in aldosterone
in specialised tissues, MR is associated with high levels of
11beta-hydroxysteroid dehydrogenase enzyme (11beta-HSD)
11beta-HSD metabolises/removes cortisol
carbenoxolone (glycerrhetinic acid from liquorice) is used in the treatment of
oral and gastric ulcer
liquorice in pregnancy has side effects (glycerrhetinic acid/glycyrrhiza)
11beta HSD in the placenta protects the foetus from elevation in cortisol from the maternal bloodstream
baby weight
mental cognition- oppositional defiant disorder