chemistry/ organic compounds
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
type of bond
low/high melting pt in organic compounds?
covalent bond
low melting, boiling pt
can undergo combustion
what is a hydrocarbon
an organic compound consisting only of hydrogen and carbon atoms without any functional groups.
what reduces and what increases the oxidation number?
H reduces (-1)
every heteroatom (N,O,S…) increases
C leaves it unaltered
what kind of bond do the molecules in alkanes form (or any single bond)?
o (sigma) bond
what kind of arrangment do sp3 orbitals have?
tetrahedral
what kind of p atomic orbitals form pi molecular orbitals to form double (sp2) or triple (sp) bonds?
non-hybridized p atomic orbitals
why do organic compounds enter reactions?
single bonds are saturated double and triple bonds are unsaturated so the goal is to have as many single bonds to obtain the most saturated compounds.
what defines the properties of the organic molecule and adds reacticity?
functional groups
HYRDOCARBONS DONT MIX WITH WATER
just dont forget!! they are also composed of apolar bonds and they can be linear, branched or cyclic
why is the 3D structure of molecules important?
because it modulates their chemical properties and determines their biological function
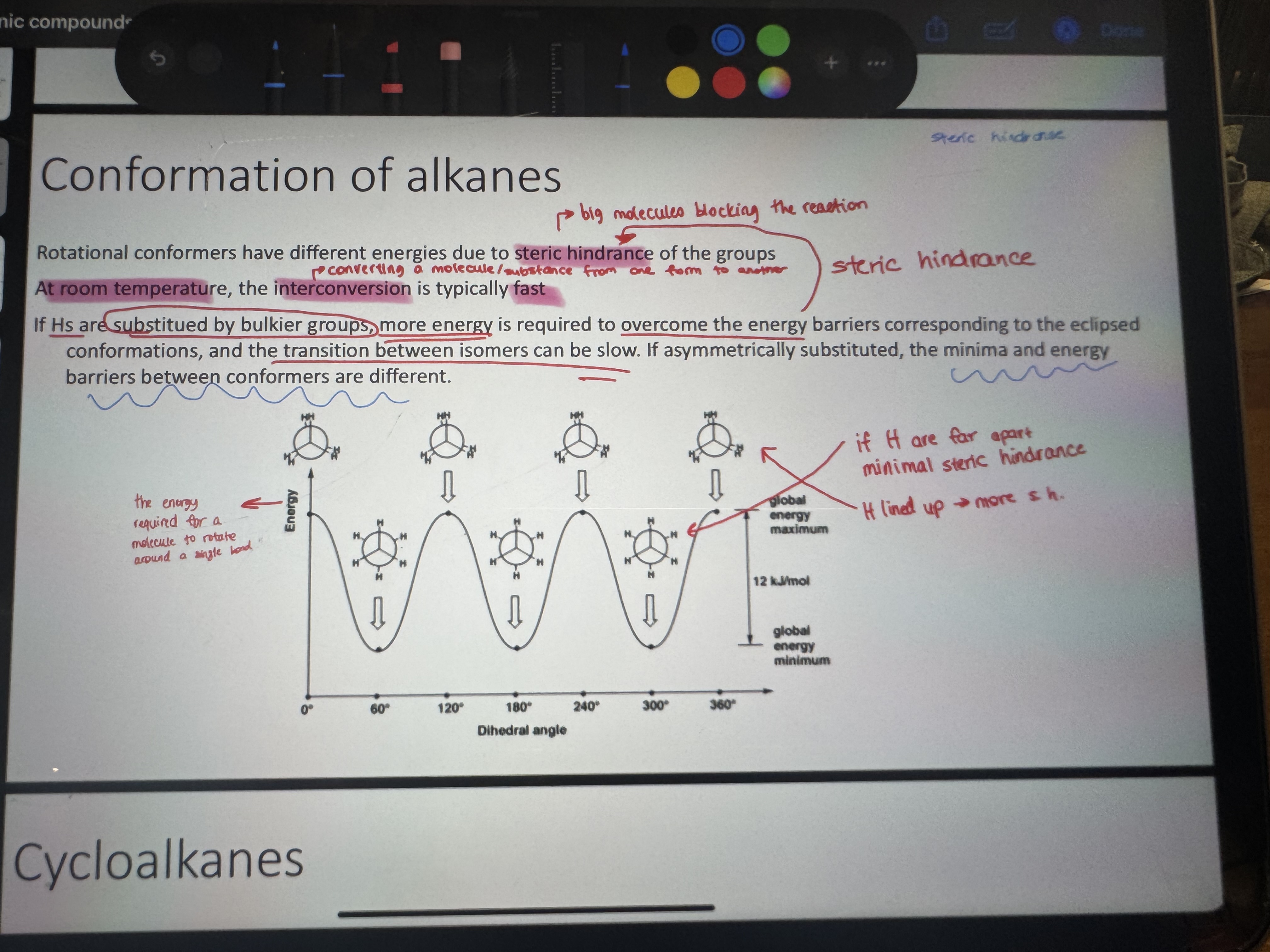
what is conformation
the shapes that molecules can adopt due to rotation about bonds (different conformation different properties)
what is a conformational isomer
same molecule different arrangment of the ch3 groups
what is steric hindrance
bic molecules blocking the reaction
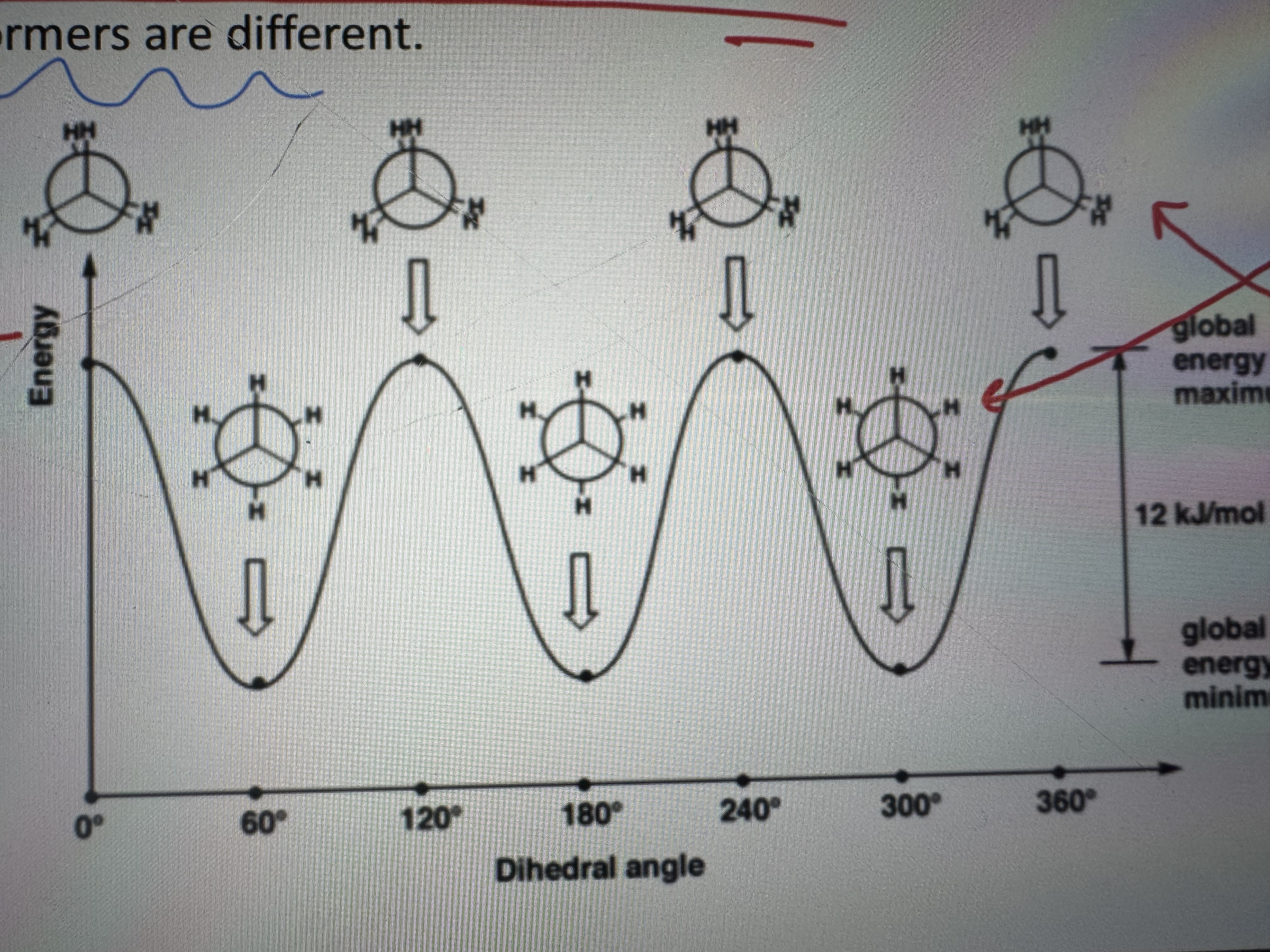
explain this
why does cyclohexane not have a planar structure?
-the sp3 interactions prevent it
-to obtain a planar structure the angles need to be 120 degrees which is much greater than the angle between hybrid orbitals
the required distortion would lead to a very high energy and thys unstable molecule
what is the chair conformation
four atoms on a plane one above and one below
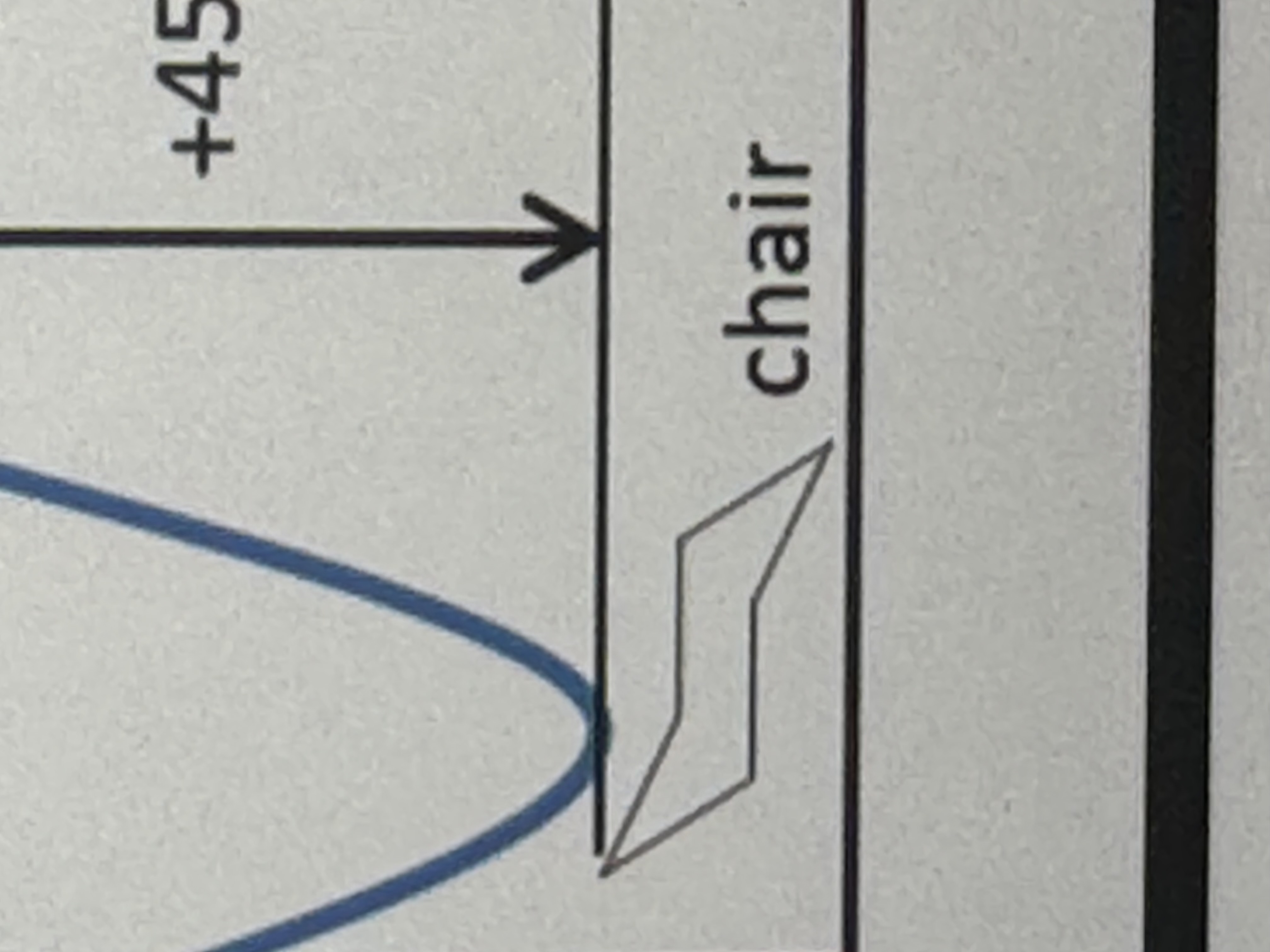
what are the orientations of a cyclic structure
axial (higher energy) or equatorial (lower energy) (both are chair cyclohexane forms)
which cylic structure is preferred
equatorial bc it has lower energy
what is the way to convert one cylic structure to another called?
chair flip
what can we replace a hydrogen in an alkane with?
a halogen or an alkyl chain
to create a structural isomer how many carbon atoms is the alkane supposed to have (minimum)
4+
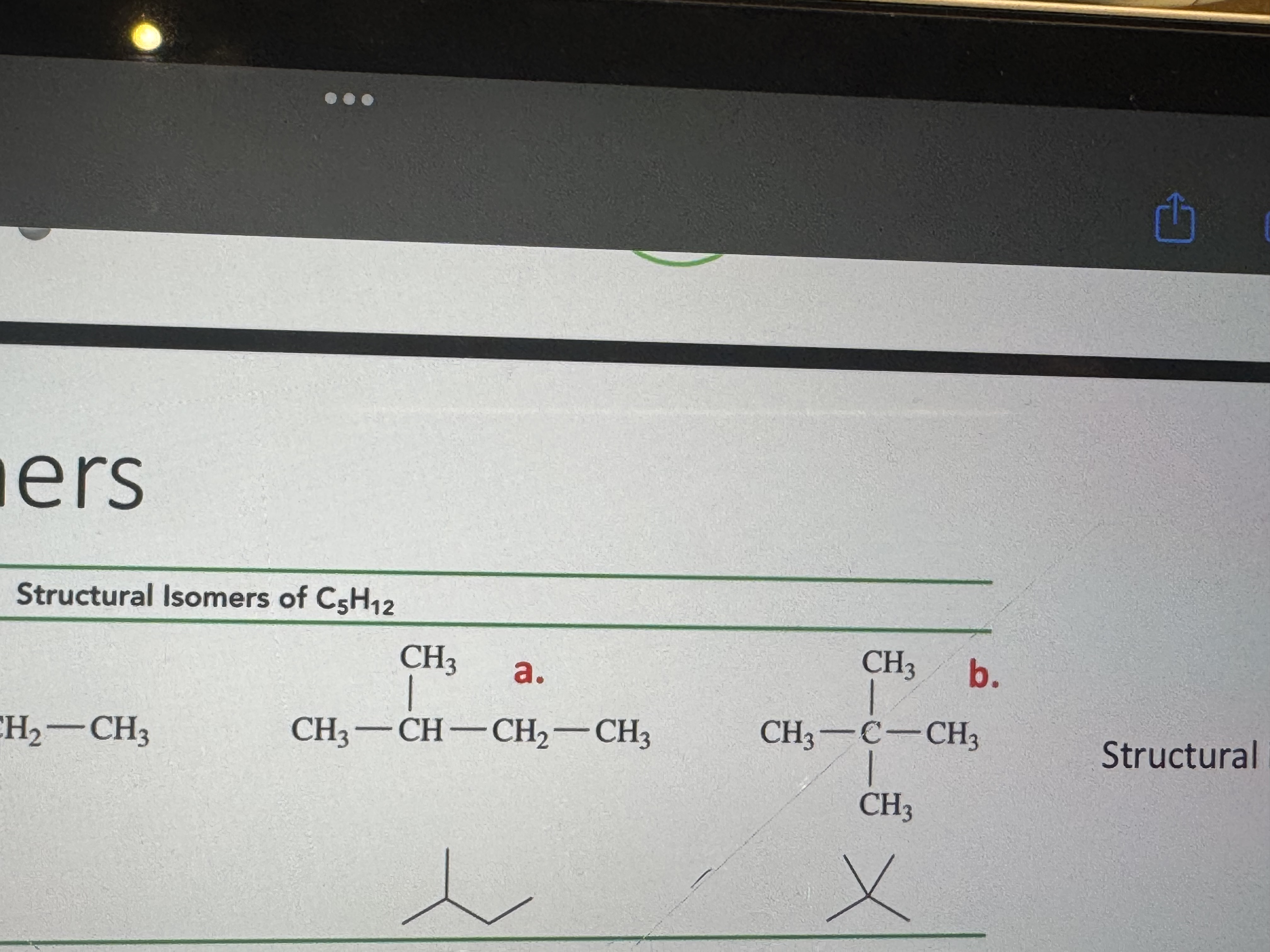
name these isomers
isopentane and neopentane
do an organic compound and it’s isomer have different chemical properties?
yes
which alkanes are used as heatinf fuels
methanes and butanes
which alkanes are volatile liquids, fuels
pentane and octane
if we get a longer alkane chain what else increases
they become waxy solids and their boiling/ melting point increase
why does the boiling/ melting point increase when the length of the C chain increase
because of the dispersion forces. the bond between c atoms are hard to break
when branching increases what else also increase
melting/ boiling point
what kind of alkanes have the highest melting/ boiling point
cyclic alkanes
why do branched alkanes have a lower boiling point?
the 3D arrangment of the substituents makes intermolecular packing less easy to achieve
why are alkanes so little reactive
the c-c bonds are very strong and difficult to break
what is added for the combustion of alkane
alkane + 02 —> CO2 + H2O + energy
what does the reaction of an alkane and a halogen create
a haloalkane
what is a nucleophilic substitution
a chemical reaction where an electron rich species replace a functional group on an electron deficient molecule
what is a nucleophile
a molecule/ ion that has at least one electron pair in a non-bonding orbital (a nucleophile always have negative charge)
what is a carbocation
a positively charged ion with a positive charge on a carbon atom
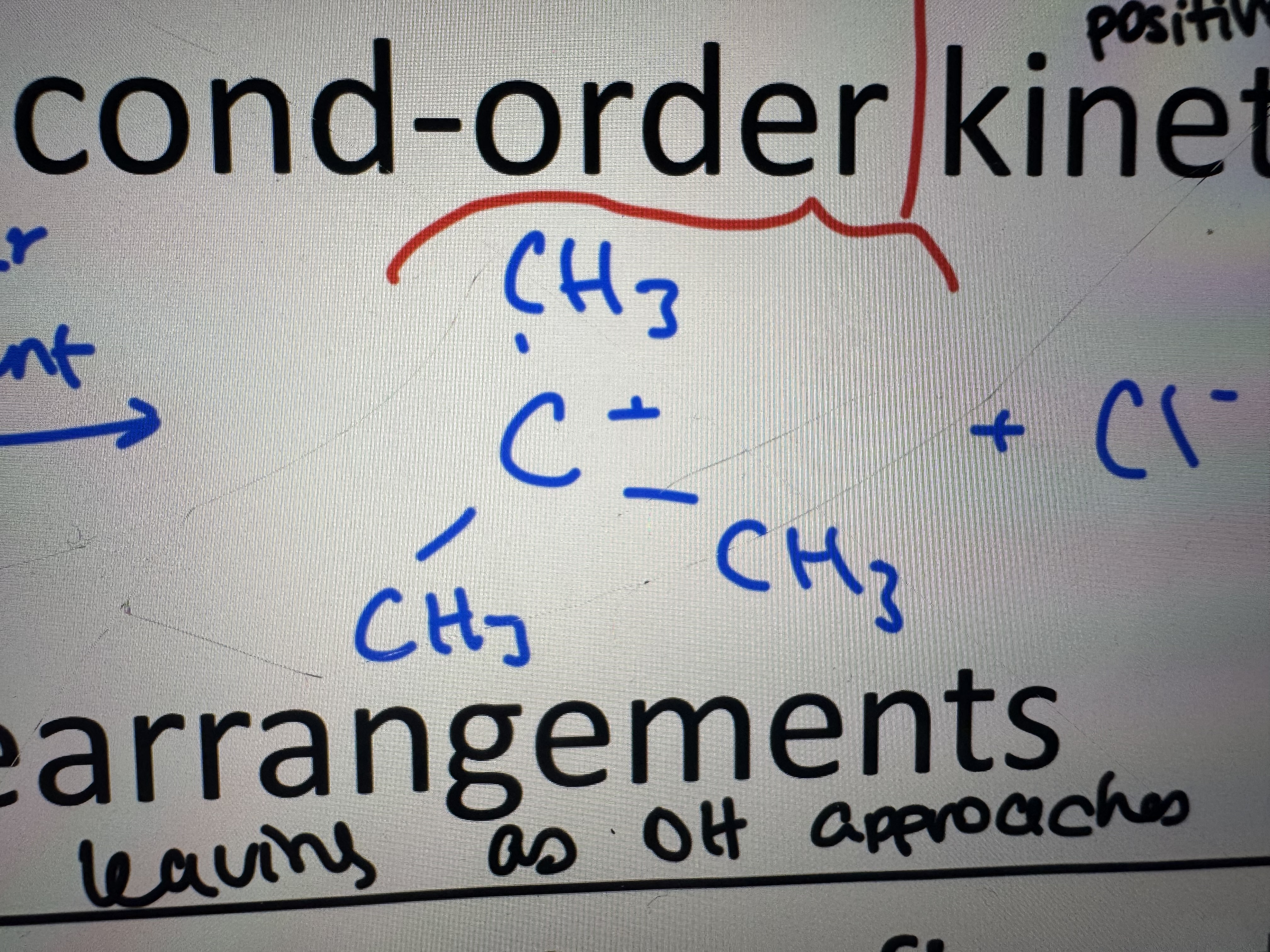
what is a sn1 reaction
formation of an intermediate carbocation with possible arrangments
what is a sn2 reaction
leaving group leaves as the nucleophile approaches. inversion of configuration of the carbon atom
why are alkanes and alkynes unsaturated hydrocarbons
because they dont contain the maximum amount of hydrogen atoms
what is a reduction reaction
reaction with a hydrogen gas to increase the number of hydrogen atoms and become alkanes
in a reduction reaction does the oxidation number decrease
yes
alkenes bond and arrangment
hybridized sp2 bond and trigonal planar arrangment
alkynes bond and arrangment
hybridized sp bond and linear comformation
do alkanes, alkenes, alkyns have free rotation
alkanes have free rotation but alkenes and alkynes dont
what causes a cis-trans isomerism
the presence of a double or triple bond that restricts the bond rotation within a molecule
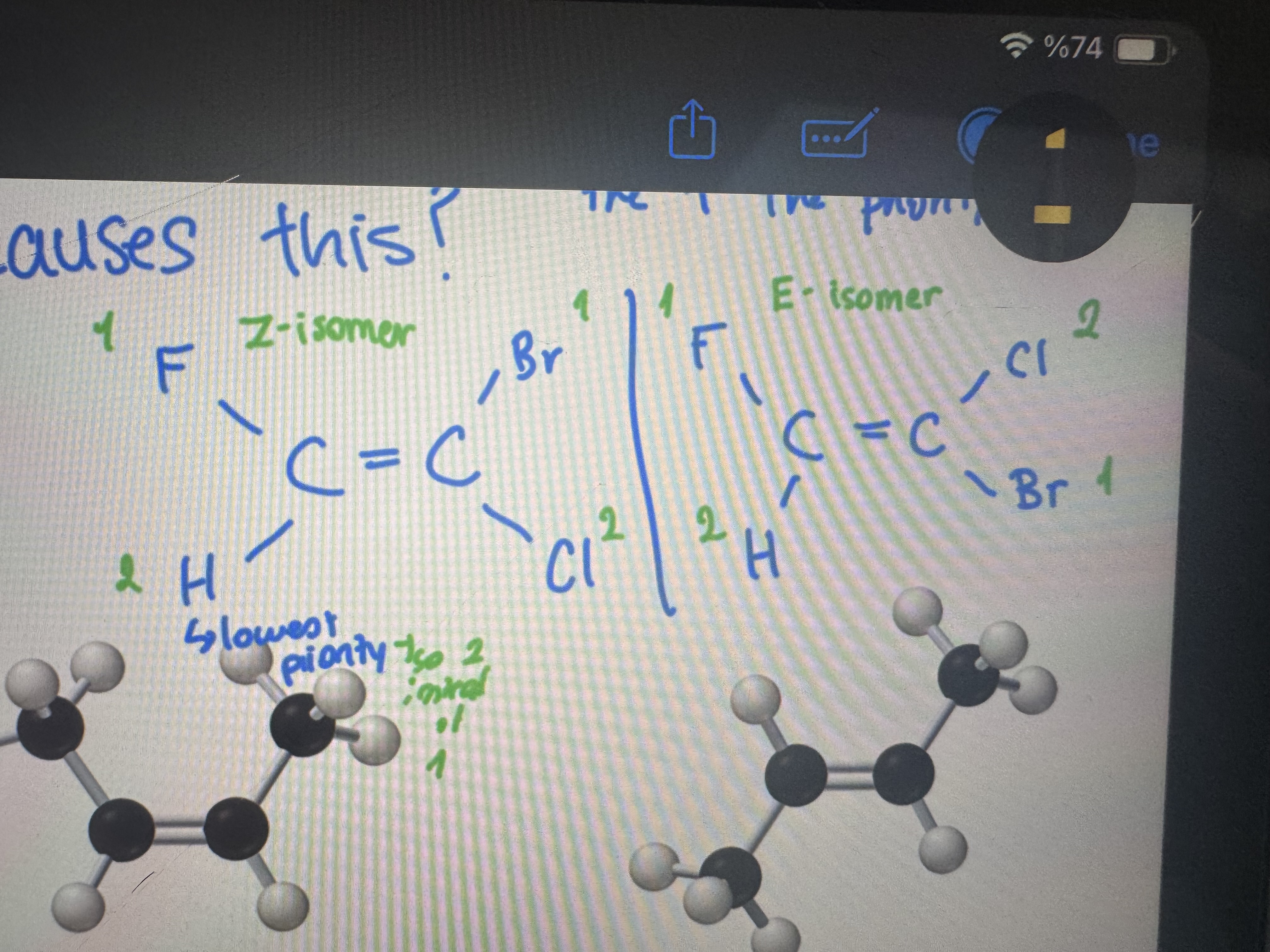
which one is a z isomer and which one is an e isomer and why
z isomer/ e isomer because of the atomic numbers we understand which is cis and which is trans
what is an electrophile
substances deficient in electrons
what is an electrophilic addition
electrons form a bond using the pi electrons of the double bond forming a carbocation. the hybridization of the atoms changes to sp3. then a nucleophile can make a bond with the carbocation. as a result the souble bond is now single and we added two dif groups to the carbon atoms
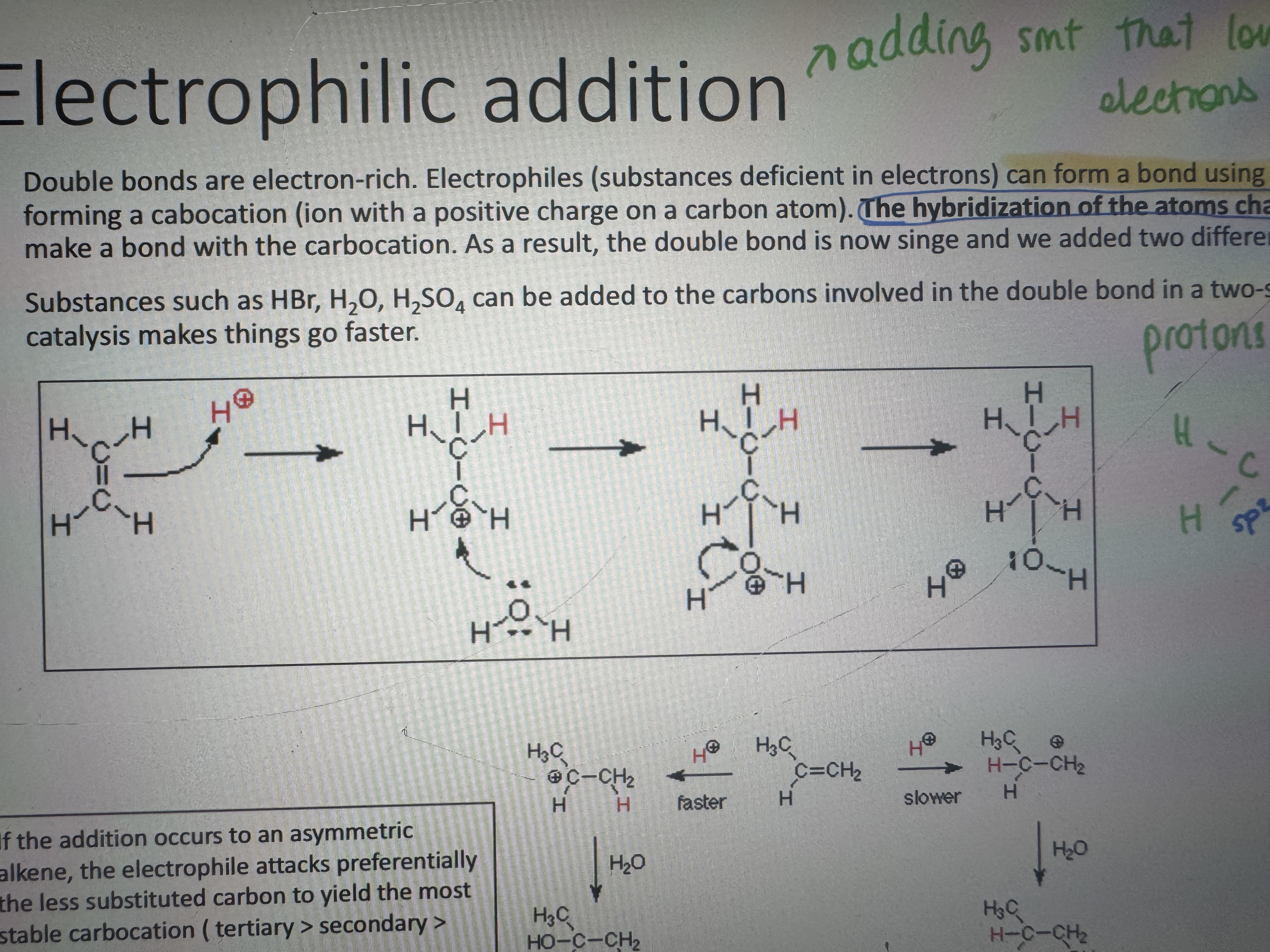
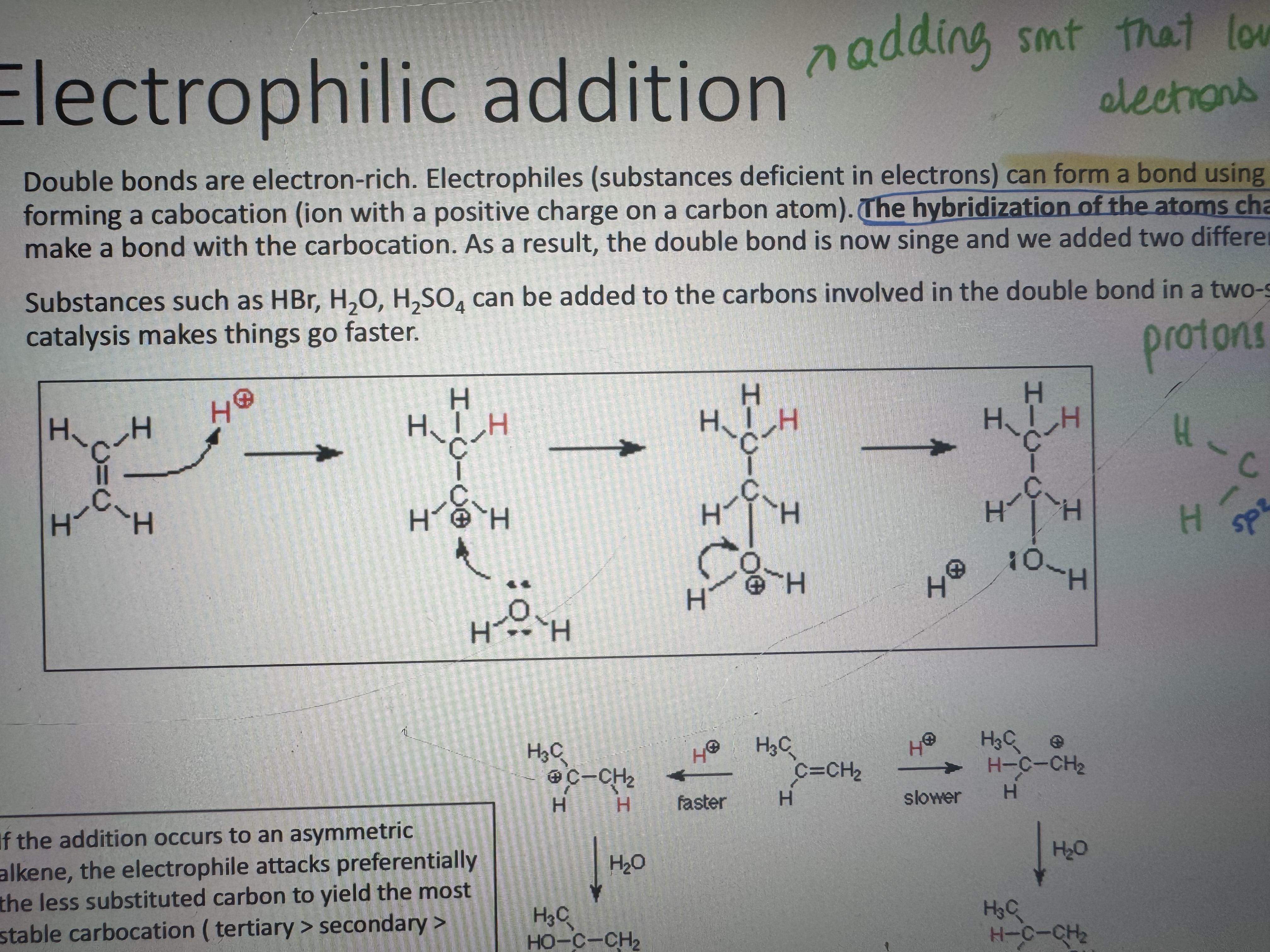
in this reaction what is the role of a proton (it means H+ i guess)
protons are not reactants they are catalysts
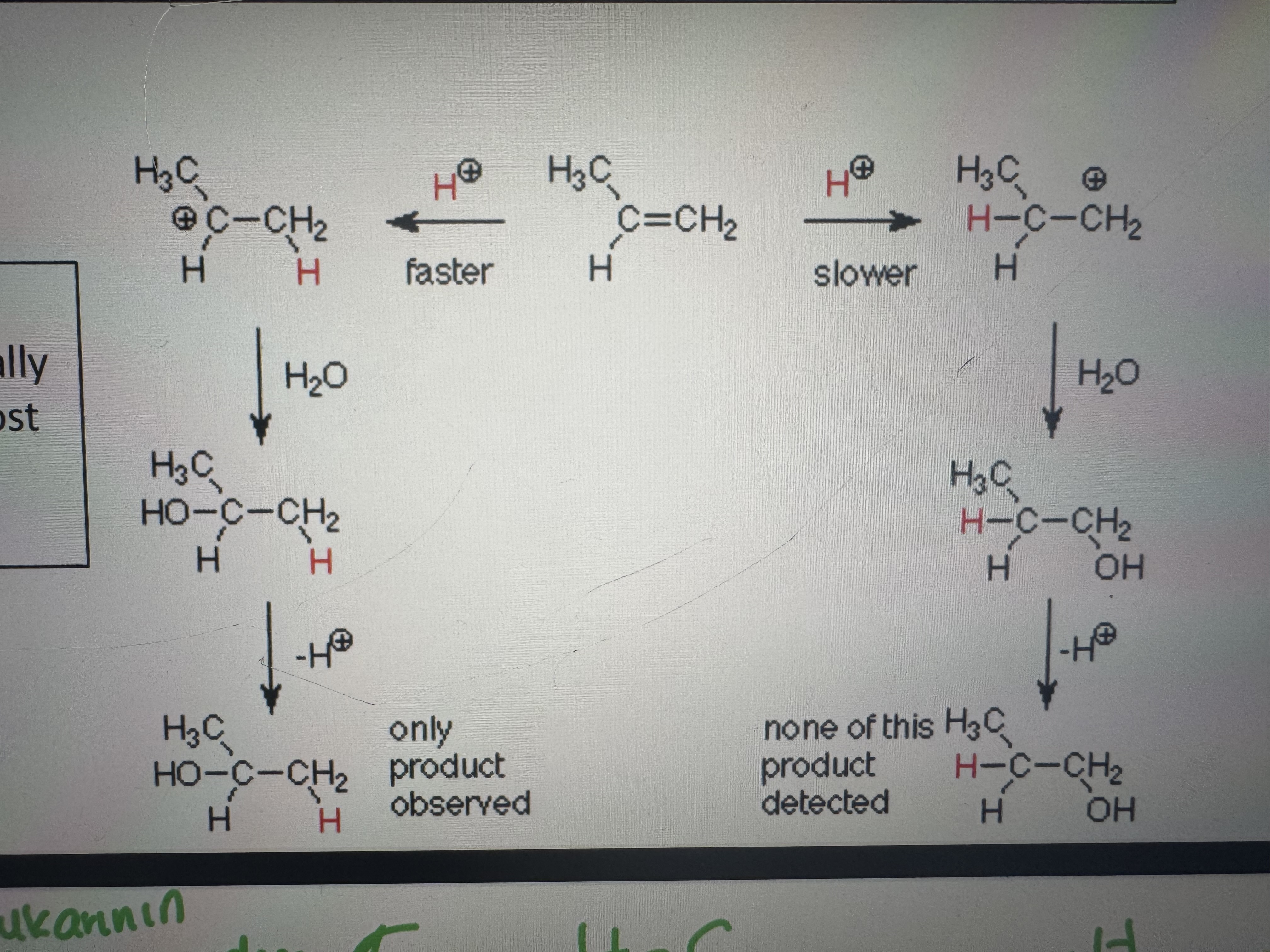
if the addition occurs to an assymetric alkene which c atom does the electrophile attack
the less substituted one to yield the most stable carbocation (tertiary>secondary>primary) (less c atom attached more chance of the electrophile attacking to c)
what do polymerization reactions require?
high temperature
a catalyst
high pressure
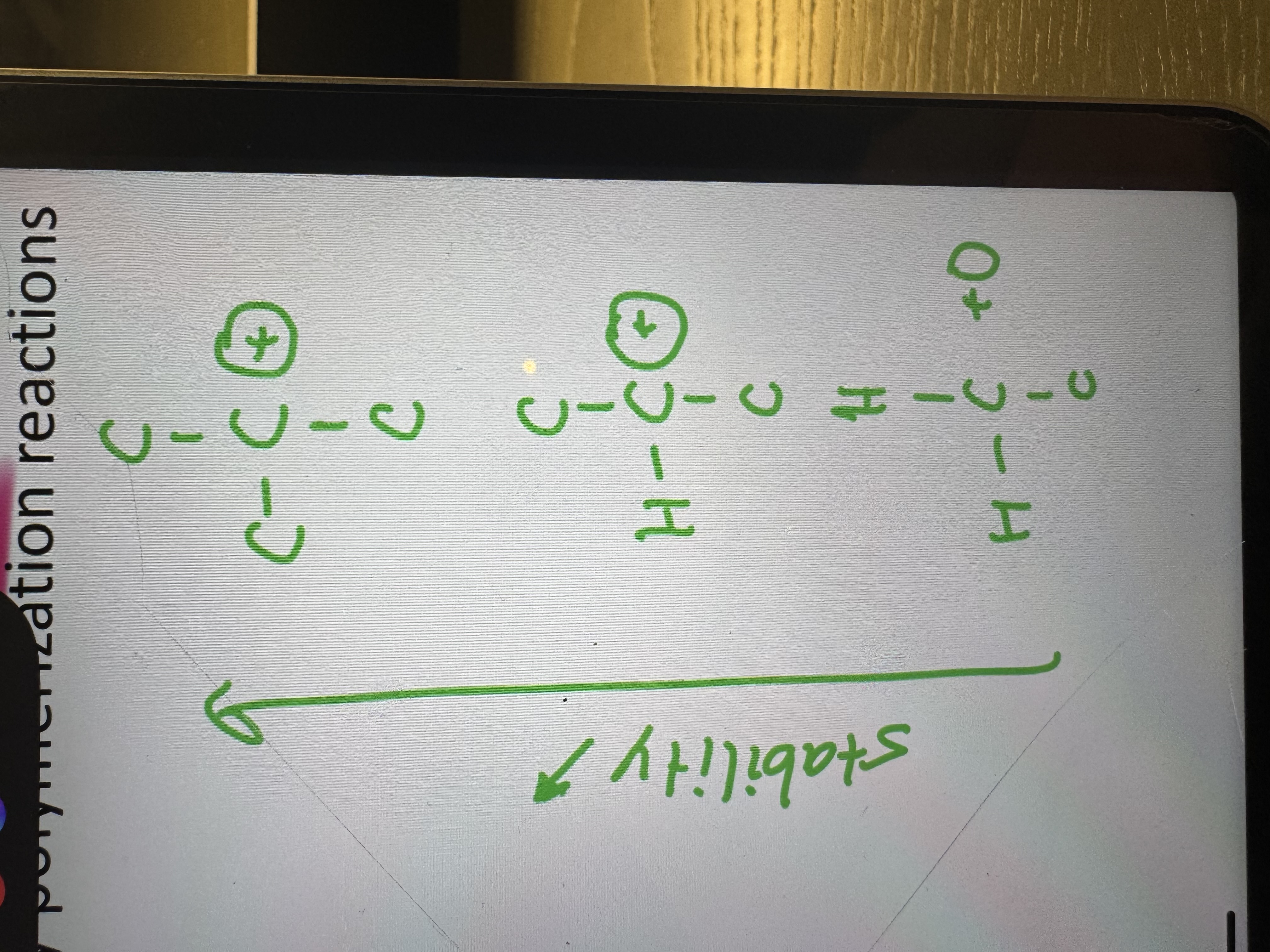
going from down to up does the stability increase or decrease
it increases (more c-c bonds more stability)
what is the bond lenght in aromatic compounds and c single and double bond lenghts?
140 pm / 154 pm / 134 pm
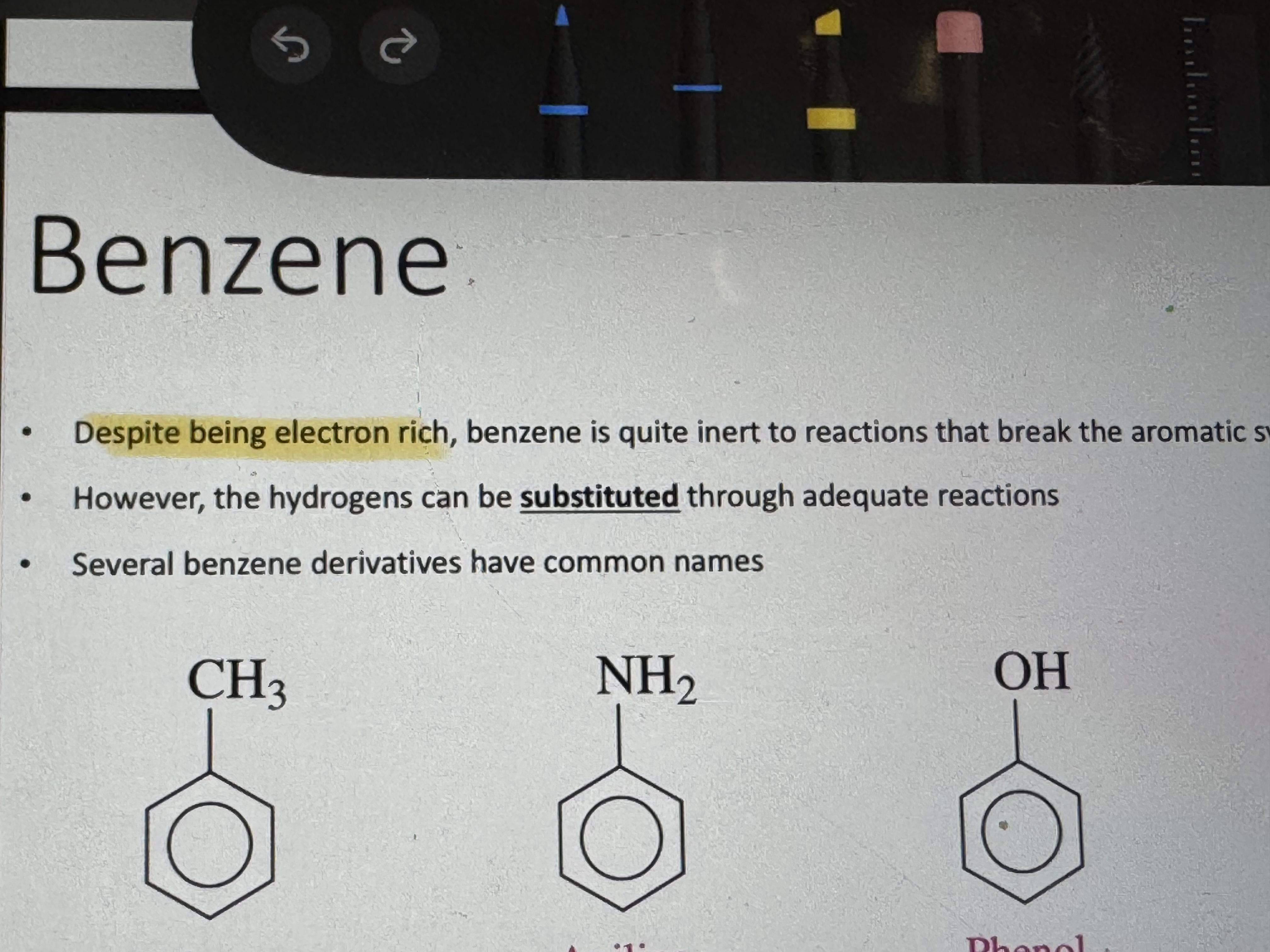
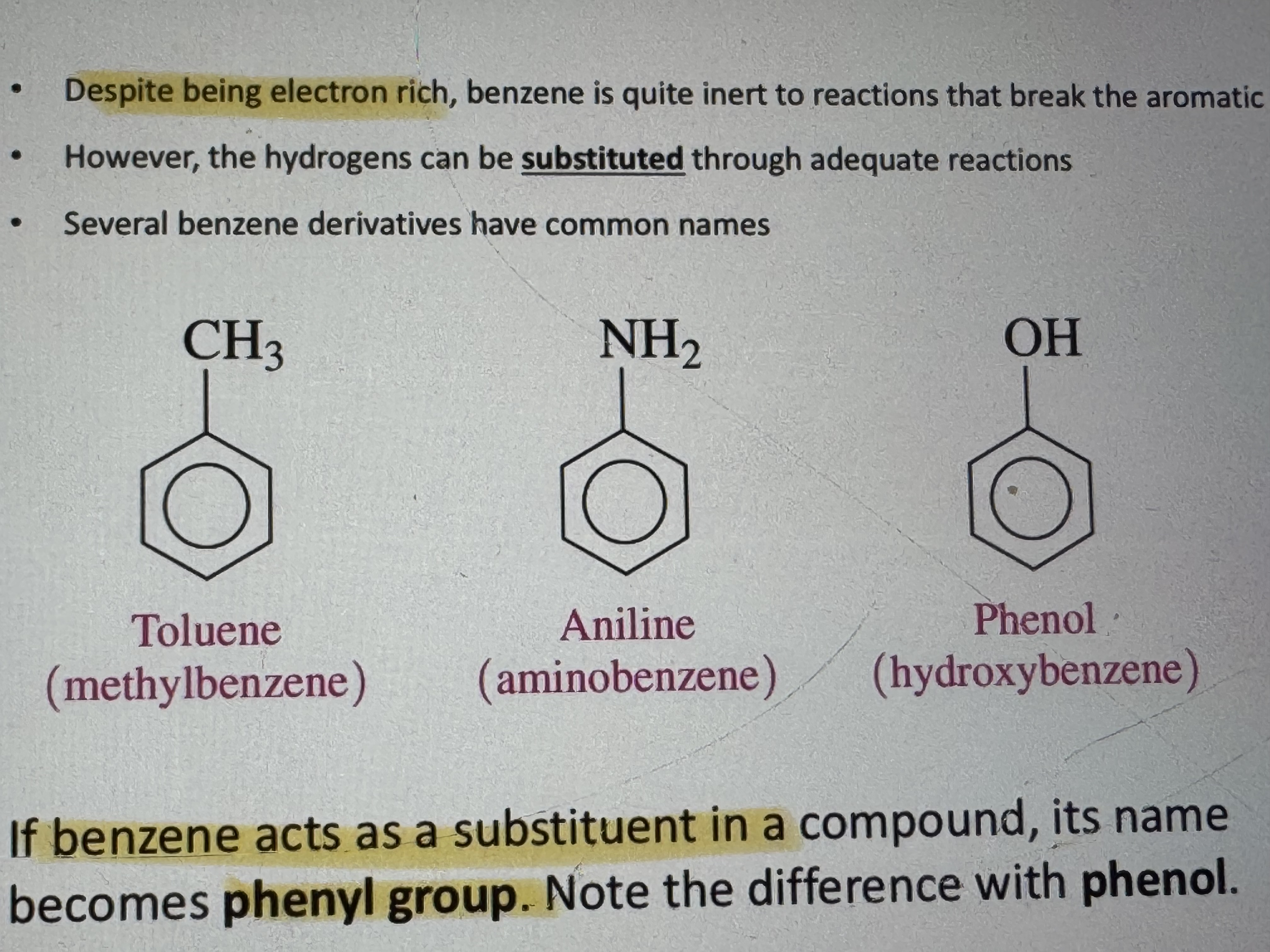
if benzene acts as a substituent in a compound what does it’s name become
phenyl group (phenol is a different things dont confuse)
what are their names
1,2 diclorobenzene
1,3 diclorobenzene
1,4 diclorobenzene
what are their other names
o-diclorobenzene
m-diclorobenzene
p-diclorobenzene
difference berween electrophile and nucleophile
electrophile has a + charge nucleophile has a - charge
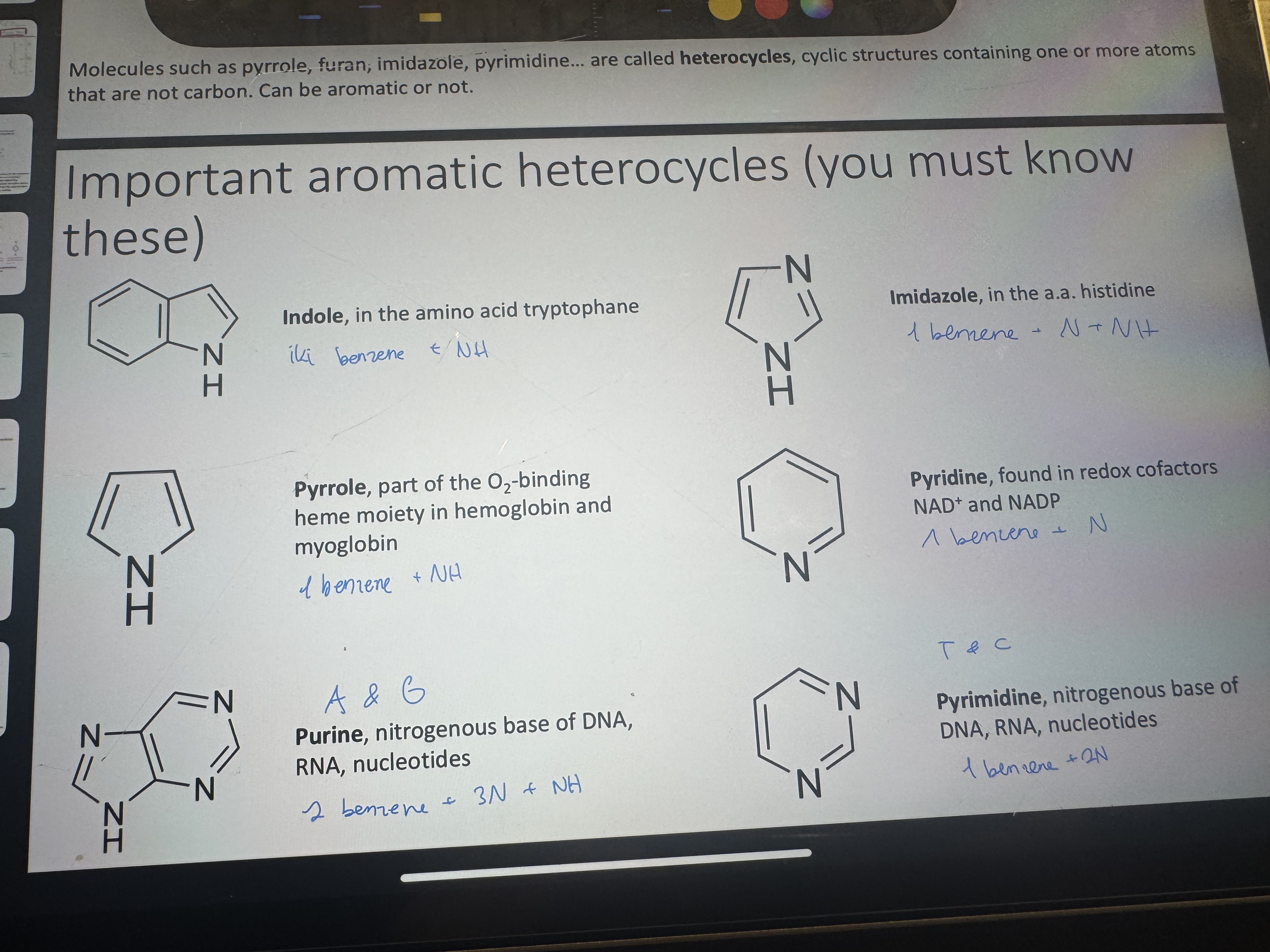
name these
ok
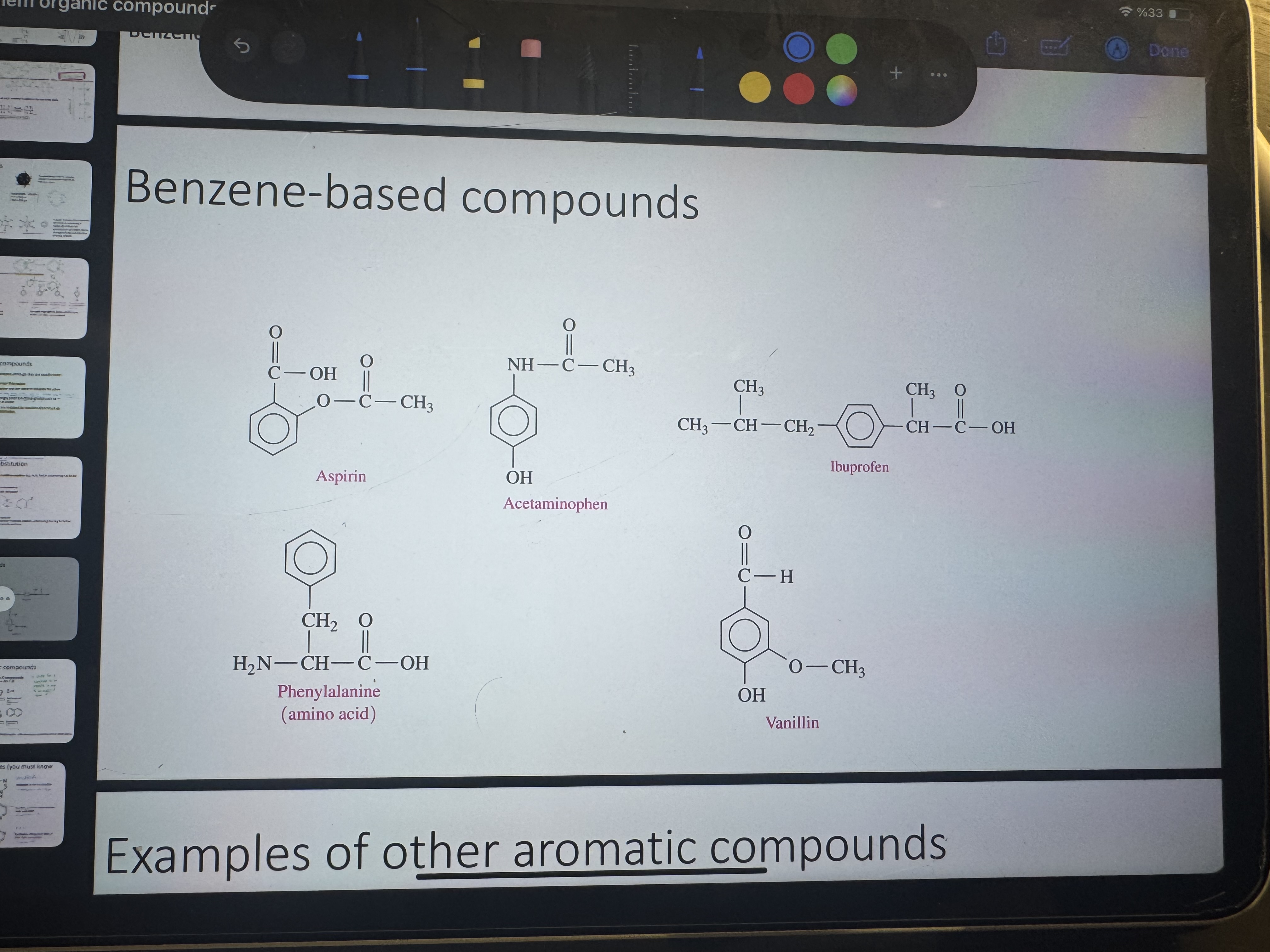
what are some benzene based compounds
aspirin, acetaminophen, ibuprofen, phenylalanine (amino acid), vanillin
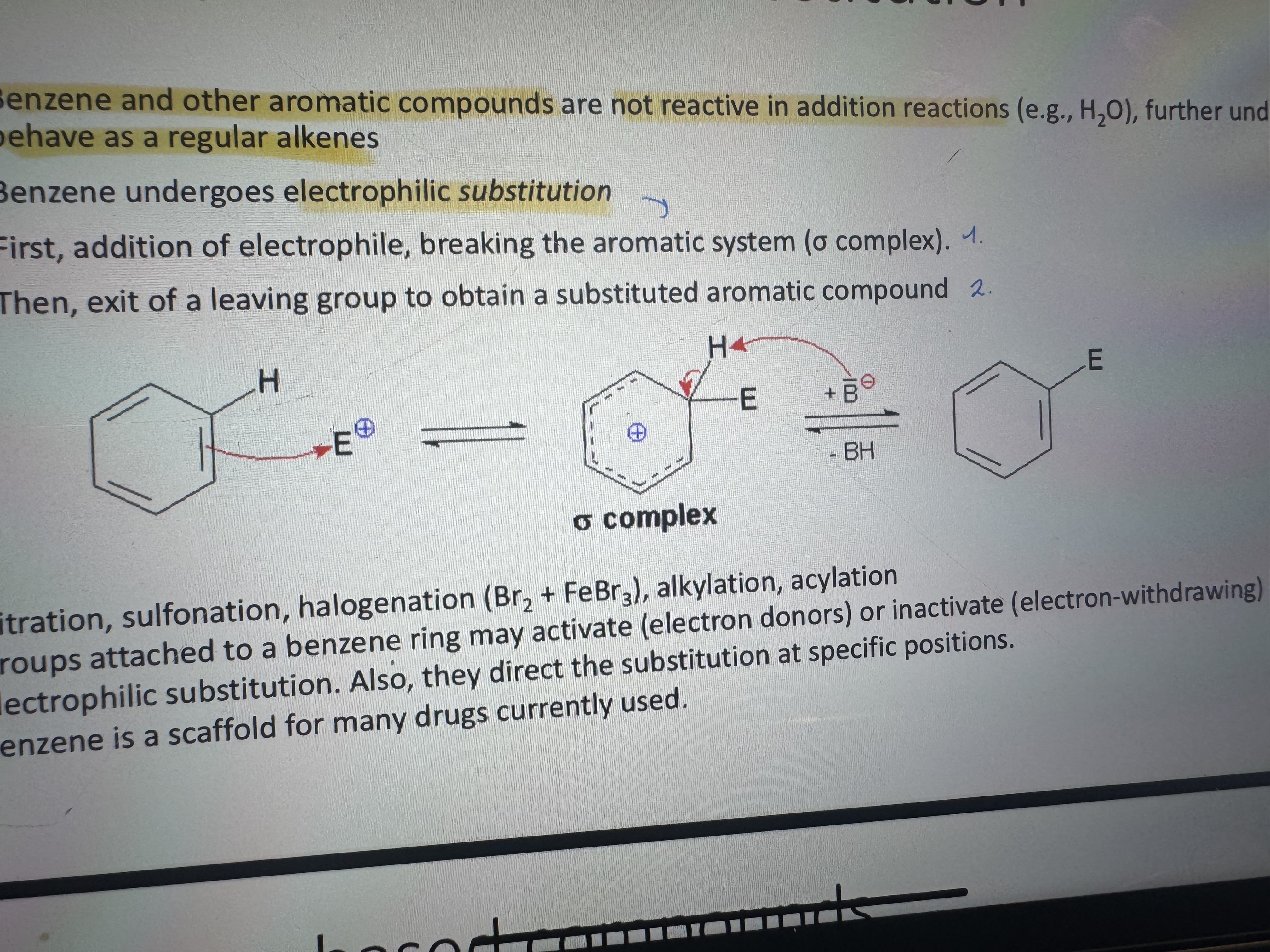
what is electrophilic substitution
first, addition of electrophile, breaking the aromatic system
then, exit of a leaving group to obtain a substituted aromatic compound