Theme 4 Module 1
1/81
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
Why is cell division essential for prokaryotic organisms?
Because in prokaryotes, cell division = reproduction, producing a new organism from a single cell.
What capabilities allow prokaryotes to reproduce successfully?
A: Prokaryotic cells can:
Make exact copies of their gemone
Segregate one copy of the genome into each daughter cell
Carry all necessary molecular machinery within a single cell
What is the outcome of prokaryotic cell division?
Two genetically identical daughter cells.
What is the process of cell division in prokaryotes called, and how does it begin?
Back (Answer):
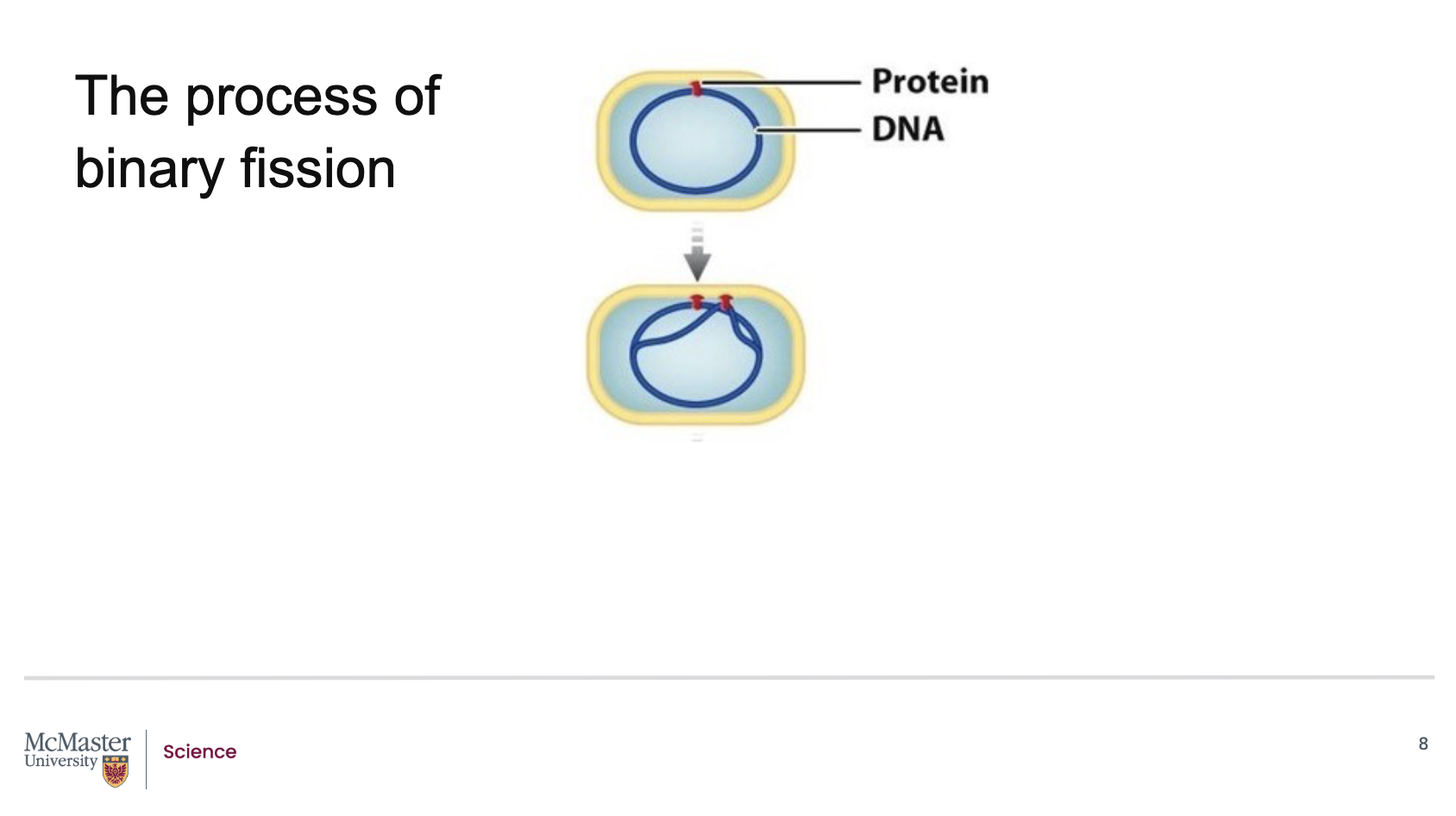
Cell division in prokaryotes is called binary fission, a type of asexual reproduction.
It begins when the bacterial chromosome attaches to the inside of the plasma membrane.
From there, DNA replication starts at the origin of replication on the chromosome.
What happens during the later stages of binary fission in prokaryotes?
Back (Answer):
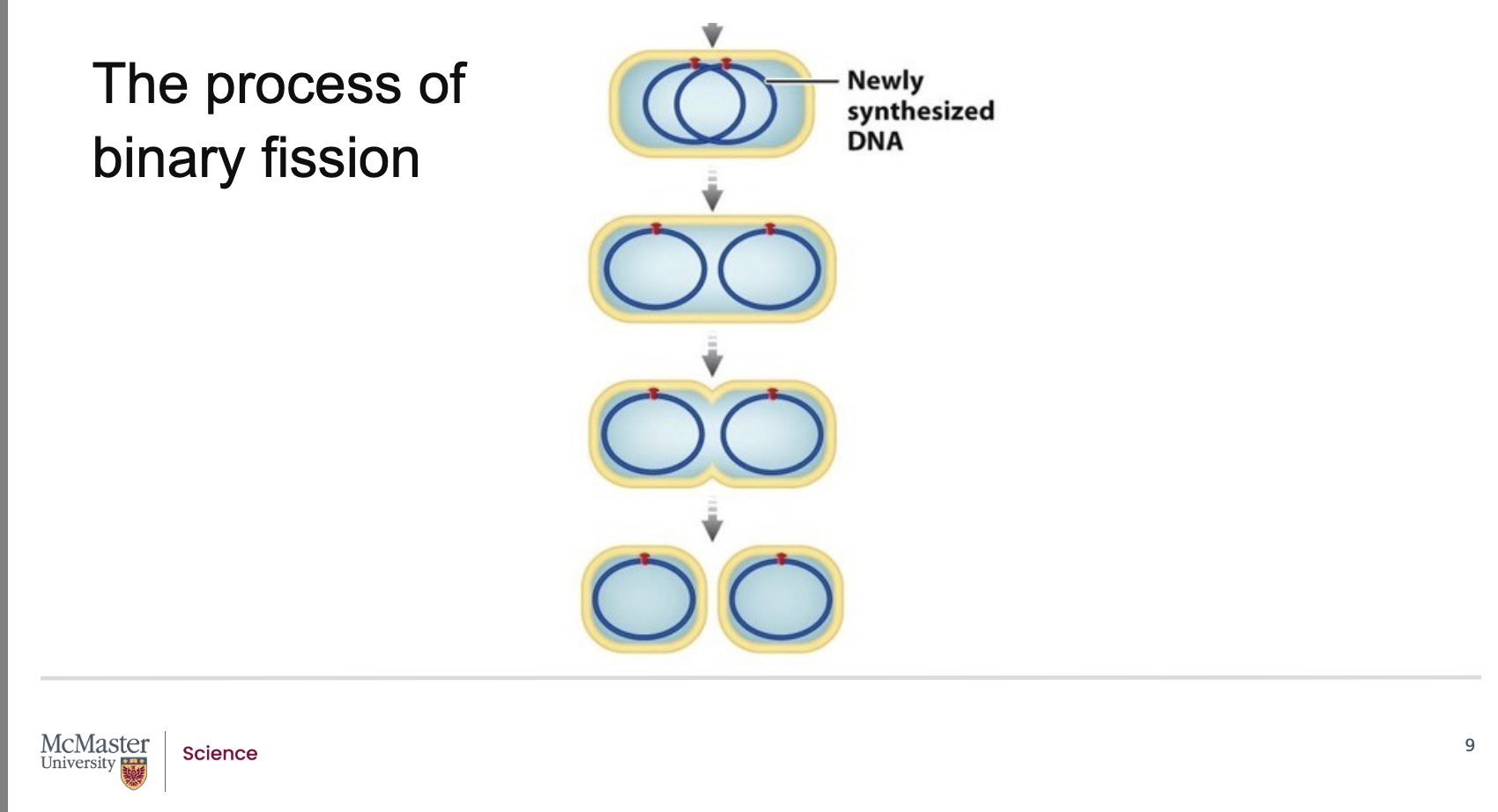
As the chromosome replicates, the cell elongates, and the new DNA attaches to the plasma membrane.
The cell keeps elongating until the two DNA attachment points reach opposite ends of the cell.
When DNA replication is finished and the cell is about double its original size, it begins to constrict at the midpoint.
New cell membrane and cell wall material form, dividing the cell into two identical daughter cells.
In contrast, eukaryotes divide using a more complex process called mitosis.
What roles does cell division play in eukaryotic organisms, and how do embryonic stem cells differ from adult stem cells?
Back (Answer):
In eukaryotes, cell division enables a single fertilized egg to develop into a complex multicellular organism.
Embryos contain stem cells that are unspecialized, can divide indefinitely, and can differentiate into many or all cell types under the right conditions.
In adults, cell division supports tissue renewal and repair.
Adult stem cells can replace certain specialized cells but are more limited—they cannot give rise to all cell types like embryonic stem cells can.
Development: Cell division allows a unicellular fertilized egg (zygote) to develop into a complex multicellular organism.
Embryonic stem cells: Present in early embryos; they are unspecialized, can reproduce (divide) indefinitely, and—under appropriate conditions—can differentiate into specialized cells of one or more types.
Adult functions: After full growth, cell division supports continual renewal and repair of cells in various tissues.
Adult stem cells: Also present in the adult body, but unlike embryonic stem cells, they cannot give rise to all cell types; instead, they replace non‑reproducing specialized cells (i.e., they have more limited differentiation potential).
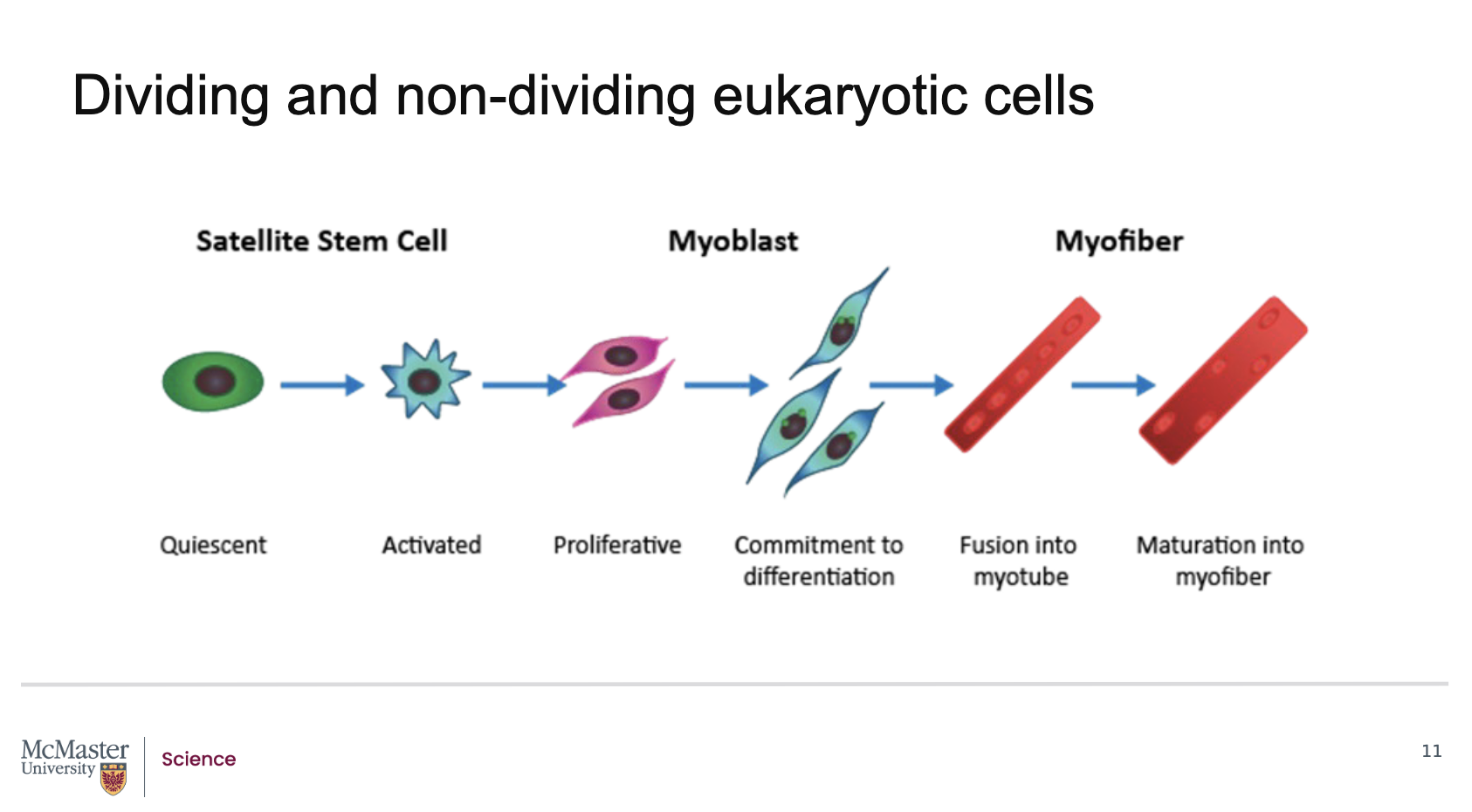
What role do satellite stem cells play in adult skeletal muscle?
In adult skeletal muscle, satellite stem cells are normally quiescent (non‑dividing) and located in the basement membrane of muscle tissue. When muscle is injured, these cells become activated, resume cell division, and contribute to muscle regeneration.
What happens after satellite stem cells are activated during muscle injury?
Activated satellite stem cells undergo:
Proliferation
Differentiation
Fusion
to form muscle precursor cells called myoblasts, which eventually develop into mature muscle cells in muscle fibers.
What are myoblasts, and what is their role in muscle regeneration?
Myoblasts are muscle precursor cells formed from activated satellite stem cells. They differentiate and fuse to create mature muscle cells that make up muscle fibers (myofibers).
Can mature muscle fibers (myofibers) divide? Why or why not?
No. Mature muscle fibers (myofibers) are terminally differentiated, meaning they cannot divide. Once formed, they are committed to their specialized function and no longer participate in the cell cycle.
What is the difference between quiescent cells and terminally differentiated cells?
Back:
Quiescent cells (e.g., satellite stem cells) can be dormant but reactivated to divide under certain conditions, such as tissue injury.
Terminally differentiated cells (e.g., mature muscle fibers) have permanently exited the cell cycle and cannot divide again.
Why is skeletal muscle considered a stable tissue, and what changes during injury?
Skeletal muscle normally has very low cell turnover and little cell division. During injury, however, satellite stem cells become activated, divide, and regenerate damaged muscle tissue.
What is one of the main distinctions between prokaryotic and eukaryotic cell division?
Eukaryotic DNA is larger, organized into linear chromosomes, and highly condensed within the nucleus, so eukaryotic cell division requires more regulated control as part of a larger cell cycle.
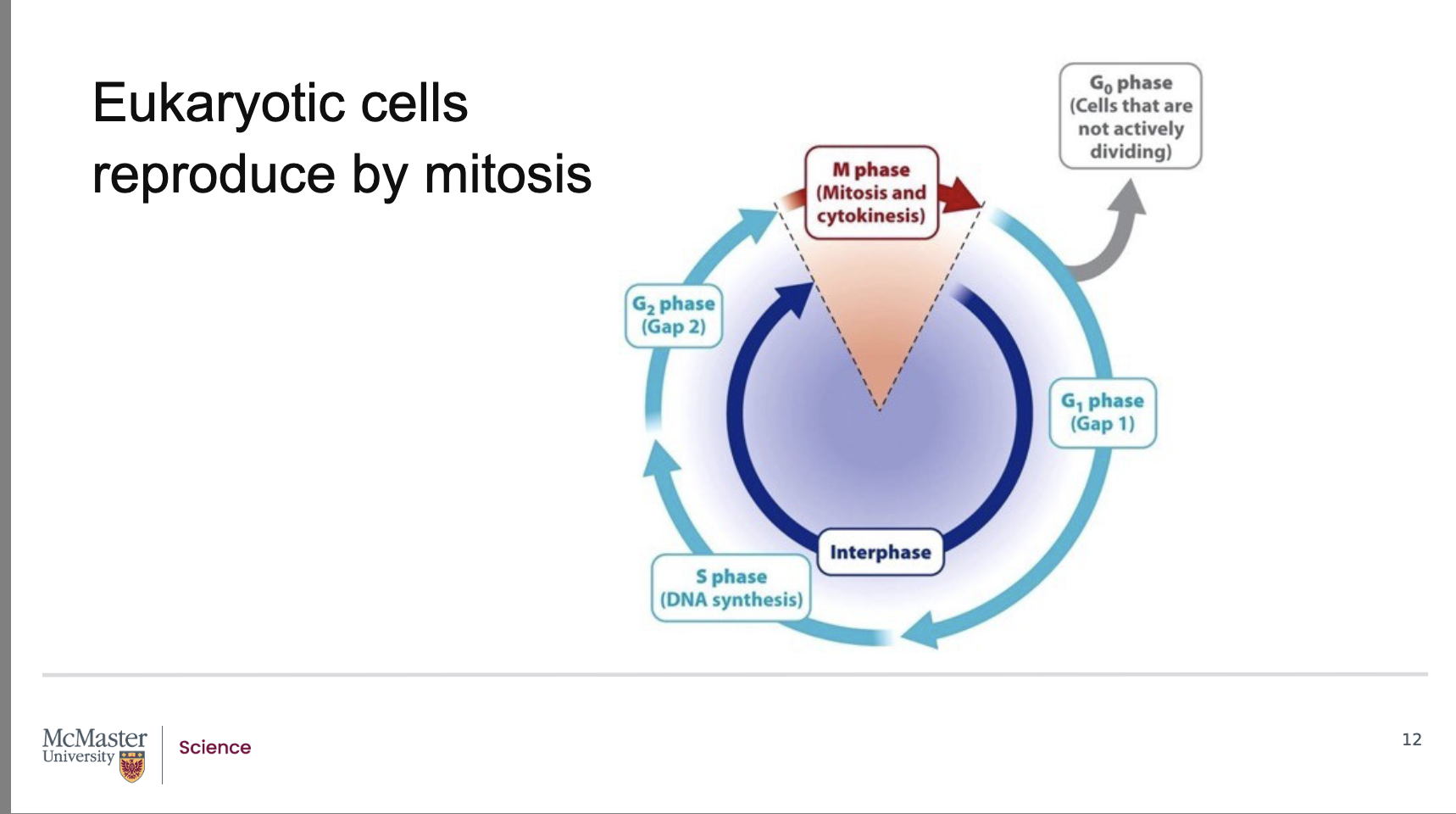
What are the two major stages of the standard eukaryotic cell cycle?
Back:
Interphase – includes:
G1 phase (first gap; growth and preparation for DNA synthesis)
S phase (DNA synthesis; replication of chromosomes)
G2 phase (second gap; preparation for mitosis)
M phase – includes:
Mitosis
Cytokinesis
What important events occur during interphase?
During interphase, cells:
Prepare for cell division
Replicate DNA in the nucleus (during the S phase)
Increase in overall size
Undergo G1 and G2 phases, which prepare the cell for DNA synthesis and mitosis, respectively.
With each mitotic cell division, the linear chromosomes of
eukaryotes must be replicated and then separated
into daughter cells. It is during the interphase stage
that cells make preparations for cell division which
include the replication of DNA in the nucleus, and an
overall increase in cell size. Replication of DNA will
occur in the S (or synthesis) phase. The G1 and G2
phases prepare the cell for DNA synthesis and mitosis
respectively
What must occur to the linear chromosomes of eukaryotes during each mitotic division?
Eukaryotic linear chromosomes must be replicated and then separated into daughter cells during mitosis.
Eukaryotic linear chromosomes must be replicated and then separated into daughter cells during mitosis.
The time required to complete the cell cycle depends on the specific cell type.
Cells that require frequent turnover, such as dividing epithelial cells of the intestine and skin, pass through the cell cycle more often.
How long does it take for the cell to pass throught the cell cycle?
The time taken for specific cells to pass through the
cell cycle depends on the type of cells in question.
For example, dividing epithelial cells of the intestine
or the skin require frequent turnover. However, not
all cells of the body participate in a regular cell cycle
that leads to regular divisions. Many cells can pause
in the cell cycle in the G0 phase. Cells in the G0 phase
pause in the cell cycle somewhere between the M
and S phase. This pause can be short, or long, ranging
from many days to more than a year. Some cells in
our body enter the G0 phase permanently and are
thus non-dividing cells. For example, cells that make
up the lenses of our eyes, nerve cells and even
mature muscle cells enter into a permanent G0 phas
Do all cells participate in regular cell division? What is the G0 phase?
Not all cells regularly divide.
Many cells can pause in the cell cycle in the G0 phase, which occurs between the M and S phases.
This pause can last days, months, or even over a year
What happens to cells that enter the G0 phase permanently?
Cells that enter permanent G0 become non‑dividing cells.
Examples include:
Lens cells of the eye
Nerve cells
Mature muscle cells
Where does the G0 phase occur in relation to the cell cycle, and how long can cells remain there?
Cells in G0 pause between M phase and S phase.
They may remain in G0 for short periods, long periods, or permanently depending on the cell type.
How do stem cells demonstrate both indefinite division and quiescence?
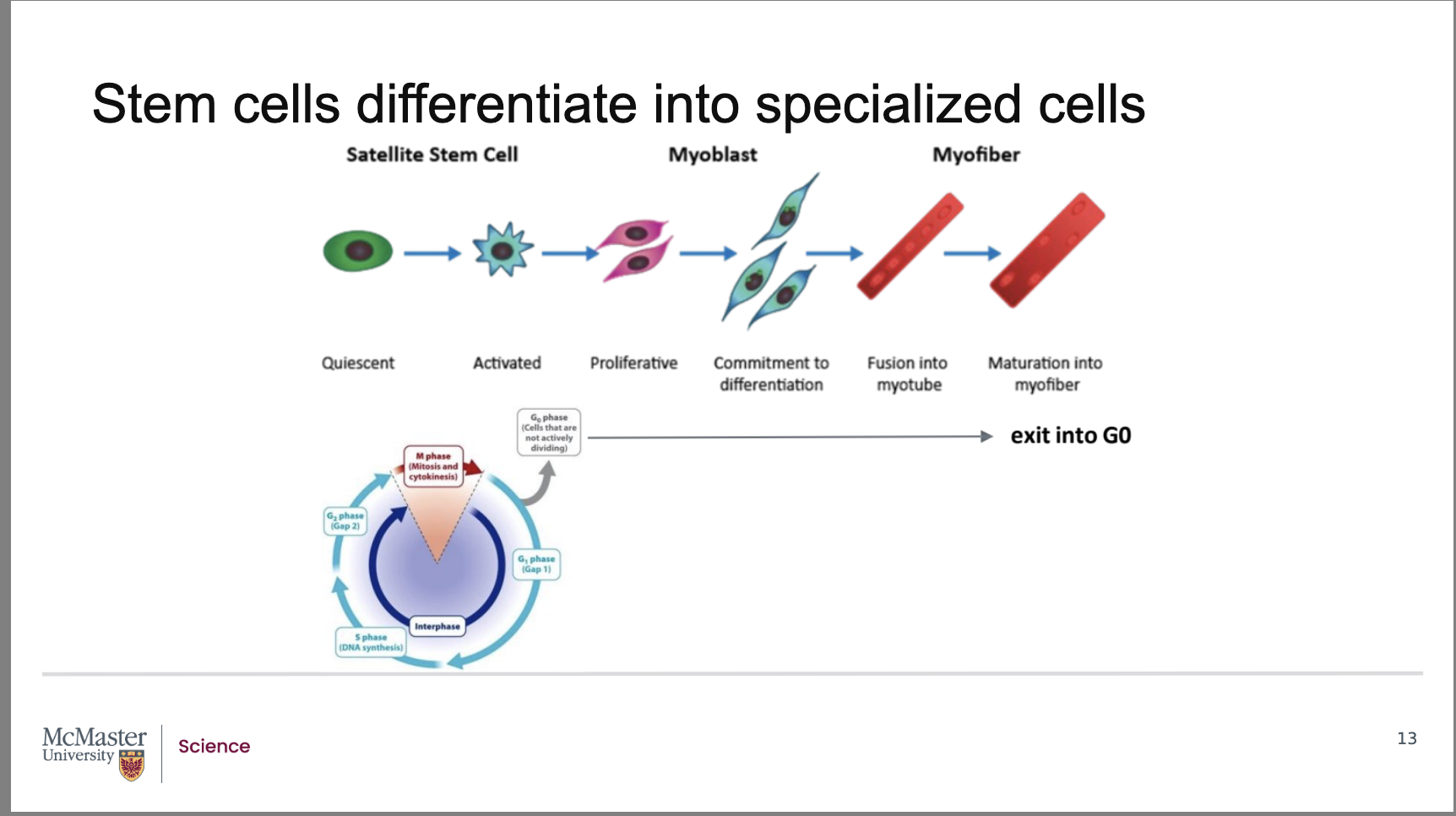
Stem cells can reproduce indefinitely, but they can also enter periods of quiescence, during which they remain in the G0 phase and undergo no cell division.
What activates quiescent satellite stem cells in skeletal muscle?
Although fully differentiated skeletal muscle has almost no cell division, injury activates quiescent satellite stem cells.
These cells exit the dormant G0 phase and re‑enter the cell cycle.
What happens after satellite stem cells re‑enter the cell cycle?
After activation and re-entry into the cell cycle, satellite stem cells undergo:
Proliferation
Differentiation
Maturation into new muscle cell precursors
These precursors ultimately fuse to repair muscle tissue by forming new muscle fibers.
Stem cells are a fine example of cells that can reproduce indefinitely, but also have periods of quiescence and therefore undergo no cell division. While fully differentiated skeletal muscle has little to no cell division, upon injury, the quiescent satellite stem cells are activated from the dormant G0 phase of the cell cycle and re-enter the cell cycle
As a result, this enables proliferation, differentiation and maturation of new muscle cell precursors that can fuse and repair the muscle tissue with new muscle fibers. Once these myofibers are formed, they once again exit the cell cycle and enter the quiescent G0 phase. How is it that these satellite cells were able to reproduce to form newly differentiated muscle cells
What happens to newly formed myofibers after muscle repair?
Once new myofibers form, they exit the cell cycle again and return to the quiescent G0 phase, becoming non‑dividing cells.
What key biological question does this slide raise about satellite stem cells?
It asks:
How are satellite stem cells able to re-enter the cell cycle and divide to form newly differentiated muscle cells when mature muscle fibers cannot?
What happens during interphase before a cell can enter mitosis?
During interphase, cells:
Replicate their DNA in the S phase
Undergo cell growth during G1 and G2 phases
These steps prepare the cell for mitosis.
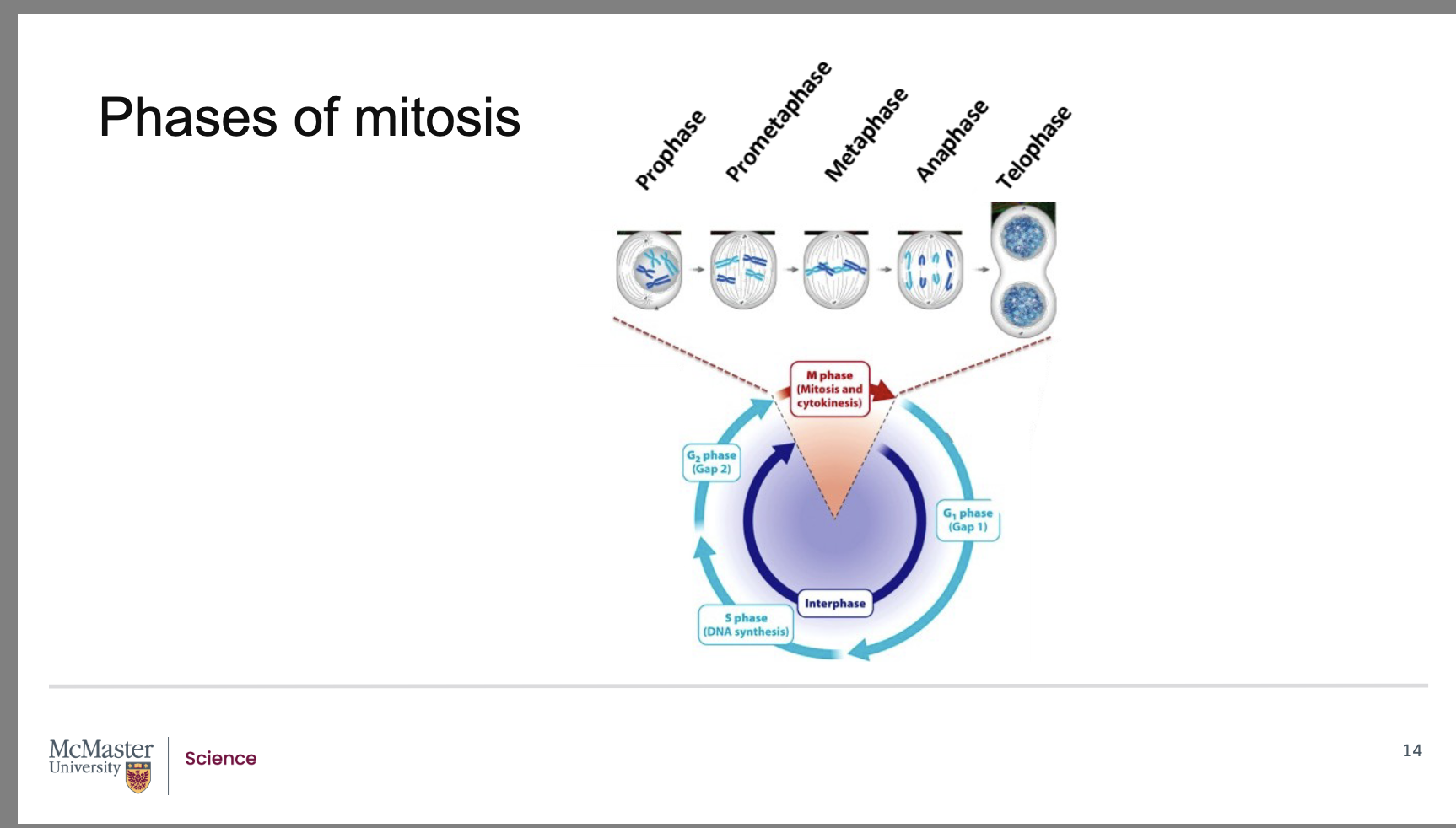
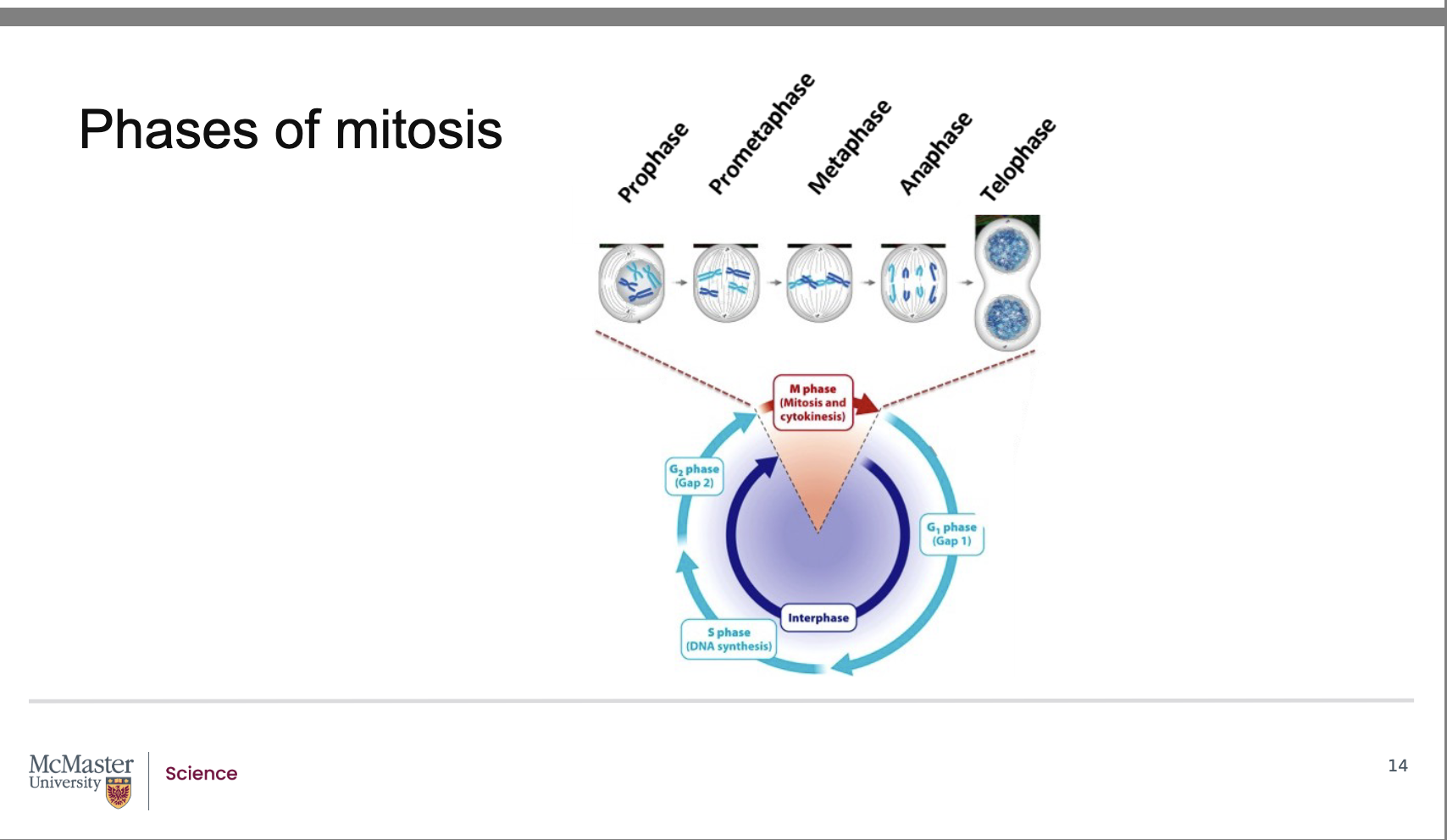
How are the stages of mitosis identified and characterized?
The five stages of mitosis are morphometrically characterized by distinctive changes in chromosome appearance and position during cell division.
Who discovered that the stages of mitosis could be identified by chromosome position and features, and when?
Walther Flemming, in 1882, discovered that mitotic stages could be distinguished by observing chromosomal position and features.
What organism did Walther Flemming study, and how did he visualize dividing chromosomes?
Flemming studied developing salamander embryos and used staining techniques to visualize chromosomes in dividing cells.
The discovery that the distinct stages of mitosis could be staged based on chromosomal position and features was identified in pioneering work by Walther Flemming in 1882. In his work, Flemming analyzed developing salamander embryos that he had stained to be able to visualize the chromosomes of the dividing cells. Further to Flemming’s work, we now know that mitosis consists of 5 stages: - -
prophase
prometaphase
metaphase
anaphase
telophase.
However, prior to entering into mitosis, the chromosomes of cells must be duplicated and condensed to allow for the daughter cells to acquire the same amount of genetic information as the parent cell in a relatively short period of time
What are the five stages of mitosis?
Back:
Prophase
Prometaphase
Metaphase
Anaphase
Telophase
What must occur before a cell can enter mitosis?
Before mitosis, chromosomes must be duplicated and condensed so daughter cells receive the same amount of genetic information as the parent cell.
Why must chromosomes be condensed before mitosis?
Chromosome condensation allows the cell to distribute genetic material accurately and efficiently to daughter cells in a short period of time.
What happens to chromosomes during interphase and S‑phase, and how are they replicated and compacted?
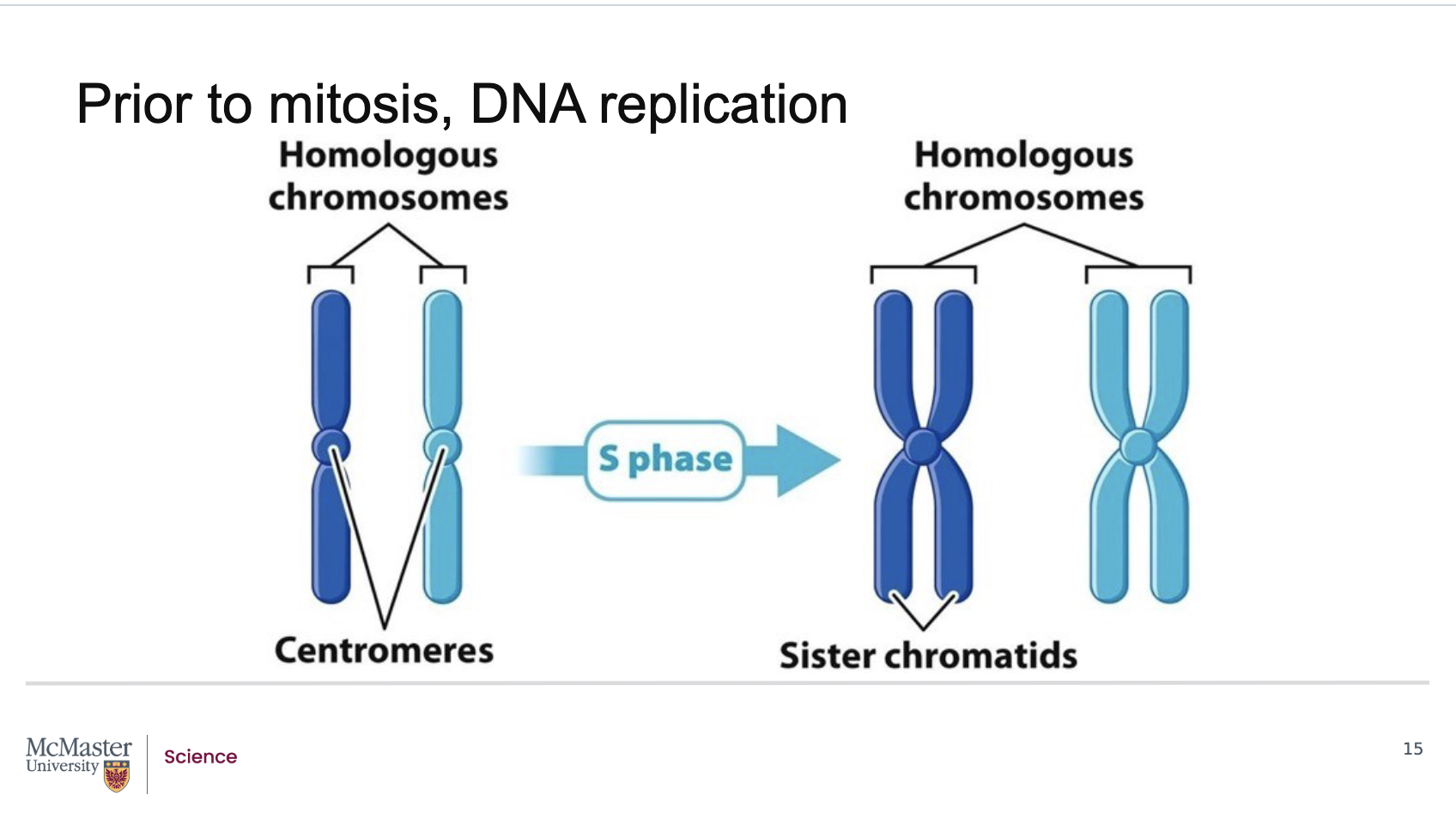
Back:
During most of interphase, chromosomes exist as long, thin chromatin fibers.
Before mitosis, exact copies of every chromosome are made during S‑phase through DNA replication, where the DNA sequence is copied from end to end.
Newly synthesized DNA associates with histones and other chromosomal proteins, enabling tight compaction.
The centromere is fully replicated, but it is so highly compacted that the paired centromeres appear fused.
What are sister chromatids, how are human chromosomes organized, and what happens to chromatids during M‑phase?
Back:
A duplicated chromosome consists of two identical sister chromatids.
Humans have 23 chromosome pairs:
we all inherit a paternal and maternal chromosome, these are what allows us to have 23 distinct chromosome pairs - where 22 are homologous chromosomes (one maternal and one paternal in origin) and 1 pair are our sex chromosomes.
22 pairs of homologous chromosomes (one maternal + one paternal)
1 pair of sex chromosomes
Chromosomes are not fully compacted until M‑phase, despite appearing so in diagrams.
As M‑phase progresses, sister chromatids separate and move into two new cells, involving major changes in chromosome dynamics.
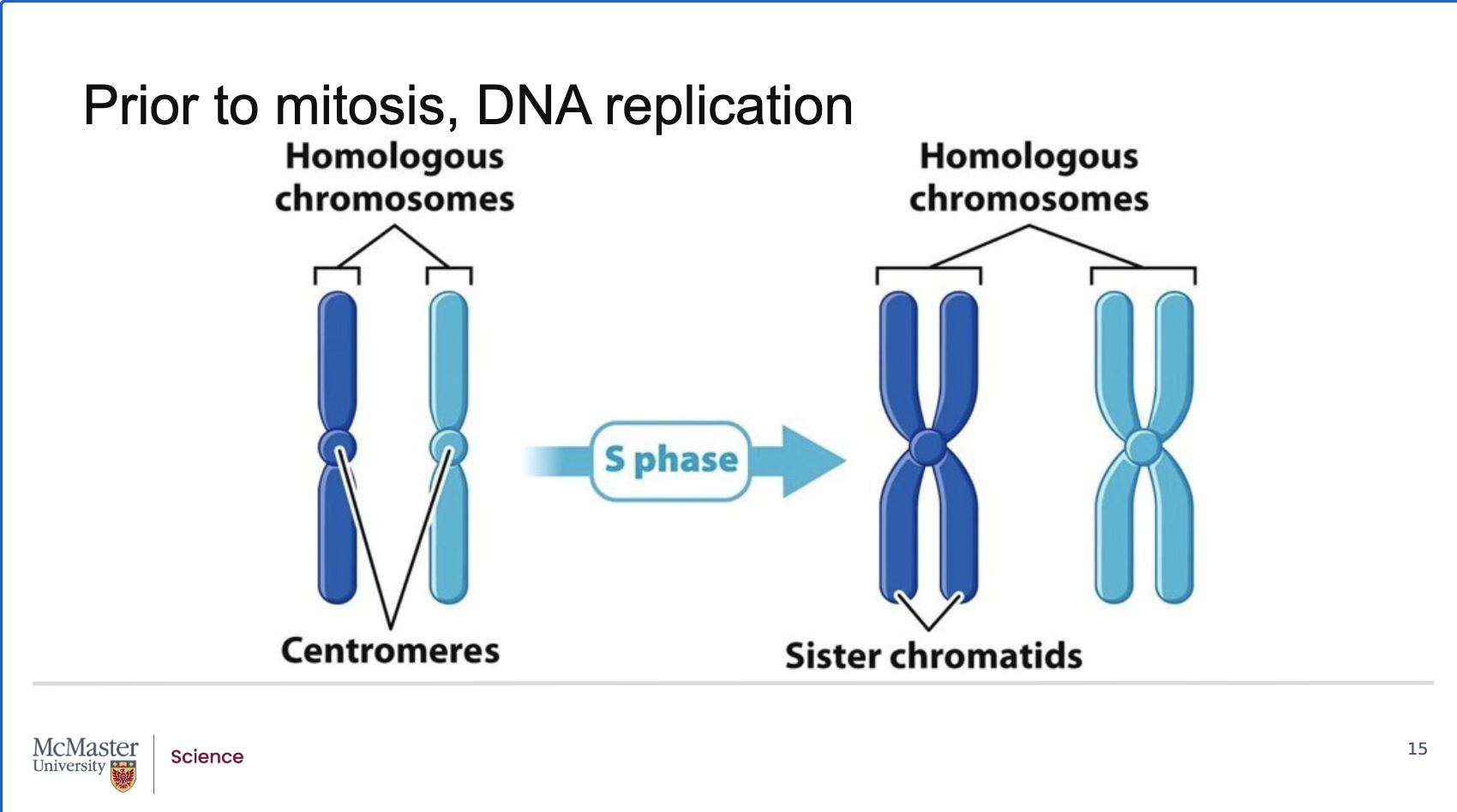
What marks the beginning of the M‑stage of the cell cycle?
The M‑stage begins immediately after the end of the G2 phase of interphase.
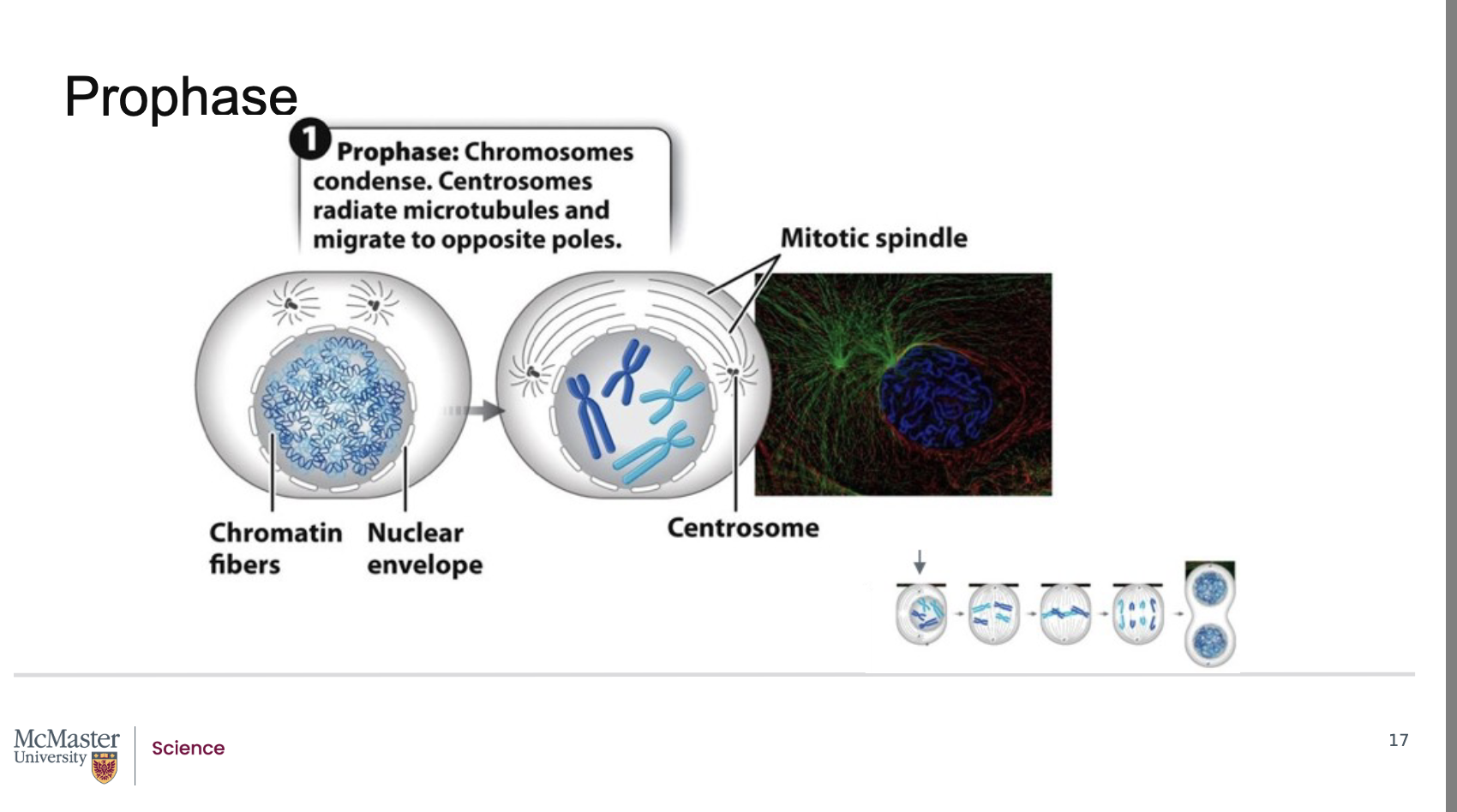
Why can individual chromosomes not be identified during most of interphase?
During interphase, chromosomes are arranged as long, thin chromatin fibers, making them not individually distinguishable.
What happens to chromosomes as a cell transitions from G2 to M‑phase?
The duplicated chromosomes condense, allowing individual chromosomes to become visible under a light microscope.
What is the first stage of mitosis, and what characterizes it?
Prophase is the first stage of mitosis.
During prophase, each chromosome appears as identical sister chromatids joined at their centromeres.
What structures begin forming during prophase and how?
Centrosomes (duplicated microtubule‑organizing centers) begin to radiate long microtubules, forming the mitotic spindle.
Where do the centrosomes move during prophase?
The centrosomes move to opposite poles of the cell.
What is the role of the mitotic spindle?
The mitotic spindle is crucial for separating chromosomes into the two daughter cells during mitosis.
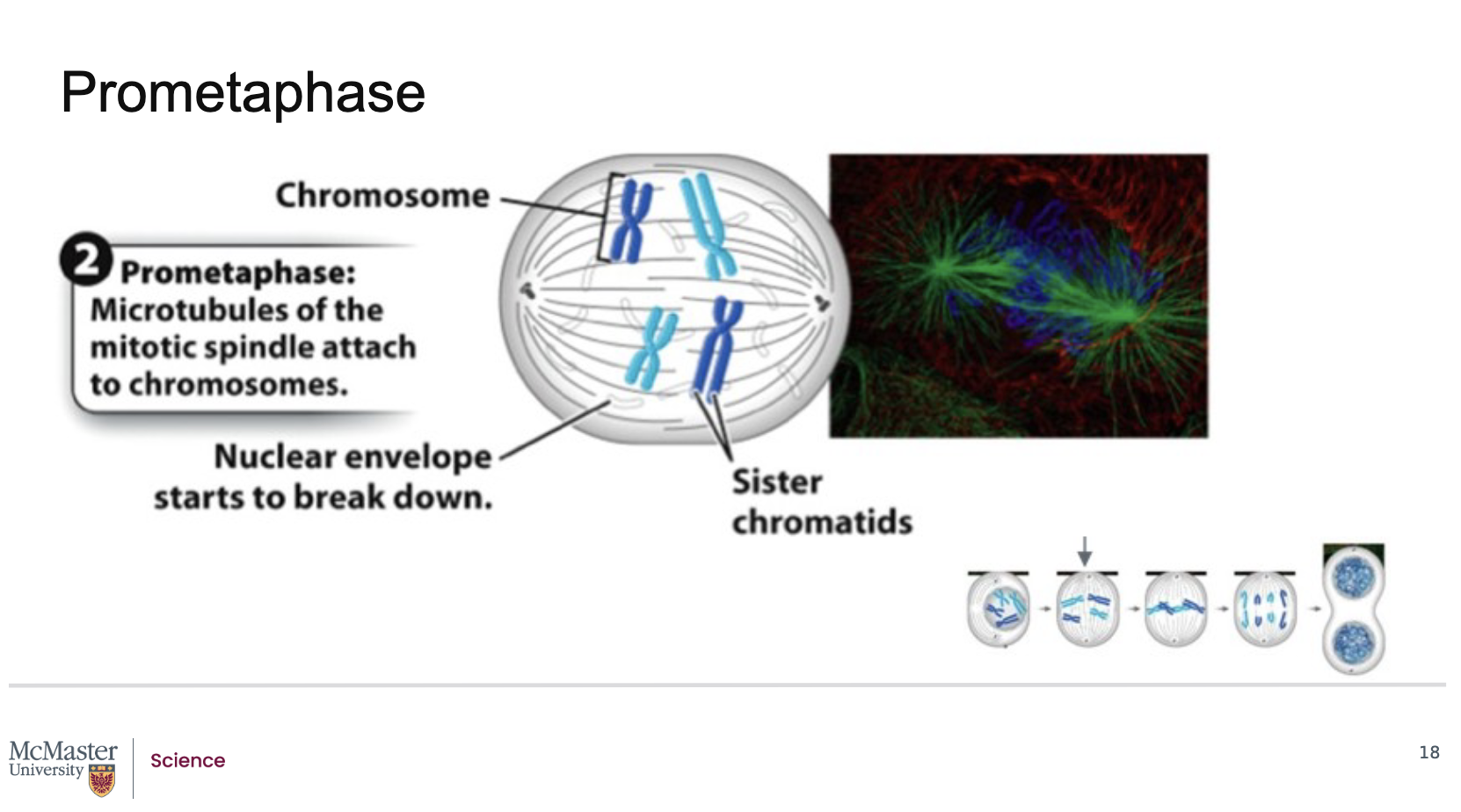
What defines prometaphase, and how does it allow microtubules to attach to chromosomes?
Back:
Prometaphase follows prophase in mitosis.
Its defining feature is the fragmentation of the nuclear envelope.
When the nuclear envelope breaks down, microtubules from each centrosome (as part of the mitotic spindle) gain access to the chromosomes.
These microtubules attach to kinetochores, which are specialized protein structures located on each of the two sister chromatids, on opposite sides of the centromere.
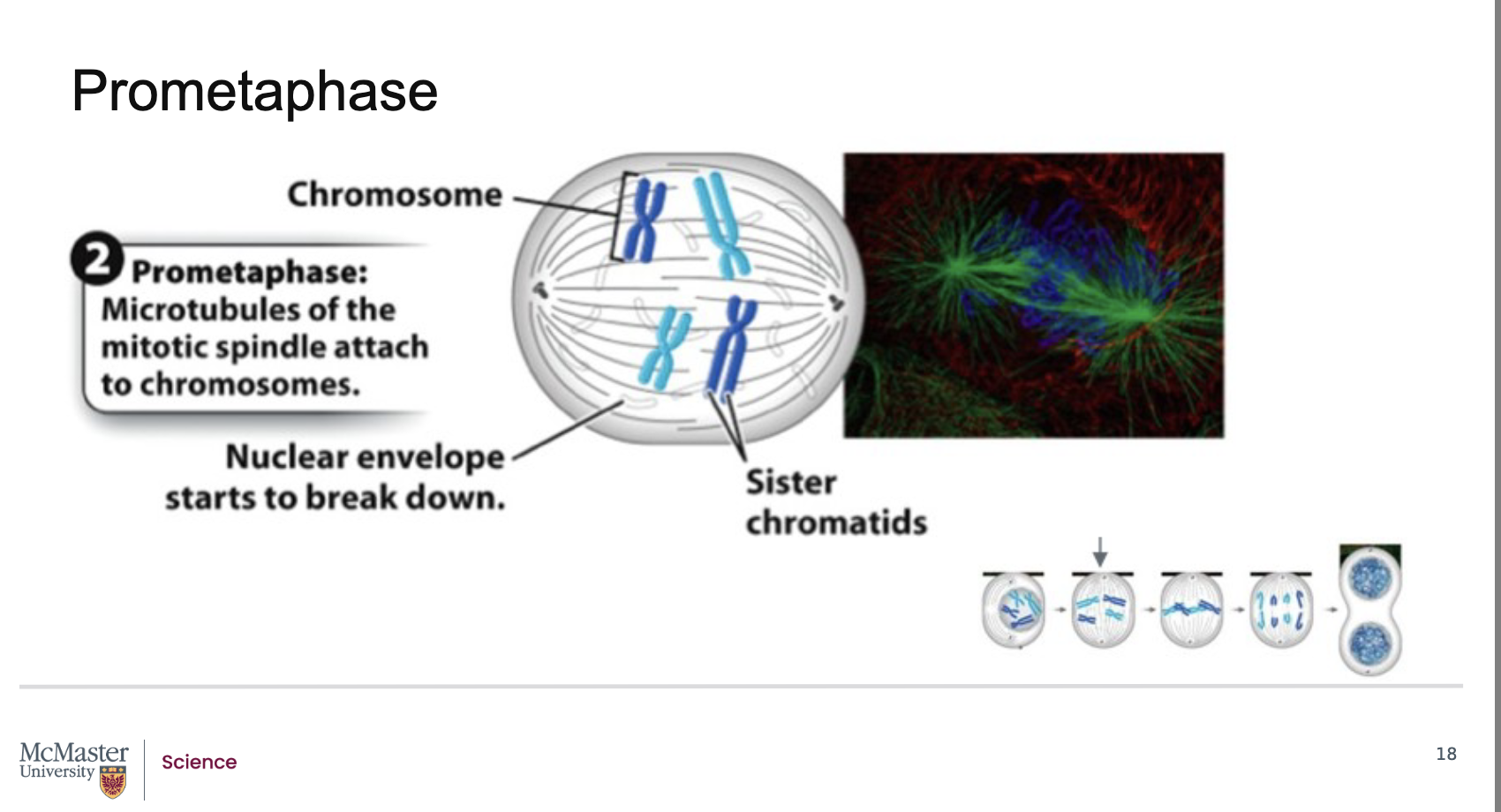
Because the nuclear envelope breaks down, the microtubules that are extending from each centrosome as part of the mitotic spindle are able to attach to specialized regions on the centromeres of the chromosomes often referred to as kinetochores. Kinetochores are specialized protein structures that associate with each one of the two sister chromatids on either side of the centromere. Some of the microtubules that radiate from the centrosome attach directly to the kinetochore regions. These kinetochore microtubules are essential to help pull the chromosomes to the poles of the cell during mitosis. Other microtubules that also radiate from the centrosome as part of the mitotic spindle are polar microtubules. These microtubules will interact with each other and help push the poles of the cell away from each other during mitosis
How do kinetochore microtubules and polar microtubules function during prometaphase?
Back:
Kinetochore microtubules:
Extend from centrosomes and attach directly to the kinetochore regions.
Essential for pulling chromosomes toward opposite poles during mitosis.
Polar microtubules:
Also extend from the centrosomes but do not attach to chromosomes.
Instead, they interact with polar microtubules from the opposite spindle pole.
Their interaction helps push the poles of the cell apart during mitosis
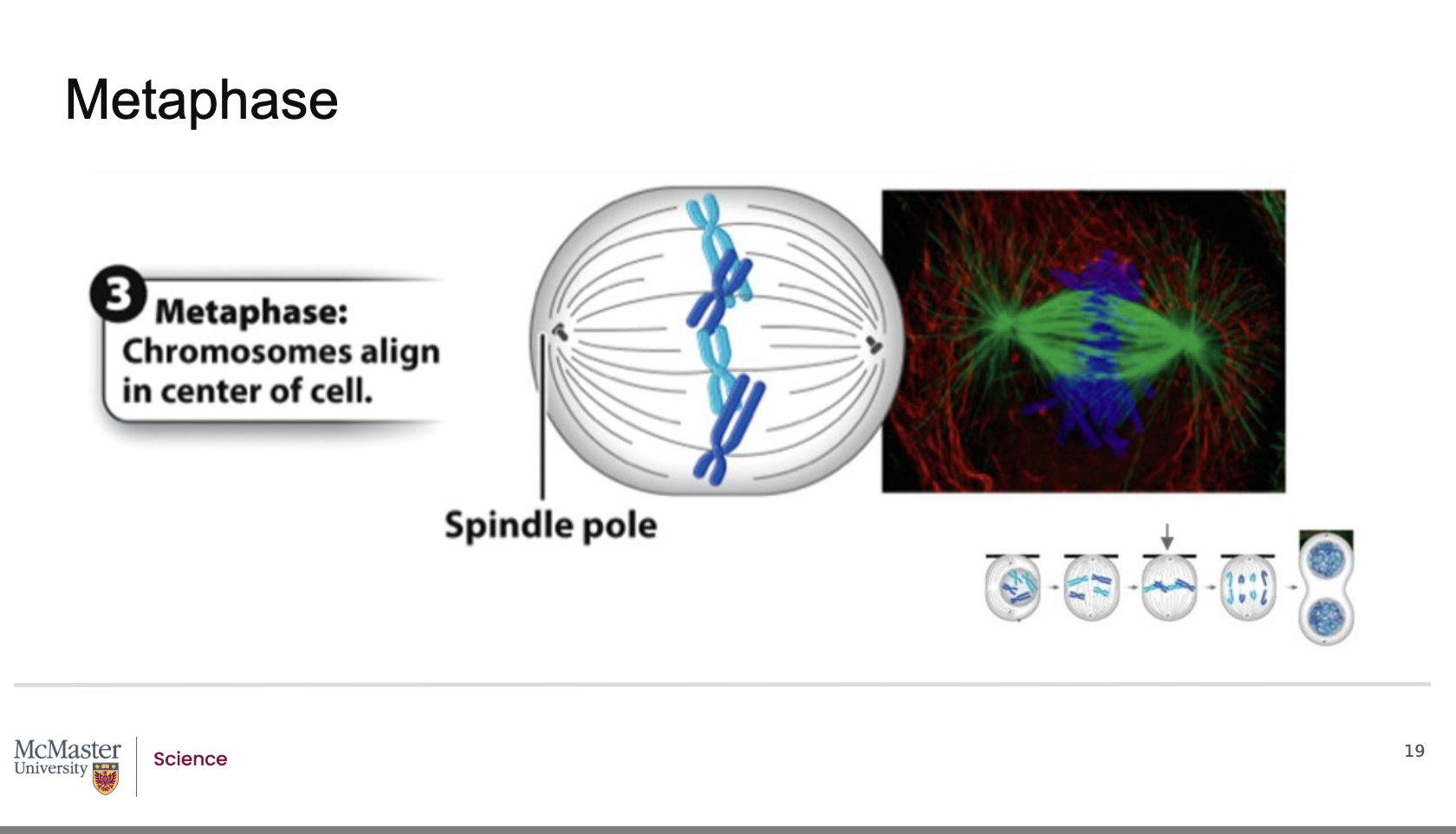
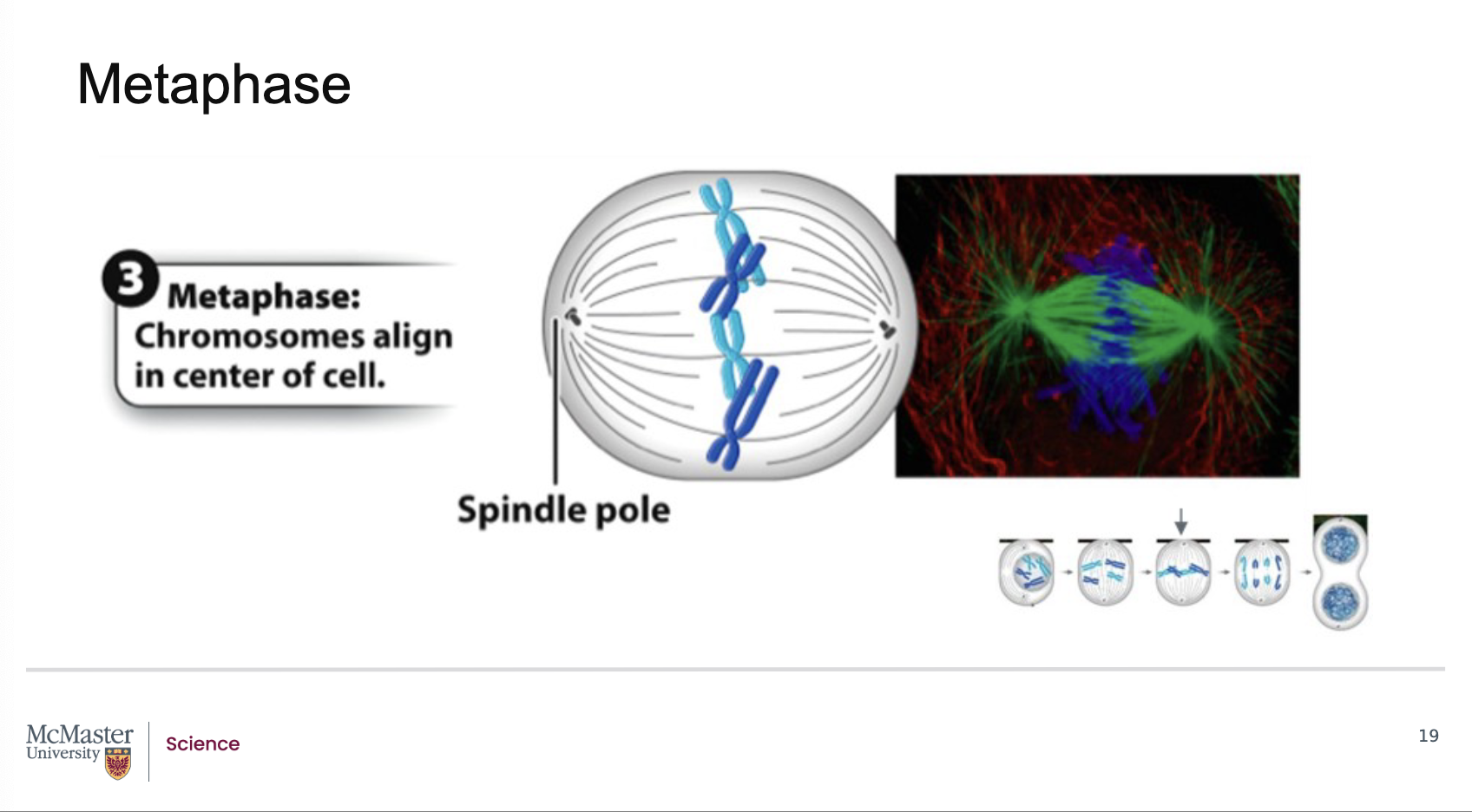
What defines metaphase in mitosis, and where do chromosomes align?
Back:
Metaphase is the third stage of mitosis.
It is marked by the alignment of chromosomes at the center of the cell.
This central region where chromosomes line up is called the metaphase plate.
How do chromosomes become aligned at the metaphase plate during metaphase?
Back:
Each chromosome has kinetochores on the sister chromatids.
Kinetochore microtubules attach to these kinetochore regions.
These microtubules exert coordinated forces that position chromosomes at the metaphase plate, ensuring proper alignment before separation
Metaphase is the third stage of mitosis and is marked
by the alignment of chromosomes at the center of
the cell in a region identified as the metaphase plate.
For each chromosome, the kinetochore microtubules
are attached at the kinetochores of each sister
chromatid and facilitate this alignment at the
metaphase plate
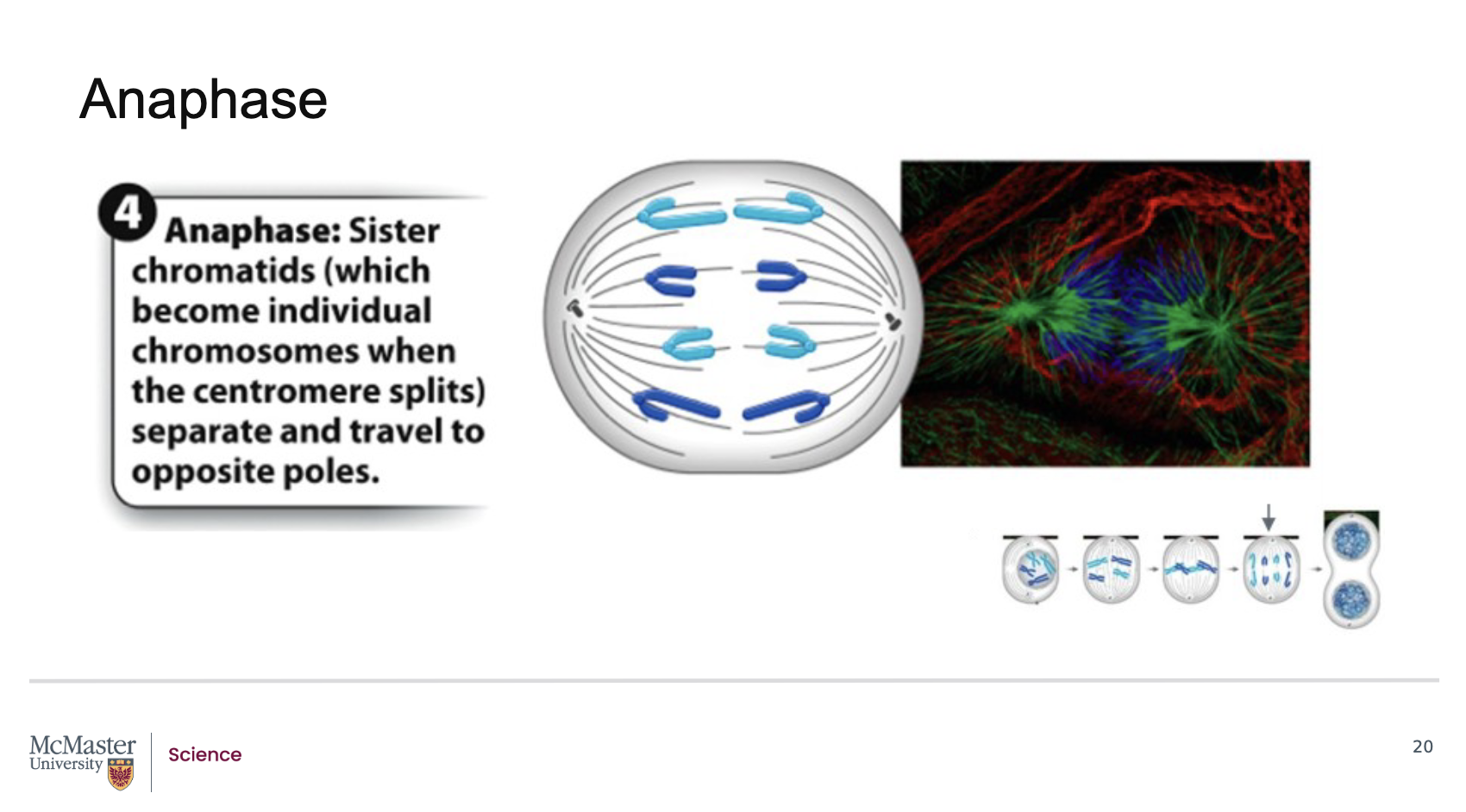
What defines anaphase and how are chromosomes separated during this stage?How do microtubules shape the cell during anaphase, and what is achieved by the end of this stage?
Anaphase is the fourth stage of mitosis.
During anaphase, kinetochore microtubules begin to shorten.
This shortening pulls the sister chromatids apart, separating them into individual chromosomes.
The separated chromosomes are pulled toward opposite spindle poles of the cell
Polar microtubules push against each other, which helps elongate the cell.
By the end of anaphase, the two ends of the cell each possess equivalent and complete sets of chromosomes, ensuring accurate genetic distribution.
What defines telophase, and what major nuclear changes occur during this stage of mitosis?
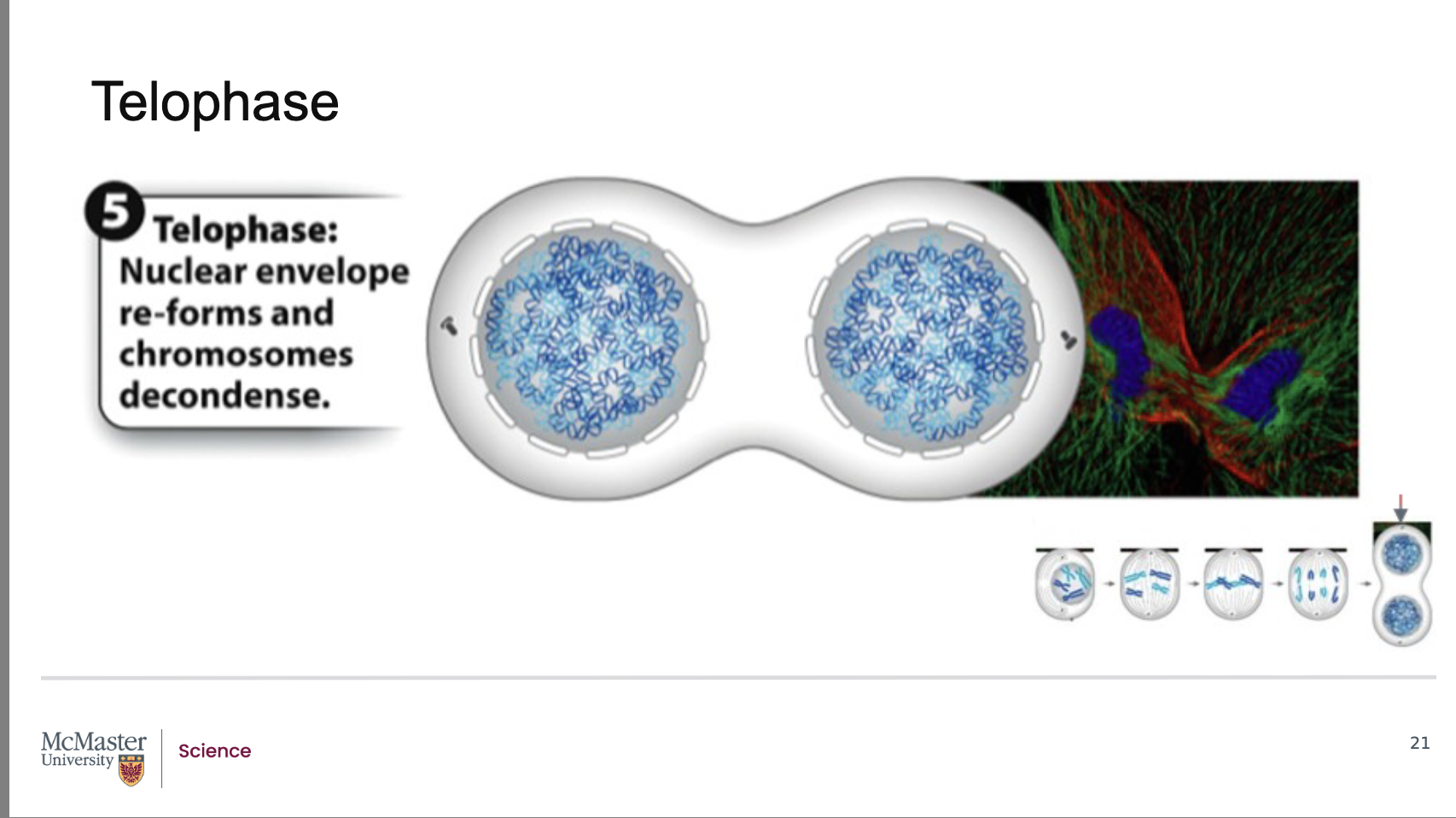
Since the end of anaphase is marked by the equal
segregation of chromosomes at two ends of a dividing
cell, the final stage of mitosis, telophase, is the stage
in which two new daughter nuclei form in the cell.
This occurs because the nuclear envelope reforms
around the chromosomes at the opposite poles of the
dividing cell. At the same time, the chromosomes
begin to decondense and spindle microtubules are
depolymerized (or are broken down). The division of
one nucleus into two genetically identical nuclei
marks the end of mitosis. However, this process of
mitosis, must now be followed up by the division of
the cell into two individual cells
Telophase is the final stage of mitosis, following anaphase.
Since anaphase ends with chromosomes equally segregated at opposite ends of the cell, telophase begins with two complete chromosome sets.
During telophase, two new daughter nuclei form.
The nuclear envelope reforms around each set of chromosomes at the opposite poles.
Chromosomes begin to decondense, reversing the compaction seen earlier in mitosis.
Spindle microtubules depolymerize (break down)
What marks the end of mitosis, and what must occur after telophase?
Back:
The division of one nucleus into two genetically identical nuclei marks the end of mitosis.
After mitosis, the cell must still divide physically through cytokinesis, producing two separate daughter cells.
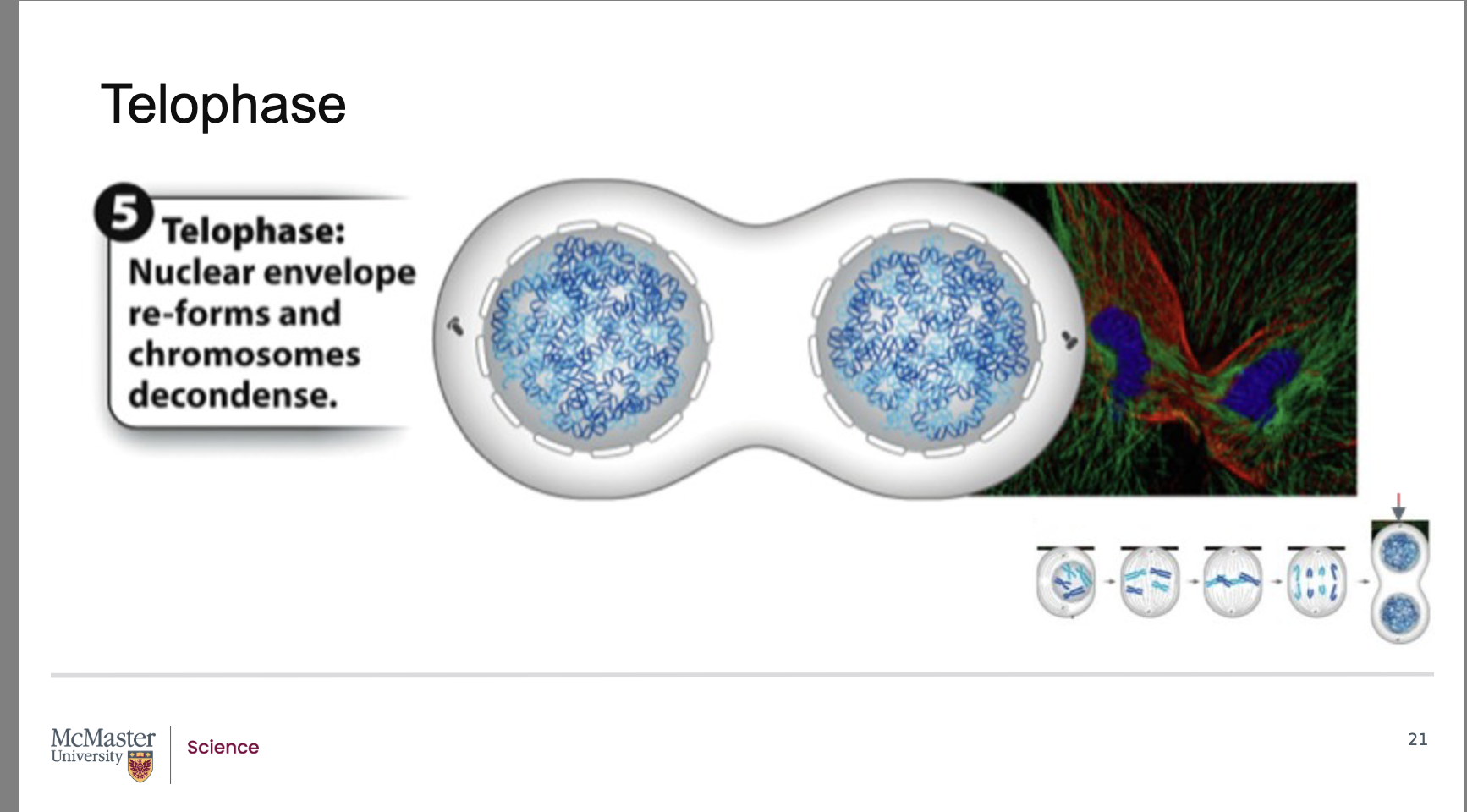
How does cytokinesis occur in animal cells, and when does it begin relative to telophase?
Back:
Cytokinesis is usually well underway by the end of telophase.
Because of this, the two daughter cells become clearly visible shortly after mitosis ends.
In animal cells, cytokinesis begins with the formation of a contractile ring made of motor proteins and bundles of actin fibers along the cell’s midline.
Contraction of this ring creates a cleavage furrow, which deepens to split the parent cell into two distinct daughter cells.
The division of the parent cell into two daughter cells
is usually well underway by the end of telophase. As
a result, the two daughter cells are distinctly apparent
shortly after the end of mitosis due to the process of
cytokinesis (or division of the cytoplasm and
therefore of the cell). In animal cells, cytokinesis
begins with the formation of a contractile ring made
up of motor proteins that contract bundles of actin
fibers along the midline of the cell. This leads to the
formation of a defined cleavage furrow which
separates the cell into two distinct and separate
daughter cells. Interestingly, while the stages of
mitosis are similar across all eukaryotic cells,
differences in cytokinesis can be observed depending
on the dividing cell type. In plant and animal cells,
the process of cytokinesis is distinct and different.
Due to the fact that plant cells have a cell wall, during
cytokinesis, plant cells lay down a newly developed
cell wall along a cell plate region in the middle of the
dividing cell. Once the forming cell wall fuses with
the original cell wall, cytokinesis is then complete in
plant cells
How does cytokinesis differ between plant and animal cells, and why?
Back:
While mitotic stages are similar across eukaryotes, cytokinesis differs depending on cell type.
In plant cells, cytokinesis is different because plants have a cell wall.
Instead of forming a cleavage furrow, plant cells build a new cell wall along a cell plate that forms in the middle of the dividing cell.
Once the new cell wall fuses with the original cell wall, cytokinesis is complete in plant cells.
Why is cell division control important, and what early ideas emerged in the 1970s about regulating the cell cycle?
Back:
Cell division is critical for developmental growth, maintenance, and tissue repair.
In the 1970s, early research suggested there are molecular regulators that control the cell cycle.
A key hypothesis was the existence of a Mitosis‑Promoting Factor (MPF) that drives the transition from G2 to M phase.
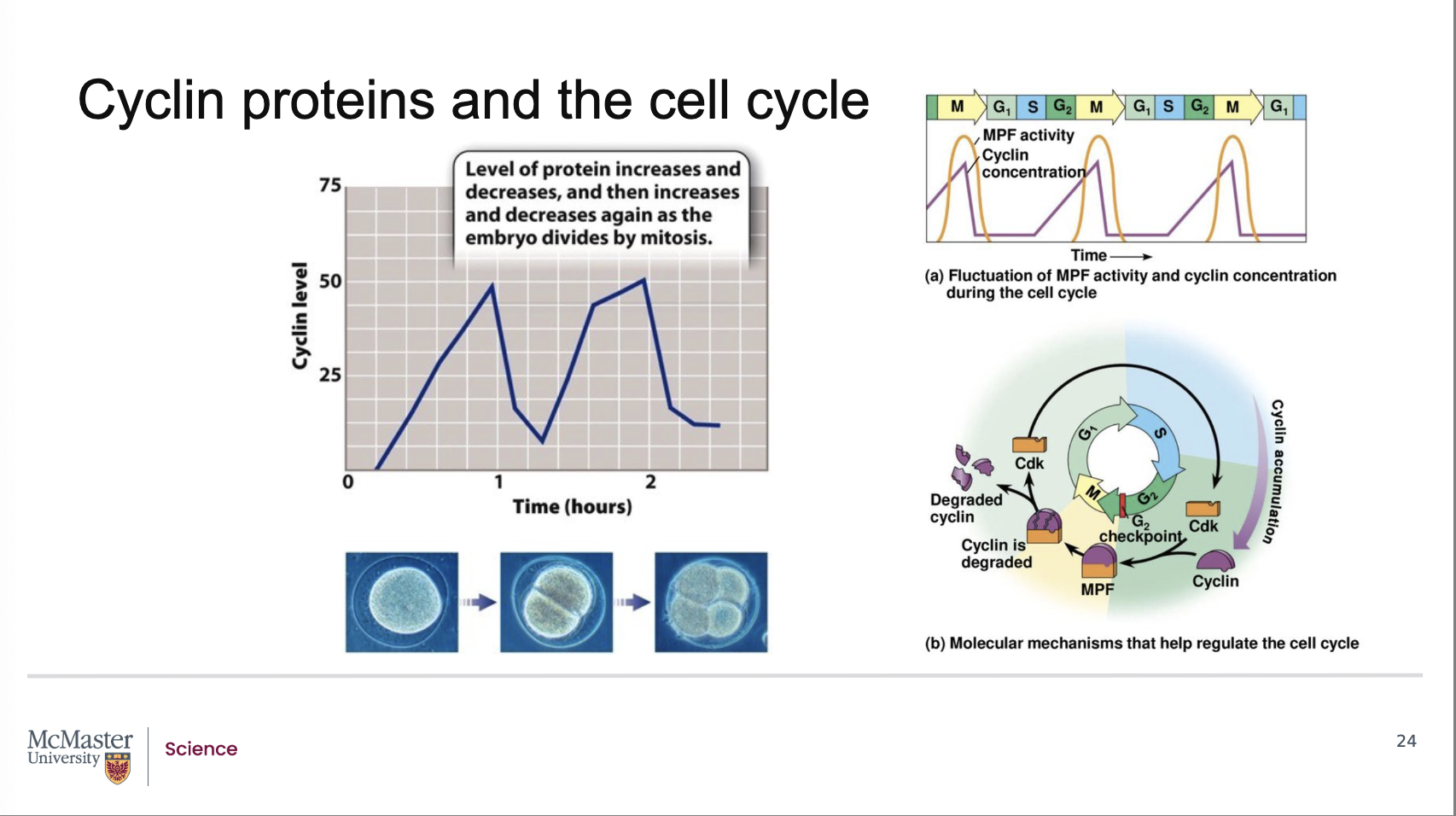
How did Tim Hunt design an experiment to track protein changes during cell division in the 1980s?
Back:
Tim Hunt studied dividing sea urchin embryos.
Added radioactively labeled amino acids, specifically [^35S]-methionine, to eggs so newly synthesized proteins would be radioactively tagged.
Rationale: Label incorporation enables quantitative tracking of new protein synthesis over time in rapidly dividing embryos.
Sampling: Collected embryo samples every 10 minutes.
Detection method: Used gel electrophoresis s (a
procedure that allows for distinct separation of
different protein types) to separate proteins and visualize changes in protein levels as bands.
What did Hunt observe about protein levels on the gels during embryonic cell divisions?
Back:
Most protein bands on the gel became darker over time, indicating increasing levels as development and cell division progressed.
One protein band behaved differently: its intensity oscillated—it increased and then decreased with each subsequent cell division.
What was the oscillating protein called, and why?
Back:
Because the protein’s abundance rose and fell cyclically with each cell division, Hunt and his team named it “cyclin.”
The name reflects the cyclic nature of its synthesis and degradation during the cell cycle.
Hunt and his team found
that most protein bands on the gel became darker as
cell division and embryonic development progressed.
However, one protein band oscillated in intensity. It
was found that this protein increased then decreased
with each subsequent cell division. Due to this cyclic
nature of this protein, Hunt and his team called this
protein cyclin. While they were not aware of the
function of this protein, they suspected that it was
intimately involved with playing some sort of
regulatory role on cell cycle progression
What did Hunt and his team infer about cyclin’s function and the control of the G2→M transition?
Back:
Although the exact function of cyclin was not yet known, its precise oscillation suggested a regulatory role in cell‑cycle progression.
The findings aligned with the concept of a mitosis‑promoting factor (MPF) that triggers G2 to M phase transition—implying cyclin could be a key component or regulator of that control system.
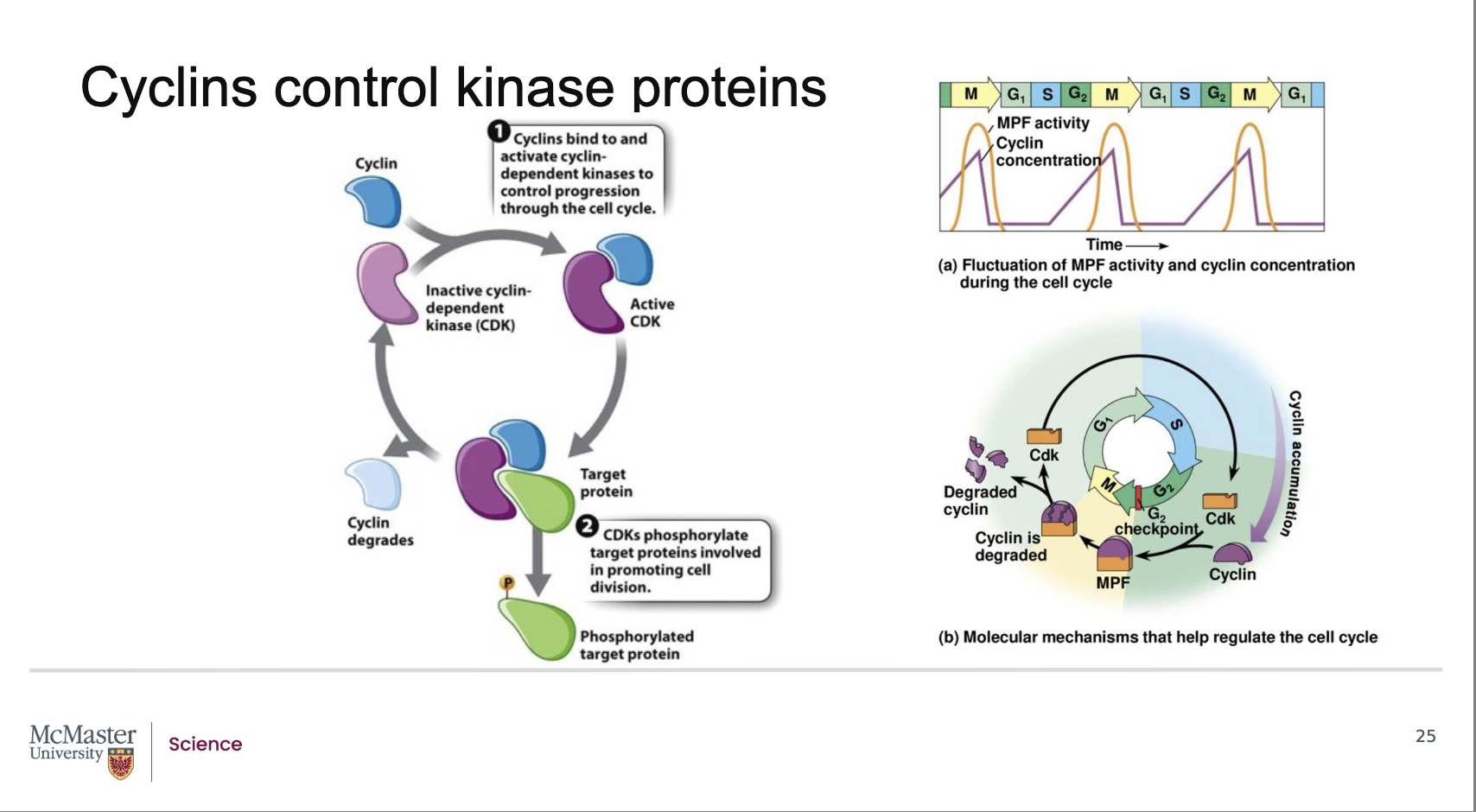
What did Hunt and his colleagues discover about the composition of the mitosis‑promoting factor (MPF)?
Follow‑up work revealed that MPF is made of two components:
A cyclin protein, whose levels rise and fall during the cell cycle.
A cyclin‑dependent kinase (CDK), an enzyme that regulates cell‑cycle transitions.
Together, the cyclin + CDK pair forms the active MPF complex that drives cell‑cycle progression, including the transition from G2 → M phase.
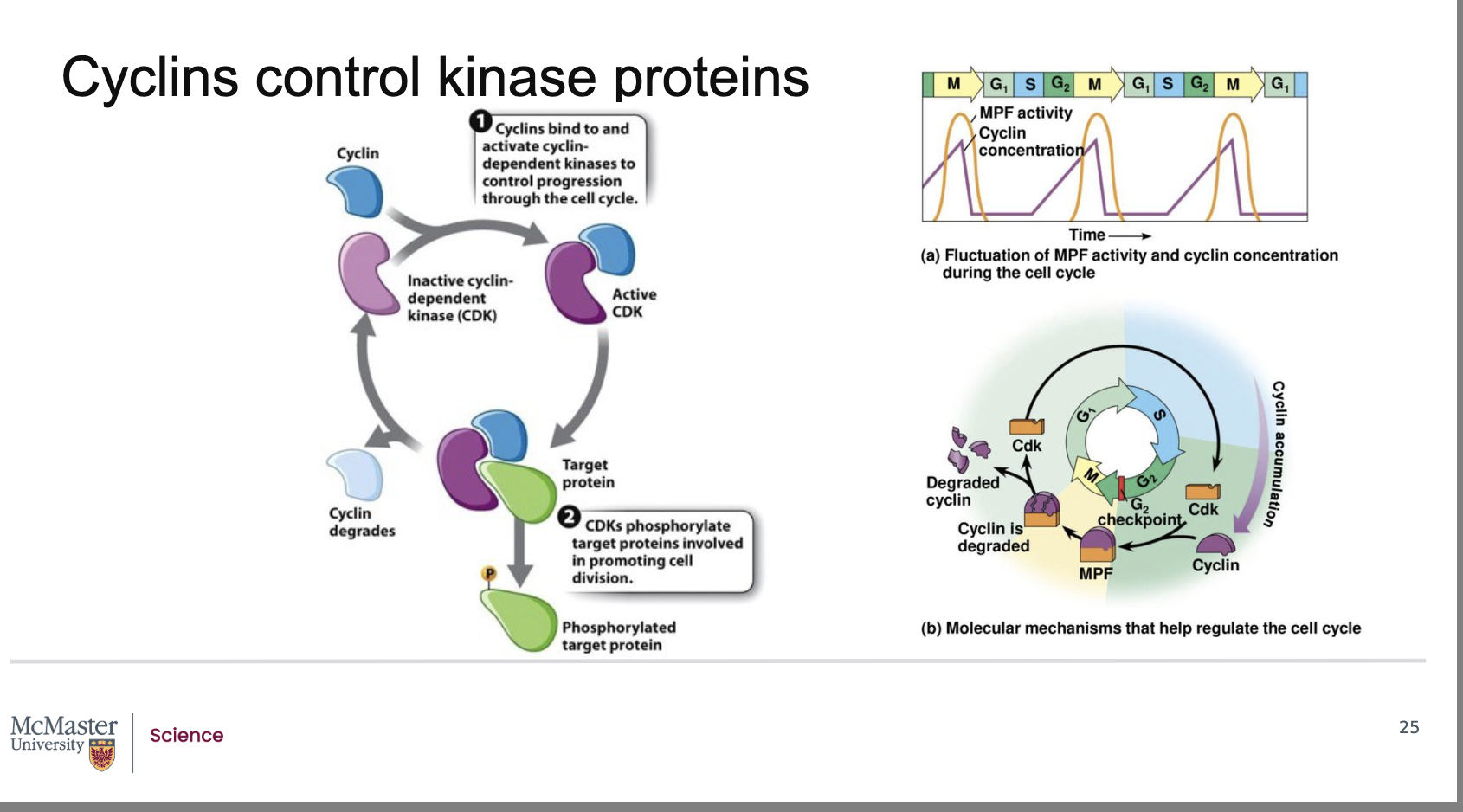
How does cyclin concentration influence the activity of cyclin‑dependent kinases (CDKs)?
Back:
The activity of a cyclin‑dependent kinase (CDK) rises and falls depending on the concentration of its specific activating cyclin protein.
CDKs alone are generally inactive; they require binding to a cyclin to become functional.
Because CDK activity depends directly on cyclin levels, CDK activity naturally oscillates with the cell‑cycle–dependent rise and fall of cyclin concentration.
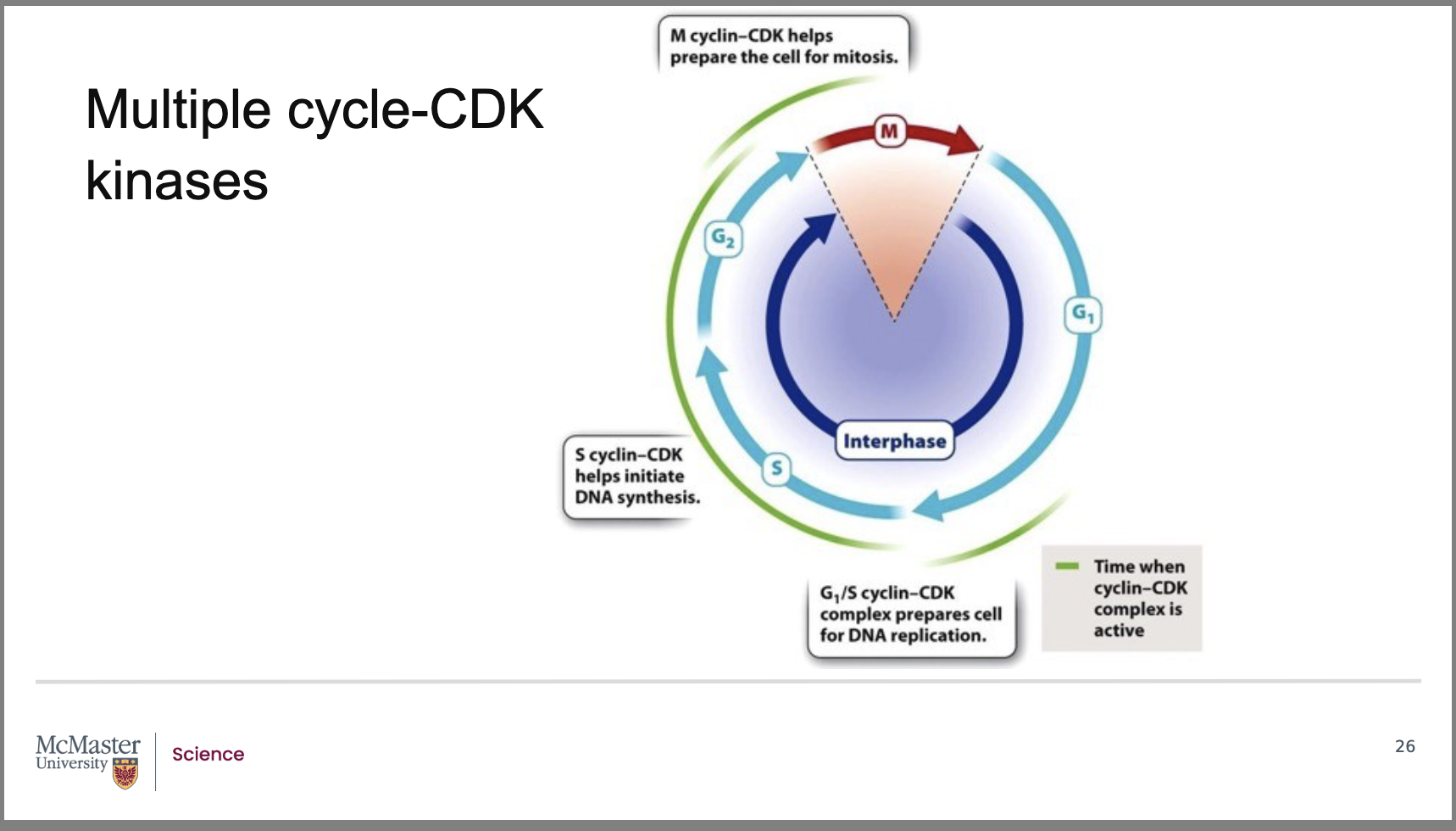
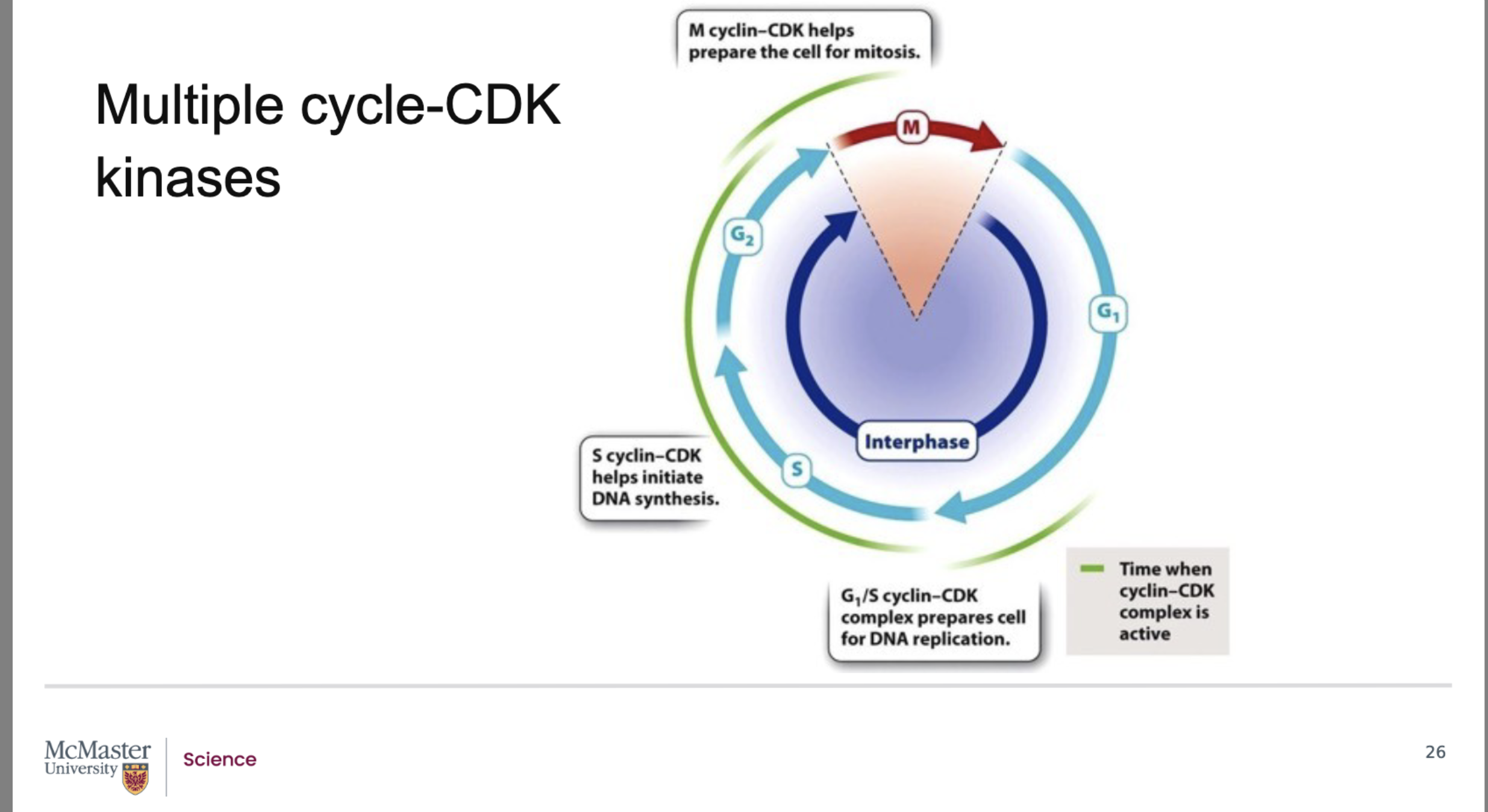
Why are there many types of cyclin–CDK complexes, and what role do they play in cell‑cycle regulation?
Back:
There are many distinct cyclin–CDK complexes.
Each complex regulates a specific stage of the eukaryotic cell cycle.
This specialization ensures that events occur in the correct order and at the correct time (G1 → S → G2 → M).
What do the G1/S and S‑cyclin–CDK complexes control in the cell cycle?
Back:
G1/S cyclin–CDK complex:
Drives the transition from G1 to S phase.
Prepares the cell for DNA replication.
Example: increases production of histone proteins, which are needed to package newly synthesized DNA.
S‑cyclin–CDK complex:
Helps initiate DNA synthesis.
Ensures replication machinery is activated at the correct time.
Front:
What does the M‑cyclin–CDK complex regulate, and how does it facilitate mitosis?
Back:
The M‑cyclin–CDK complex initiates the M phase (mitosis).
It phosphorylates key structural and regulatory proteins that drive mitotic events, including:
Breakdown of the nuclear membrane.
Regulation of microtubule assembly needed to form the mitotic spindle.
These phosphorylation events coordinate the major structural changes required for successful mitosis.
What are cell‑cycle checkpoints, and what roles do they play in regulating the cell cycle?
Back:
Cell‑cycle checkpoints are key regulatory control points that act as cellular surveillance systems.
Their primary purpose is to monitor the accuracy and safety of cell‑cycle progression.
If something goes wrong during a specific phase, checkpoints can block cyclin–CDK activity, halting the cell cycle.
Checkpoints ensure that:
A cell does not enter the next phase prematurely.
Damage can be repaired before the cell continues dividing.
Checkpoints protect cells from replicating damaged DNA, incomplete DNA, or misaligned chromosomes.
What are the three major checkpoints in the eukaryotic cell cycle and what does each monitor?
Back:
G1 DNA Damage Checkpoint (End of G1)
Ensures DNA is undamaged before entering S phase.
Prevents replication of damaged or mutated DNA.
G2 DNA Replication Checkpoint (End of G2)
Ensures all DNA has been completely and correctly replicated before mitosis.
Prevents cells from entering M phase with incomplete or faulty replication.
M‑Phase Spindle Assembly Checkpoint (Before Anaphase)
Ensures every chromosome is attached to a spindle microtubule.
Prevents sister chromatids from separating until all chromosomes are properly bi‑oriented.
What outcomes do checkpoints ensure in terms of DNA integrity and cell‑cycle transitions?
Back:
G1 DNA Damage Checkpoint:
Only undamaged DNA proceeds into S‑phase for replication.
G2 Replication Checkpoint:
Cells enter mitosis only when all DNA has been fully replicated.
M‑Phase (Spindle) Checkpoint:
Cells complete mitosis only when all chromosomes are properly attached to spindle fibers.
These checkpoints prevent genomic instability, mutations, and improper chromosome segregation.
How do checkpoints mechanistically pause the cell cycle?
Back:
Checkpoints detect DNA damage, incomplete replication, or chromosome misalignment.
In response, they trigger pathways that block cyclin–CDK complex activity, which is required for progression to the next phase.
By inhibiting cyclin–CDK activity, checkpoints pause cell division, giving the cell time to:
Repair DNA,
Finish replication,
Correct spindle attachment,
before continuing.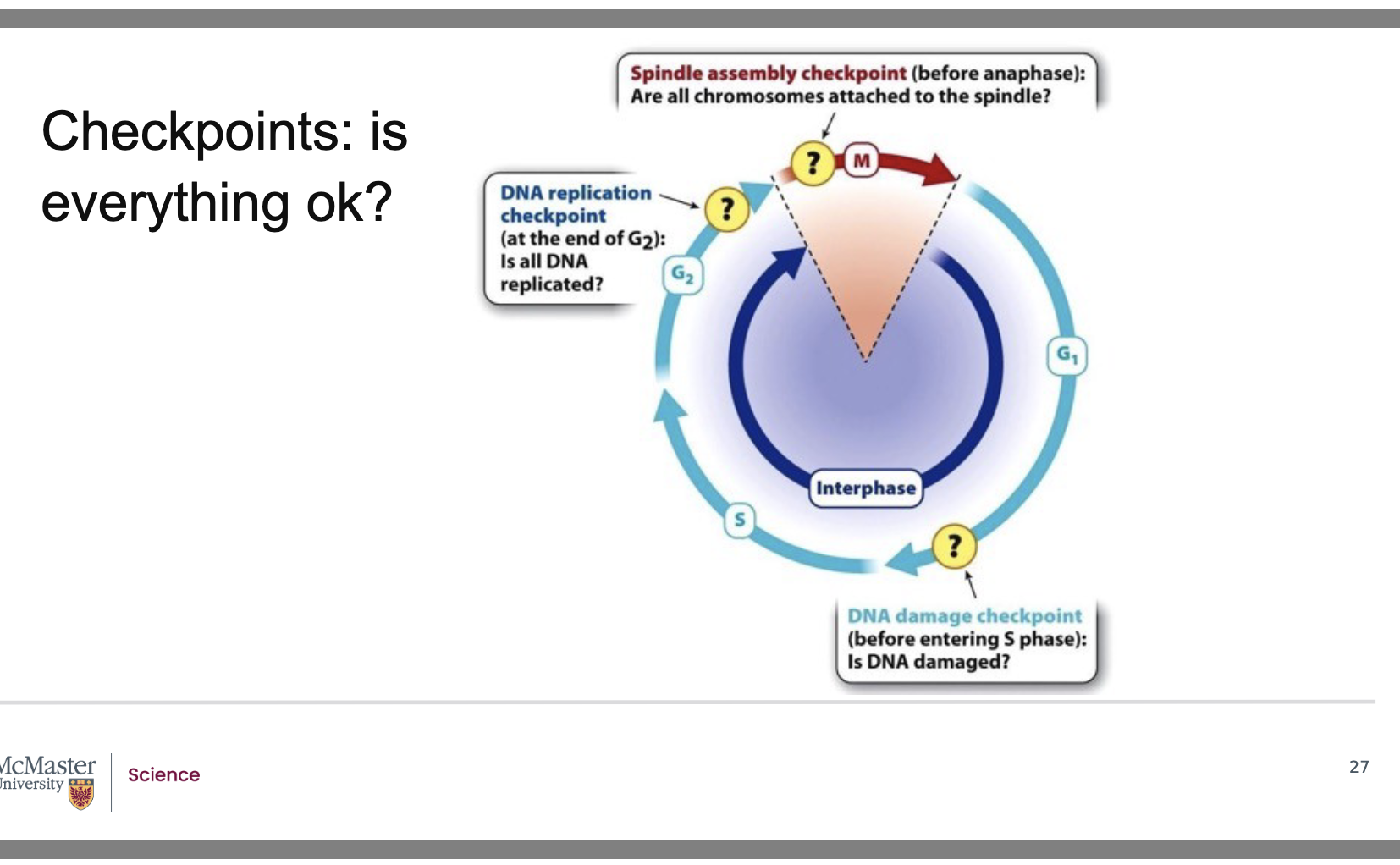
This ensures that cells only move forward when all conditions are correct.
What triggers the DNA damage checkpoint and what is its purpose?
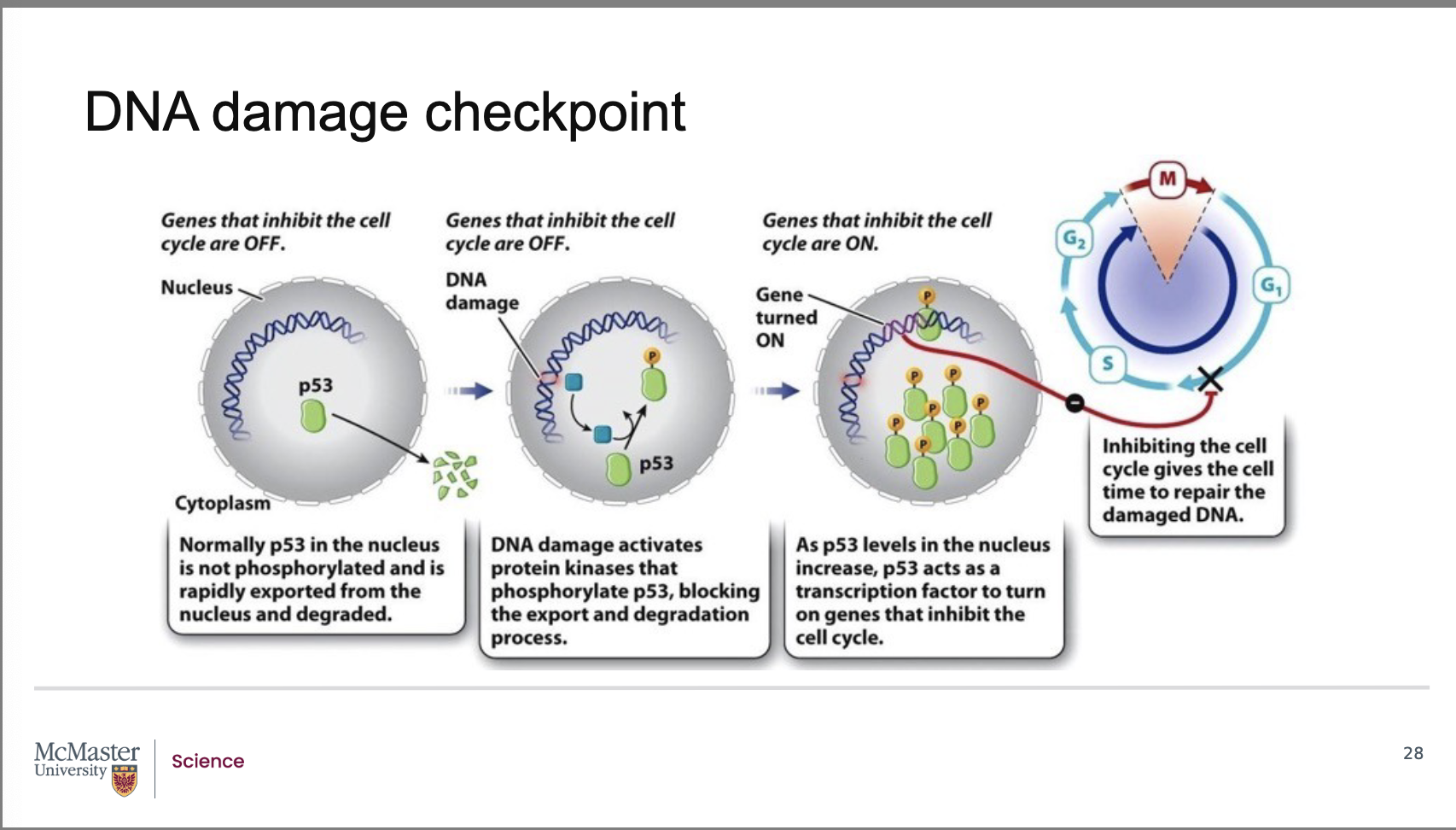
Back:
The DNA damage checkpoint is activated when structural damage occurs to DNA, especially double‑stranded breaks in the phosphodiester backbone.
Under normal conditions, genes that inhibit the cell cycle are turned off, allowing normal progression.
When DNA damage is detected, the checkpoint serves as a mechanism to block cyclin–CDK activity, thereby pausing the cell cycle until repairs can be made.
How is p53 activated during the DNA damage checkpoint, and what is its normal behavior?
Back:
p53 is normally present at very low levels in the nucleus.
Under normal conditions:
p53 is exported from the nucleus
p53 is rapidly degraded
When DNA damage occurs:
Specific protein kinases phosphorylate p53.
Phosphorylation prevents its degradation.
This allows p53 to accumulate in the nucleus.
Activated p53 acts as a transcription factor, turning on genes that inhibit the cell cycle.
How does p53 stop the cell cycle after DNA damage, and which cyclin–CDK complex is affected?
Back:
p53 induces transcription of a CDK inhibitor protein.
This inhibitor binds to and blocks the activity of the G1/S cyclin–CDK complex.
As a result, the cell cycle is arrested in the G1 phase, preventing the damaged DNA from entering S phase.
This pause allows time for the cell to repair its damaged DNA before replication begins.
When is the spindle assembly checkpoint active, and what does it monitor?
Back:
The spindle assembly checkpoint becomes active as early as prometaphase.
It monitors how well sister chromatids are attached to microtubules of the mitotic spindle at their kinetochore regions.
Its goal is to ensure that each sister chromatid is properly attached before anaphase begins.
How do cells detect improperly attached chromatids, and what signals are produced?
Back:
Unattached kinetochores generate a biochemical “wait” signal.
This signal recruits spindle assembly checkpoint proteins to the kinetochore.
These proteins are activated when the kinetochore experiences a lack of tension—a sign that attachment to microtubules is incomplete or incorrect.
As long as any kinetochore is not attached or not under tension, the checkpoint blocks progression into anaphase.
How is the spindle checkpoint resolved, and how does this allow sister chromatid separation?
Back:
Once every sister chromatid is correctly attached to a spindle microtubule, tension is generated at all centromeres.
This tension causes the checkpoint proteins to be removed from the centromere region.
Removal of these proteins activates separase, a specialized enzyme.
Separase breaks the attachments between sister chromatids, allowing them to separate and enter anaphase.
What happens to genes that inhibit cell‑cycle progression when the cell is not experiencing DNA damage?
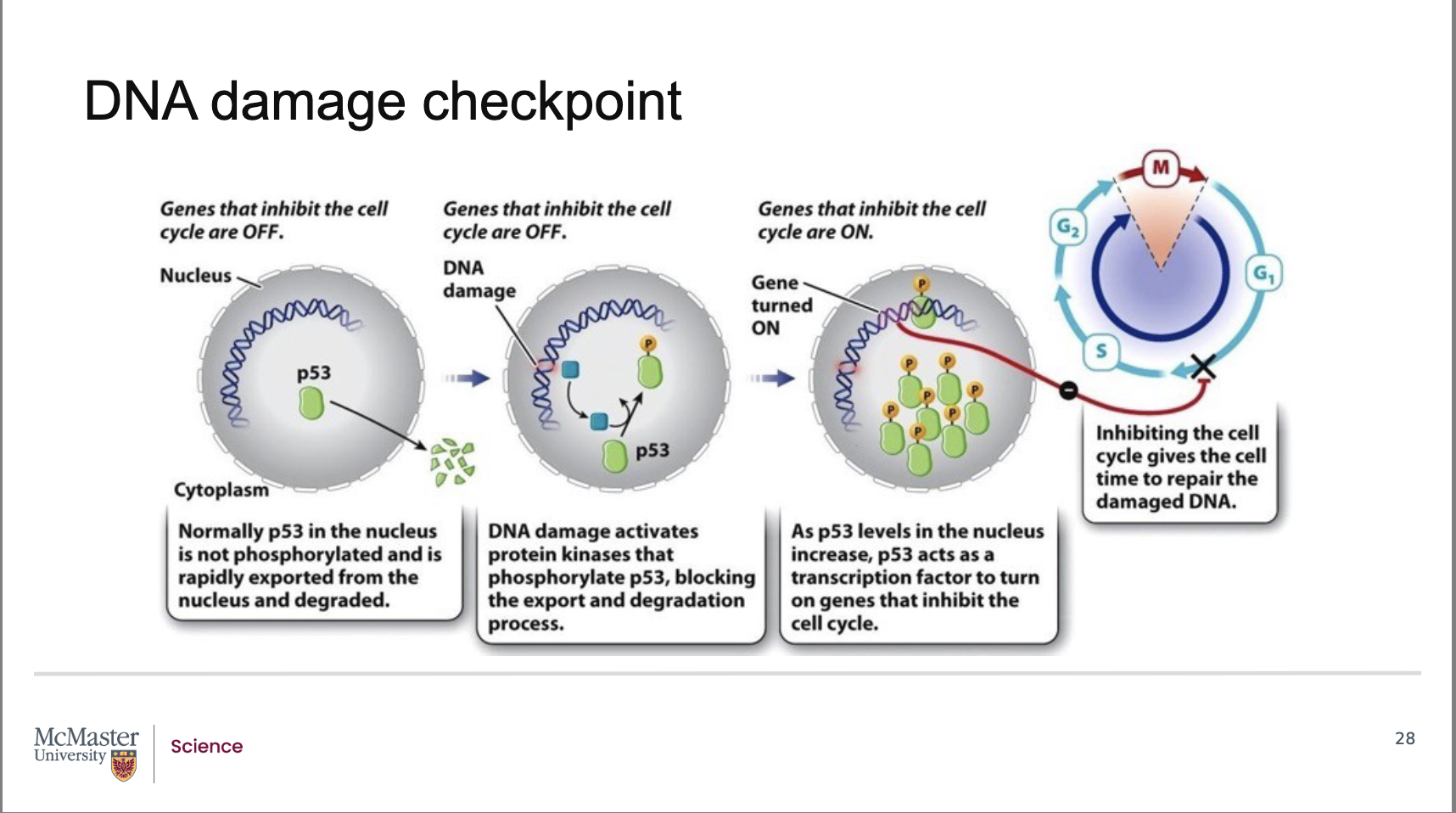
Under normal, undamaged conditions, the genes that inhibit the cell cycle are turned off.
This allows the cell cycle to proceed normally through G1 → S → G2 → M.
Only when DNA damage is detected are these inhibitory genes turned on through p53 activation
What specific type of DNA damage activates the checkpoint machinery?
Back:
The checkpoint is activated by structural damage to DNA, including:
Double‑stranded breaks
Breaks in the phosphodiester backbone of DNA
These are serious lesions that must be repaired before DNA replication or mitosis.
What proteins first respond to DNA damage, and what do they do to p53?
Back:
When DNA damage occurs, specific protein kinases detect the damage.
These kinases phosphorylate p53.
Phosphorylation prevents p53 from being exported and degraded, allowing p53 levels to rise
Why does p53 accumulate in the nucleus when phosphorylated?
Back:
Normally, p53 is:
Produced at low levels
Exported from the nucleus
Rapidly degraded
Phosphorylation blocks p53's export and degradation, allowing it to accumulate inside the nucleus.
This buildup is required for p53 to function as a transcription factor.
Once activated, how does p53 stop the cell cycle?
Back:
Activated, phosphorylated p53 acts as a transcription factor.
It turns on genes that inhibit the cell cycle.
These genes encode a CDK inhibitor protein.
Which CDK complex is blocked during the DNA damage checkpoint, and what is the effect?
Back:
The CDK inhibitor produced in response to p53 binds to and blocks the G1–S cyclin–CDK complex.
This arrests the cell in G1, preventing entry into S phase.
The pause gives the cell time to repair damaged DNA before replication begins.
At what stage is the spindle assembly checkpoint active, and what does it monitor?
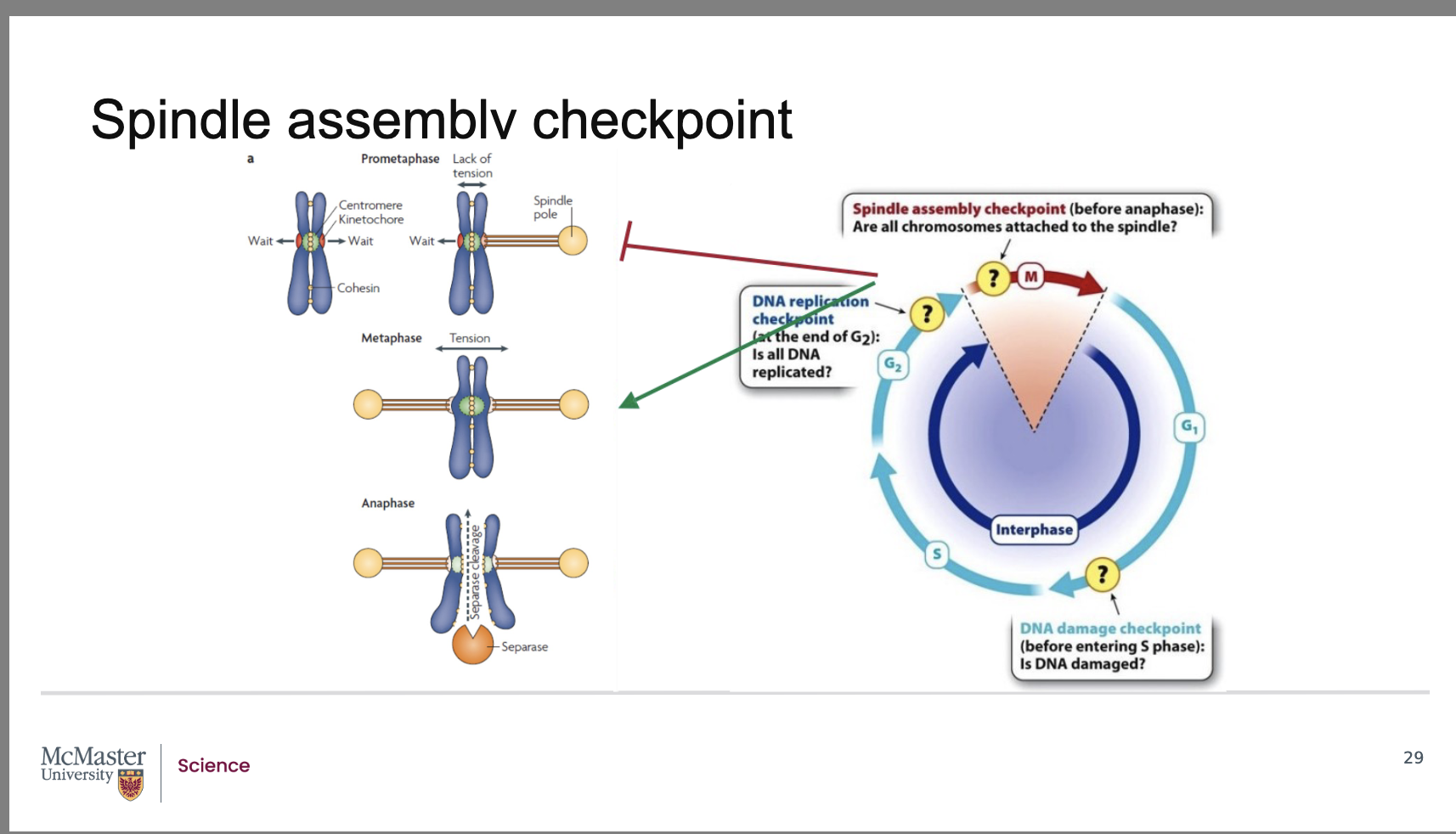
Back:
The spindle assembly checkpoint becomes active as early as prometaphase of mitosis.
It monitors how securely sister chromatids are attached to microtubules of the mitotic spindle at their kinetochore regions.
This checkpoint ensures proper chromosome–spindle microtubule attachment before allowing the cell to proceed to anaphase.
How does the cell detect improper kinetochore attachment, and what signals are produced?
Back:
If a sister chromatid has an unattached kinetochore, that kinetochore produces a “wait” signal.
This signal recruits spindle assembly checkpoint proteins to that kinetochore.
These checkpoint proteins become activated when there is a lack of tension in the centromere region, which indicates:
The chromatids are not properly attached to spindle microtubules
OR attachment is incomplete or incorrect
While any kinetochore remains unattached or under low tension, the checkpoint blocks the progression from metaphase to anaphase.
How is the spindle assembly checkpoint satisfied, and what enzyme triggers chromatid separation?
Back:
The checkpoint is satisfied only when each sister chromatid is correctly attached to a kinetochore microtubule.
Proper attachment generates tension across the centromere, which removes checkpoint proteins from the kinetochore.
Once these checkpoint proteins are removed, separase, a specialized enzyme, becomes active.
Separase breaks the protein connections holding sister chromatids together, allowing them to separate and move into anaphase.