Module 3: DNA, Proteins, and Amino acids
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What are the individual units DNA is made of
(deoxy)-nucleotides
What are the 3 components of a nucleotide
5 carbon sugar
deoxyribose
missing a hydroxyl group at 2’ carbon
phosphate group
1 of 4 possible nitrogenous bases
where is the nitrogenous base always attached on the sugar
1’
where is the phosphate group located
on the 5’ of one sugar, and 3’ of other nucleotide
what bonds are between nucleotides
phosphodiester bonda
what are the bonds between the bases of DNA
hydrogen bonds
what are the base parings and how many h-bonds between them
A → T: 2
C → G: 3
What 2 family names do the base pairs fall under and why
pyrimidines
thymine and cytosine
have single ring structure
purines
adenine and guanine
double ring structure
Geometry is the same!
why can’t other bases pair (ex: G and T)
angles in which bases attach are not correct
do not have same geometry + angles
hence cannot form strong base bonds
disturb the helix structure
what other interactions are occurring with the base pairs
pi-pi interactions when the base pairs aromatic rings stack next to each other and share electron probabilities
what are the two repeating and alternating spaces + what do they do
major and minor grooves
act as base pair recognition and binding sites for proteins
due to patterns of hydrogen bond acceptors and donors that proteins can interact with in the groups
allow for proteins to act in a seq or non-seq manner → carry out specific tasks
major
contains bp specific info
minor
largely bp non-specific
Differences between RNA
RNA:
Uracil instead of thymine
ribonucleic acid
has O at 2’ of ribose sugar
DNA:
deoxyribonucleic acid
H instead of hydroxyl group at 2’ position of ribose sugar
3’ carbon contains linkage to hydroxyl group
important in DNA replication
5’ carbon linked to phosphate moiety
part of a molecule that is given a name because it is identified as a part of other molecules as well.
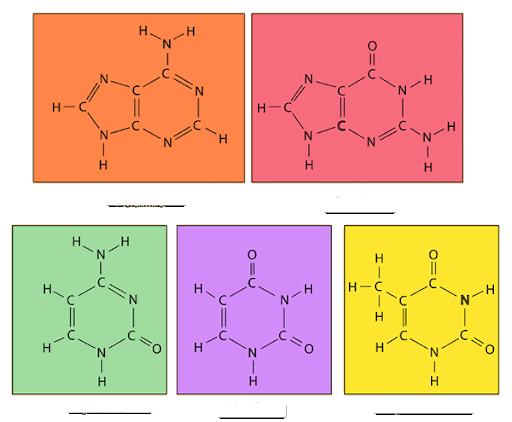
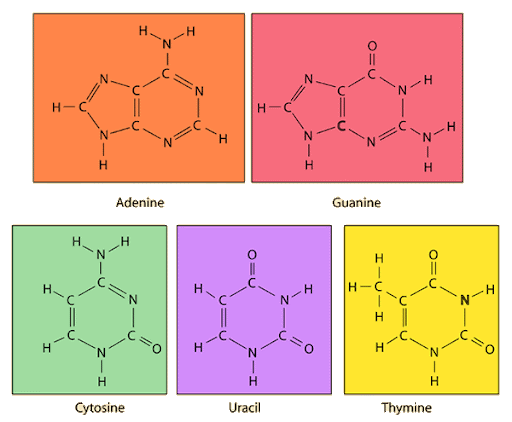
identify the nitrogenous bases:
Why are nucleic acids negatively charged?
phosphate group is a strong acid with pKa of 1
makes dna and rna acidic
in aq environment like cells with a pH of around 7, the monomeric unit becomes the acid, hence will donate a proton (deprotonation)
will then carry a net negative charge
RNA hydrolysis
both DNA and RNA backbones undergo slow-enzymatic hydrolysis of phosphodiester bonds
RNA is quicker than dna due to the 2’ C position on the ribose ring having a more reactive hydroxyl group
in test tube: under alkaline conditions
alkaline conditions = presence of OH ions
OH group of the 2’ C directly involved
cyclic 2’,3’-monophosphate nucleotides are first products
second products: mixture of 2’-and 3’-nuceloside monophosphates
what is a DNTP and why are they needed
dNTP (deoxy nucleoside triphosphate) is a monomer that contains an energy packed triphosphate moiety at the 5’ end
used to couple the condensation reaction between 3’ OH and a 5’ hydroxyl of a deoxynucleoside monophosphate
condensation of a adding a deoxynucleotide to a polynucelotide chain is a thermodynamically unfavoured rxn
hence dNTP is reacted with water to form a dNMP and a free pryophosphat
v favourable rxn
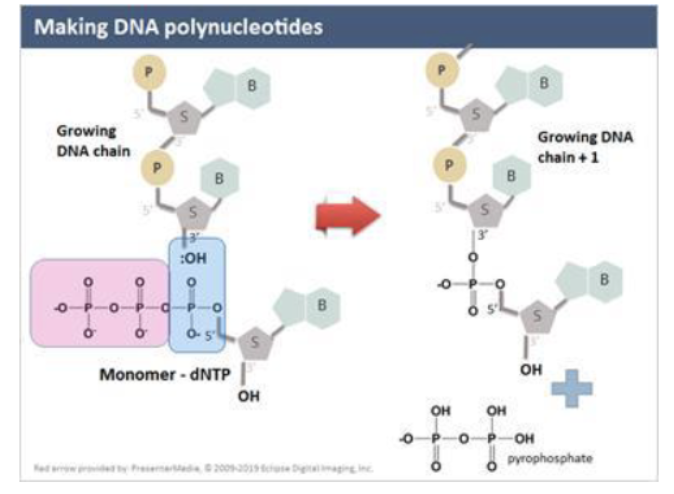
How are DNA polynucleotides made
poly = many, nucleotides bound together
a chain of nucleotides typically has one 5’ C with a free phosphate group and a free 3’ OH group
adding deoxynucleotide:
5’ end of dNTP is added to the 3’ OH of the growing DNA polynucleotide chain
covalent bond to the phosphorus atom of the free nucleotide closest to the 5’ O
bond between the first phosphorus atom and the oxygen atom linking it to the next phosphate group breaks
the pyrophosphate molecule is readily hydrolyzed (split w addition of water) releasing a large amts of E → drives rxn fwd
all these rxns are coupled together
end up w a negative dG of overall adding a monomer to a growing DNA chain
Why is DNA antiparallel
one stand goes 5’ → 3’, other goes 3’ → 5’
defined through the numbering of the ribose sugar
if the free phosphate group that is on the fifth carbon and the free OH group on the 3rd carbon that dictates
difference between nucleoside and nucleotide
nucleotide: consists of a nucleobase and ribose sugar, no phosphate
nucleotide:consists of a nucleotide, ribose sugar, and 1-3 phosphate groups
what are proteins made of
amino acids → polypeptide chain → protein
what is the basic structure of an amino acid
carboxylic acid attached to a carbon atom called the alpha carbon (where all the components of the amino acid are attached)
amine attached to alpha carbon
where monomeric form of the protein polymer gets name
alpha carbon attached to R group (side chain)
contains beta carbon where a lot of other groups attach that differentiates the amino acids
what is chirality
any molecule whose reflection in a mirror cannot be superimposed (reflected back)
most common form occurs when a C-atom is attached to 4 non-identical group
only one of the 20 amino acids is achiral (due to H as the R group)
What is L chirality
chiral form of amino acids
prefix L means levo
L-amino acid rotates polarized light to the left
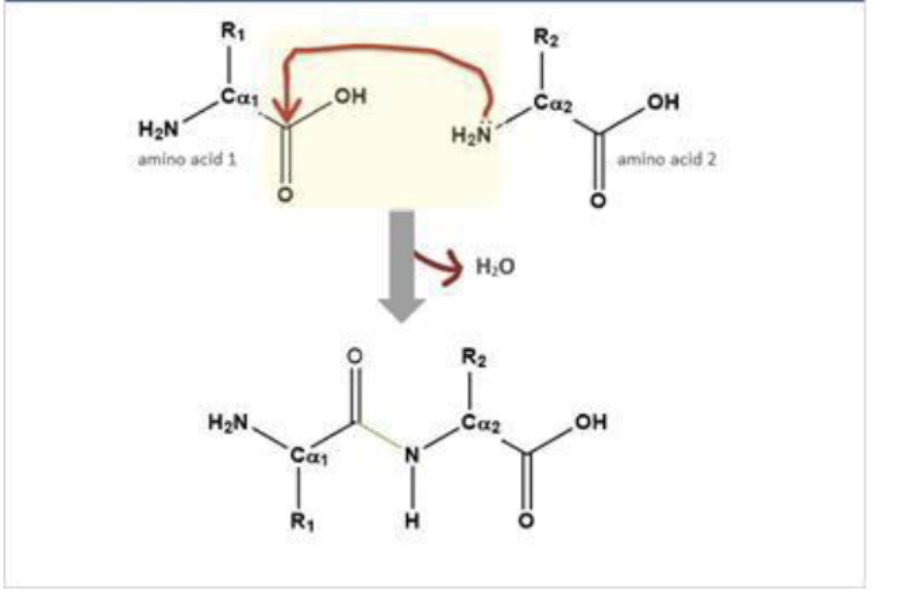
How are peptide bonds formed and what are they?
link amino acids together (covalent bond)
formation:
nucleophilic addition-elimination rxn between the carboxyl and amino groups of adjacent aminos acids
initiated by a nucleophilic attack from the e- pair on the amino group of aa2 to the carbonyl carbon on aa1 (nucleophilic addition)
gives a water molecule (elimination)
why is a peptide bond rigid
it is a planar rigid bond
there is not a lot of rotation around it
due to the fact that it has double-bond like character
kept in a rigid planar state by resonance delocalization of its electrons
also means the peptide N is slightly positively charged and carbonyl O is slightly neg charged
DOES NOT MEAN THE ENTIRE POLYPEPTIDE is rigid
still 2 freely rotating single bonds p
backbone features of a polypeptide chain
nitrogen (alpha carbon) - carbonyl carbon - nitrogen-Ca-carbonyl carbon (x2)
directionality:
amino group at one end and other end has carbonyl carbon
beginning called: amino or N terminal
end called: carboxyl or C terminal
can write out the amino acid seq of a polypeptide chain just by referring to the unique side chain (R group)
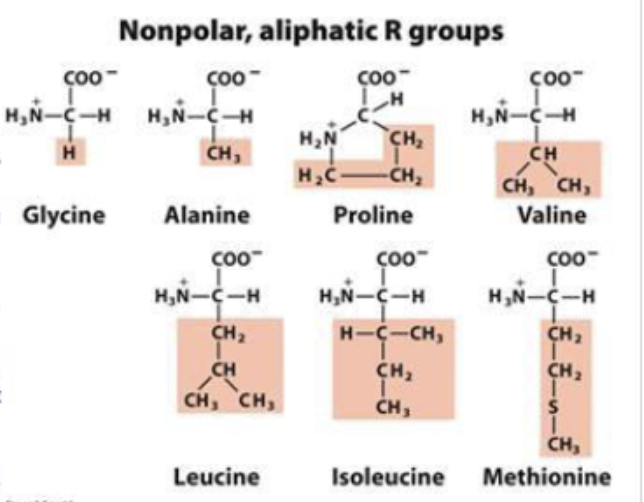
What does it mean if the amino acid is non-polar (due to the side chain)
non-polar and hydrophobic
stabilize proteins structure through the hydrophobic effect
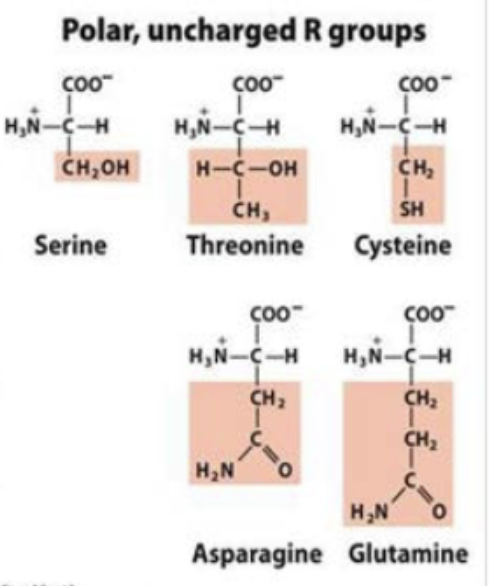
polar uncharged R groups
have polar side chains that can form H-bonds with water or other polar molecules
ser and thr found on surfaces of proteins
cys sidechain can form h-bonds w water or other polar molecules
asn and gln very polar but have non ionizable un-charged polar side chains
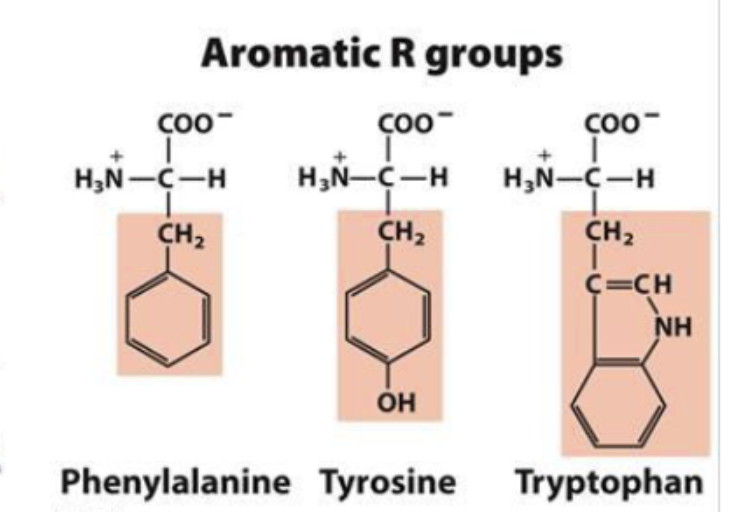
aromatic R groups
hydrophobic and aromatic
absorb UV light at wavelength of 280nm
can use to measure protein concentrations
phe most hydrophobic amino acid
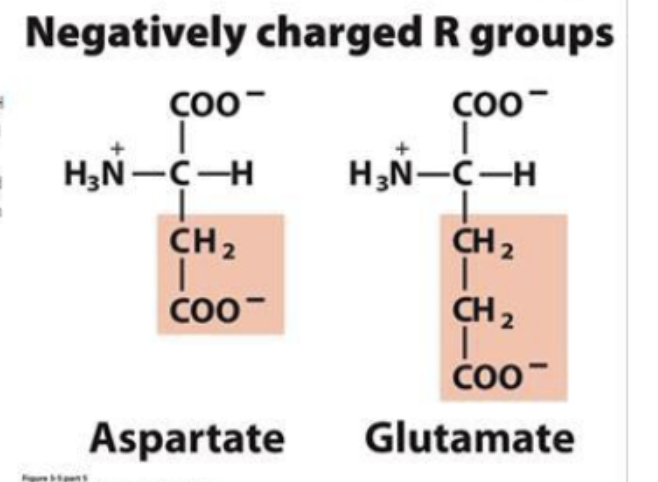
negatively charged R groups
amino acids that carry negative charges at pH 7
hydrophillic and typically found on surfaces of proteins in contact w aq envion
positively charged R groups
basic in side chain character
very polar and usually found on the exterior of proteins or in the substrate binding clefts of enzymes