coordination chemistry II: bonding, spectrochemical series
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
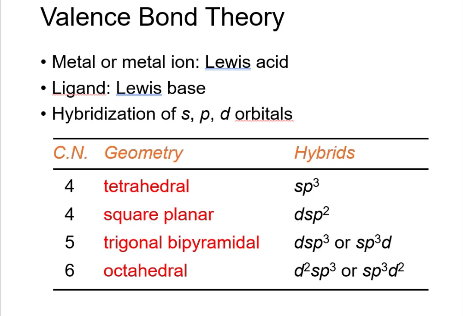
Valence bond theory:
Metal or metal ion: lewis ___
ligand: lewis ___
Hybridization of _, _, and _ orbitals
acid, base, s,p, and d
tetrahedral hybridization
sp3
square planar hybridization
dsp2
trigonal bipyramidal hybridization:
dsp3 or sp3d
octahedral hybridization:
d2sp3 or sp3d2
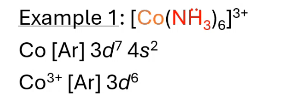
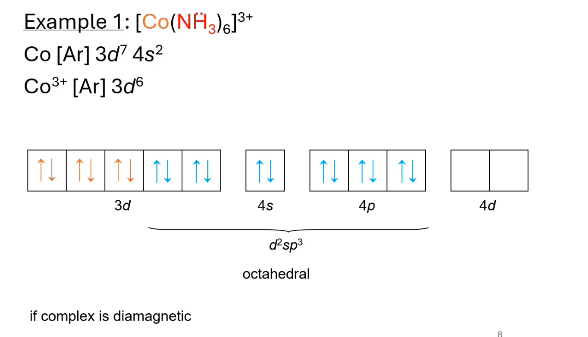
what hybridization? what does valence electron configuration look like? paramagnetic or diamagnetic?
octahedral (d2sp3), diamagnetic
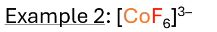
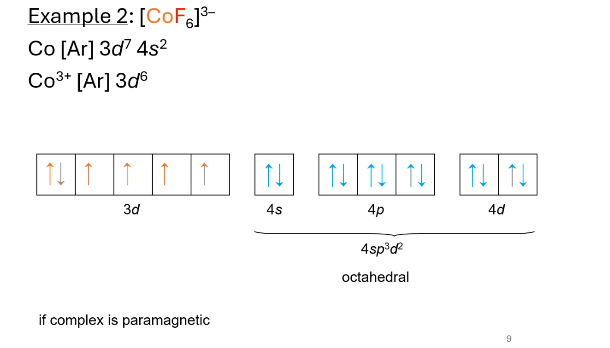
what hybridization? what does valence electron configuration look like? paramagnetic or diamagnetic?
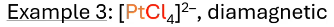
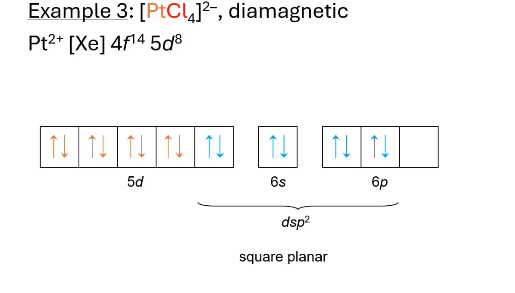
what hybridization? what does valence electron configuration look like? paramagnetic or diamagnetic?
Crystal field theory: Three types of interactions are considered. what are they?

the more electron-electron repulsion you have the ____ in energy it is
higher
the repulsion between ligand lone pairs and the d orbitals on the metal results in a ____ of the energy of the d orbitals
splitting
electrons in lower d orbitals (?) is canceled out by the destabilizing effect of the electrons in the upper orbitals (?)
t2g, eg
(crystal Field Stabilization Energy) CFSE=?
(#e x0.4∆0) - (#e x 0.6 ∆0)
In an octahedral complex, ___ and ____ have the highest electron-electron repulsion because the orbitals directly overlap with the incoming ligand, sit along axes, higher in energy.
dx2-y2 and dz2
in an octahedral complex, dx2-y2 and dz2 have the highest electron-electron repulsion because ?
the orbitals directly overlap with the incoming ligand, sit much higher in energy
in an octahedral complex, __, __, and __ orbitals sit much lower in energy (less electron-electron repulsion) because they do not sit directly along the axes
dxy, dyz, dxz
in an octahedral complex, dxy, dyz, dxz sit in the ___ orbital (lower in energy)
t2g
in an octahedral complex, dx2-y2 and dz2 sit in the ___ orbital (higher in energy)
eg
what is crystal field stabilization energy
value of energy required in order for lower energy orbitals to get back to where it wants to be before the ligands came in
the more electrons in the lower orbitals, the ____ the CFSE
greater
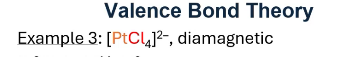
2 electrons per chlorine, they each have their own electron pair through covalent bonding (VBT)
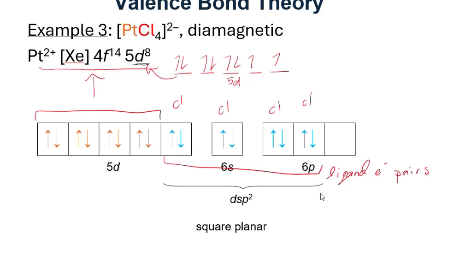

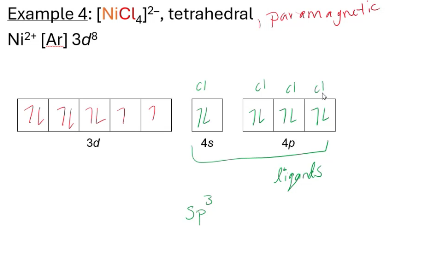
everything need to know about VBT

complete this using crystal field theory

What is spin pairing energy
the energy required to place two electrons with opposite spins in the same orbital , overcoming the repulsion between them
if pairing energy is ____ than delta, the complex is high-spin
higher
if pairing energy is lower than delta, the complex is ___
low-spin
low-spin states are associated with?
strong field ligands
high-spin states are associated with?
weak field ligands
in a tetrahedral field, which orbitals have the highest and lowest electron-electron repulsions? what is the highest energy orbital called? the lowest?
highest: dxy, dyz, dxz
lowest: dx2-y2 and dz2, opposite of octahedral complex
t2, e
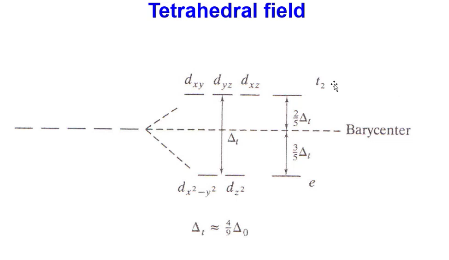
Tetrahedral fields can have ___.
distortion
factors affecting magnitude of CFSE:
Geometry of the field:
Tetrahedral are aways ___ or __
weak field or high-spin
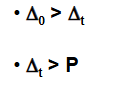
factors affecting magnitude of CFSE:
∆0~ ?
square planar can be either __ or __
high or low-field
factors affecting magnitude of CFSE:
charge on metal ion - greater the charge, ___ the magnitude of ∆
greater the charge, on more ____ are pulled, ___ split
greater (ex: +3 > +2)
metal ligands, greater
factors affecting magnitude of CFSE:
greater the size, ___ the magnitude of ∆
ligand experience less _____ for a large metal ion
greater (ex: Mo3+ > Co3+), steric hindrance
factors affecting magnitude of CFSE:
relative ability of ligand to split: spectrochemical series
goes from high spin to low spin
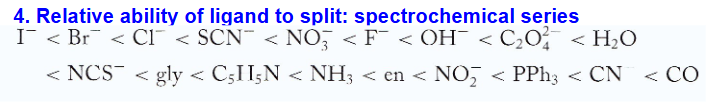
greater splitting means ___ bond
stronger
splitting is was reveals colors in ____
spectrochemical series
crystal field theory cannot explain the?
spectrochemical series
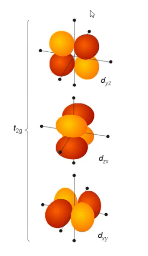
these are non-bonding orbitals because
they are not aligned on the axes, no significant overlap
Ligand field theory:
the ___ set of orbitals is non-bonding, and the __ set of orbitals is antibonding
t2g, eg
this is just the octahedral complex
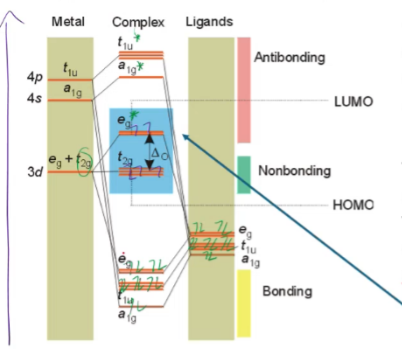
Blue complex emits ___ and vice versa
orange
red complex emits ___ and vice versa
green
Violet complex emits ___ and vice versa
yellow
The moment resulting from “electron spin”: spin-only magnetic moment, ___
the moment resulting from orbiting: orbital magnetic moment, __
μs, μL
μs =?


know low spin wont happen because of Cl ligand spectrochemical series

if magnetic moment is 5.8, what is the oxidation state?
5.8 is close to 5.92 (5 unpaired electrons) so could be d5, so oxidation state could be +3
The colors exhibited by most
transition metal complexes arises from the _____. As electrons transition from the lower t2g set to the eg set, _____ range is absorbed.
splitting of the d orbitals, light in the visible
∆o increases with increasing ____ on the metal and going ___ a group of metals.
oxidation number, down
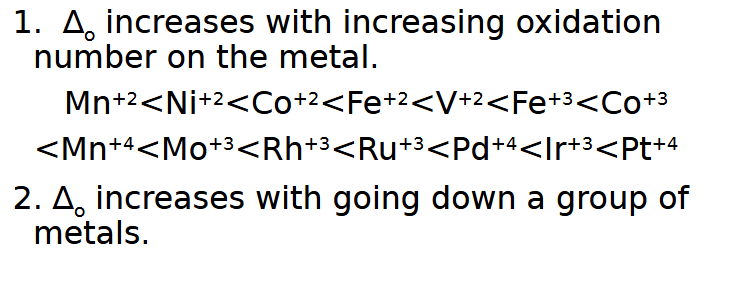
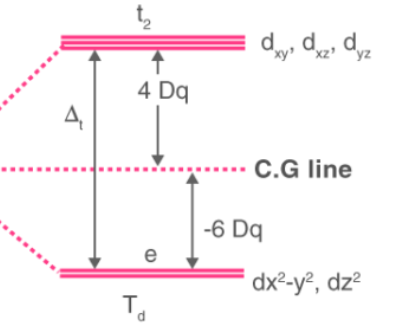
which geometry describes this d-orbital splitting energy diagram
tetrahedral
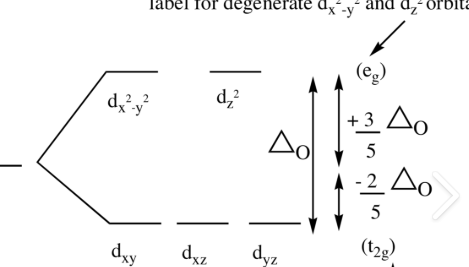
which geometry describes this d-orbital splitting energy diagram
octahedral
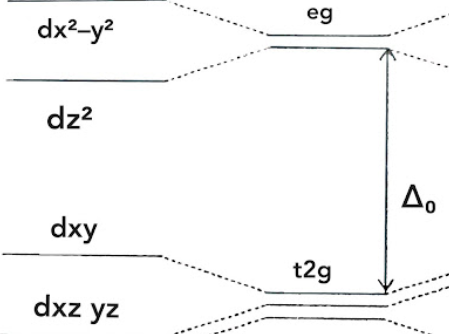
which geometry describes this d-orbital splitting energy diagram
Jahn-Teller elongation
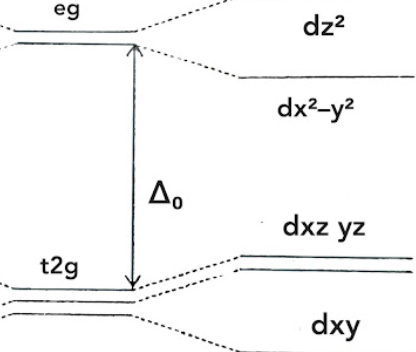
which geometry describes this d-orbital splitting energy diagram
Jahn-Teller compression

Find n,