PHGY 170 Mod 4 - Cell Communication & Cell Death
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Extracellular Communication
when a signal is received from outside the cell
Intracellular Communication
external signals cause changes within a cell
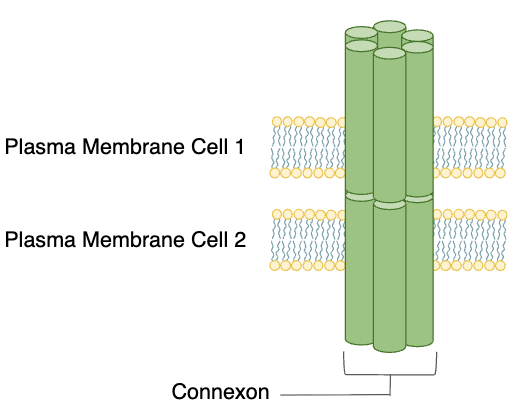
Direct Cell-to-Cell communication: Gap Junctions
cells that are touching can communicate using gap junctions
gap junctions are made of connexons which dock together to form channels from one cell to another
allows chemical signals to move directly between cells
Specifics of Gap Junctions
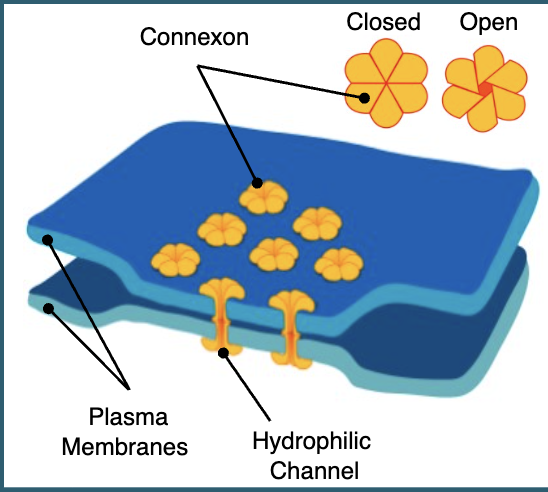
not all biomolecules can pass through gap junctions
only small particles such as ions and small signalling molecules can pass, while larger molecules such as proteins and carbs cannot
excitable cells like cardiac muscle can pass electrical signals as well as chemical signals through gap junctions
gap junctions are not open doors that allow a constant free exchange of signals
they are highly regulated and can open and close as appropriate
this gating is a defence mechanism so a cell can protect itself if something dangerous is happening in a neighbouring cell
cell to cell communication: secretions
cells that are not touching can communicate through secretions
Autocrine secretions → substances are released and have an effect on the same cell
Paracrine secretions→ substances are released and have an effect on nearby cells
Endocrine secretions → substances are released and have an effect on distant cells
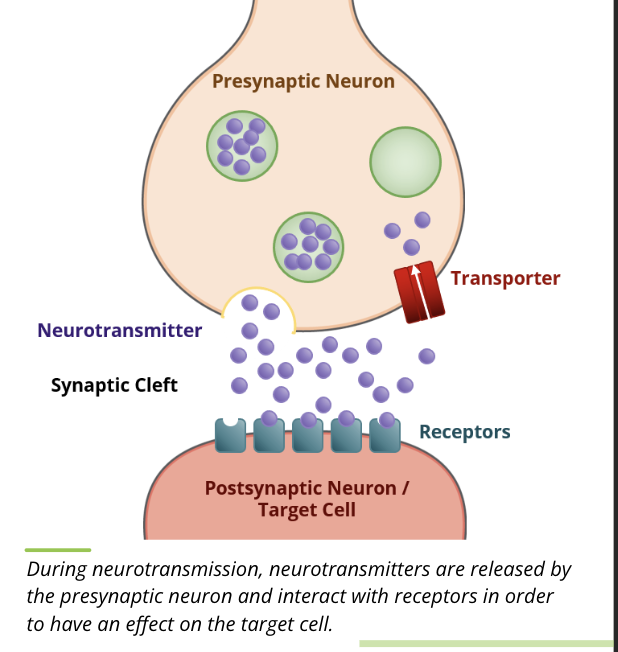
Neurotransmitters → substances are released by a nerve terminal into the synapse
Secretions: neurotransmitters
synaptic secretions occur where a nerve cell axon terminates on a target cell
when an excitatory signal comes down the axon to the synapse, neurotransmitters are released into the synapse where they either bind to a receptor on the target cell, are degraded by enzymes in the synapse, or are taken back up by the nerve cell
regardless of their fate, the presence of neurotransmitters in the synapse is very transient
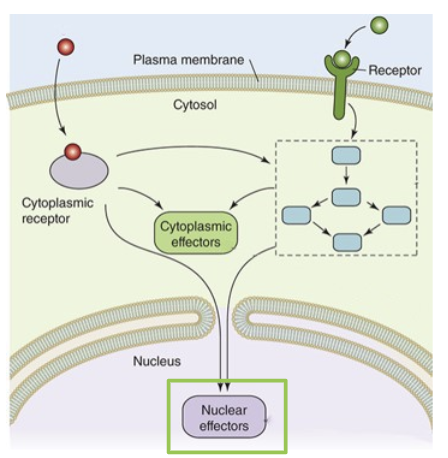
components of the signal transduction pathway
signal → can be either membrane permeable or membrane impermeable
receptors → the receptors interact with the signal
signalling proteins → signalling proteins help conduct the signal intracellularly
second messengers → non-protein molecules that help conduct the signal intracellularly
Structure of a signalling pathway
Membrane Permeable Signal Molecule→ molecules bind to receptor proteins in the cytosol
Membrane Impermeable Signal Molecule → binds to transmembrane cell surface receptor proteins which activate second messengers
Signalling Proteins and Second Messengers → amplify, process, and distribute incoming signals from both classes of signal receptor proteins
Cytoplasmic Effectors → some signals are sent to effector proteins in the cytosol. this is typically a fast, short lived response to the activation of a signalling pathway
Nuclear Effectors → some signalling pathways terminate at effectors in the nucleus. these effectors are transcription factors that control gene expression. this results in a slower, more prolonged response to a signalling pathway
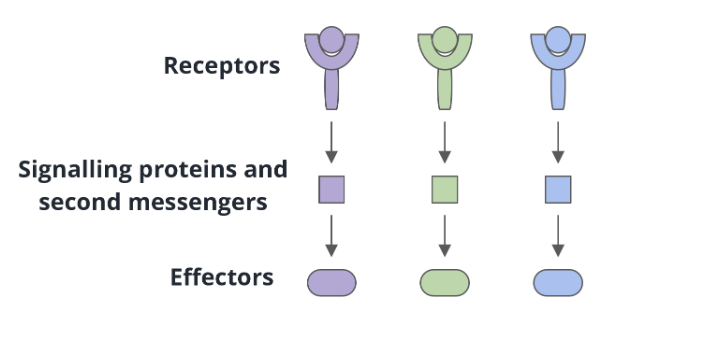
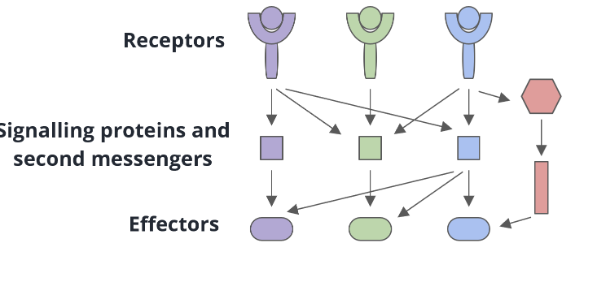
Linear signal transduction
one receptor interacts with one signalling protein or second messenger
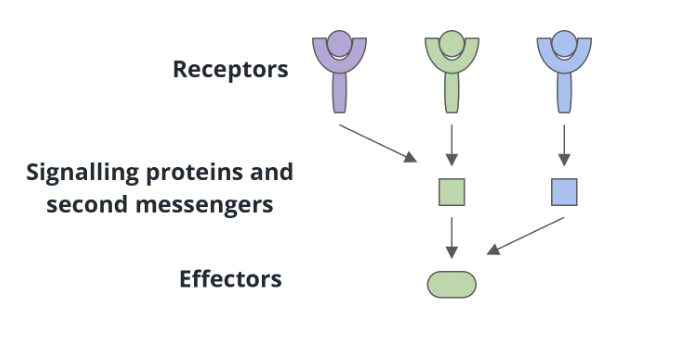
convergent signal transduction
several receptors share common signalling proteins or second messengers
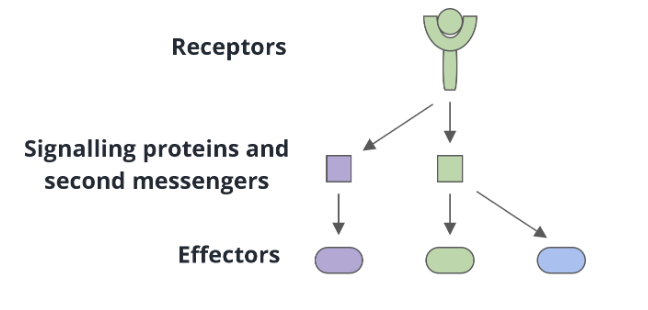
divergent signal transduction
a single receptor can interact with multiple signalling proteins or second messengers
multi-branched signal transduction
a combination of convergence and divergence may be happening at the same time
membrane impermeable ligands
the majority of signal molecules are membrane impermeable
ligands that cannot penetrate the membrane bind to receptor proteins on the cell surface
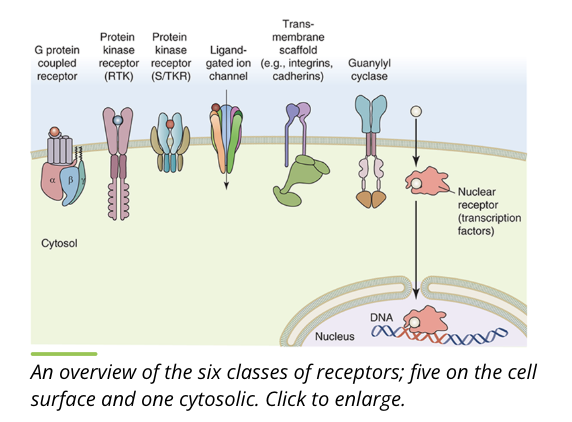
cell surface receptors can be grouped into 5 classes based on structure, binding partners, and cellular location
membrane permeable ligands
membrane permeable signal transduction molecules are mainly steroids
ligands that are able to penetrate the membrane are not limited to membrane receptors and can interact with cytosolic receptors
physical signals
physical signals like pressure, temperature, and light can trigger the signal transduction pathway instead of ligands
receptors (overview)
receptors are often found on the plasma membrane but can also be found in the cytoplasm of a cell
g protein coupled receptors (GPCR)
ion channels
guanylate cyclase
protein kinase receptors
transmembrane scaffolds
nuclear receptors
the 6 classes of receptors detect an array of environmental stimuli
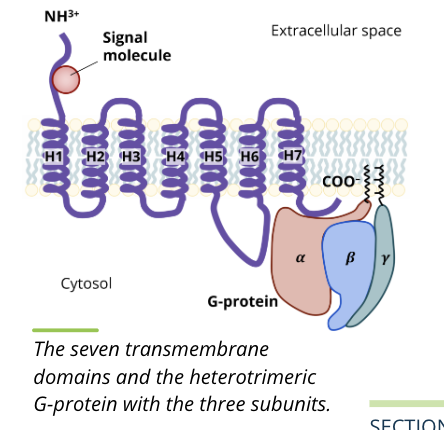
G protein coupled receptors
GPCRs represent a superfamily of receptors with hundreds of genes coding for different receptors
involved in many functions such as smell detection, and fight or flight activation
structure
a combination of seven transmembrane domains (H1 to H7) as well as heterotrimeric G protein with aplha, beta, and gamma subunits that interact with each other
function
the binding of a ligand to a GPCR causes a conformational shape change in the receptor that leads to the activation of the coupled G protein subunits
Ion Channel Receptors
aka ligand gated channels
a type of channel that can exist in the plasma membrane
transmit signal information by permitting ions to flow from one side of the membrane to the other
when their specific ligands bind, the channels udnergo a conformational change in shape that opens their pores and allows the ions to flow through
unlike other receptor types, these proteins are not enzymes
ion channels are responsible for voluntary muscle contraction
this type of signalling is common for much of the communication between nerve cells through the release of neurotransmitters
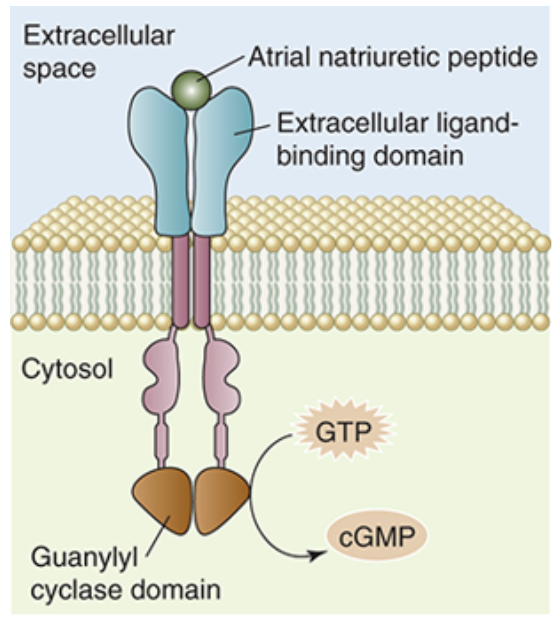
Guanylate Cyclase Receptors
a receptor that can be found both bound to the membrane and soluble within the cytosol
Structure
membrane bound guanlyate cyclase contains an externalized ligand binding domain, a transmembrane domain, and an internal catalytic domain
the soluble form of guanylate cyclase serves as a target for some membrane soluble ligands in addition to mediating some intracellular processes
Function
when the catalytic receptor is activated, the catalytic domain of membrane bound guanylate cyclase converts guanosine triphosphate (GTP) into cyclic guanosine monophosphate (cGMP)
cGMP then binds to other signalling proteins to initiate cellular processes
guanylate cyclases play an important role in vision as they help convert a light signal into an electrical signal in the eye
Protien Kinase Receptors
human cells express hundreds of different protein kinases
not all protein kinases are cell surface receptors, many are cytosolic proteins that also participate in signal transduction, alter enzyme activity, or other cellular processes
the action of protein kinases is to phosphorylate other proteins that contain serine, threonine, or tyrosine residues
protein kinase receptors are especially important in clinical settings because their dysfunction is associated with the development of a number of cancers
there are 2 classes of protein kinase receptors:
receptor tyrosine kinases (RTK)
serine/threonine kinases receptors (S/TRK
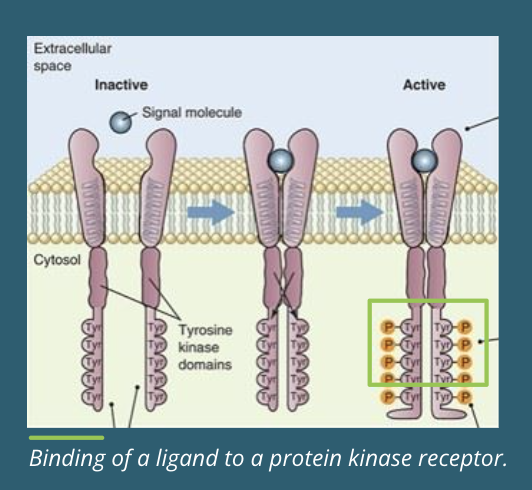
Protein Tyrosine Kinase Receptor Ligand Binding
Inactive → before ligand binding the inactive receptors are separate polypeptides with inactive tyrosine domains
Dimerization → binding to a signalling molecule causes the two subunits of the receptor to join together or “dimerize,” forming a dimer. once dimerized, the kinase is active
Transautophosphorylation → transautophosphorylation occurs when the cytoplasmic tail of one subunit is brought close to the tyrosine kinase domain of the other subunit, and the opposite domain is phosphorylated on specific tyrosine amino acids
Binding Sites → the resulting phosphotyrosine amino acids are binding sites for additional signalling proteins that pass the signal along the pathway
Resetting → the ligand is released, and the amino acids are dephosphorylated by phosphoprotein phosphotases. the kinase resets itself to its inactive state of two separate polypeptides
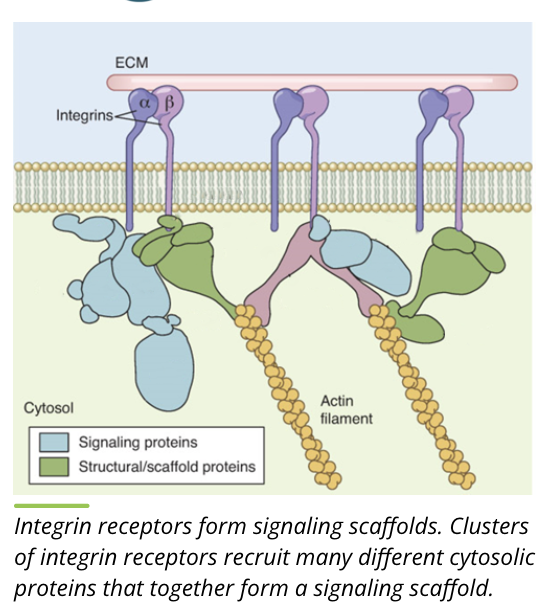
Transmembrane Scaffold Receptors
transmembrane scaffolds are different from other membrane receptors in that they do not always have a distinct single function
this type of receptor tends to form in large clusters of receptors abd signalling proteins with complex interactions
by doing this, they can regulate signal transduction
the scaffold proteins themselves determine which signalling proteins can ind to a complex, associating with the membrane receptor, and form what is called a signalling scaffold
Functions
bring signalling proteins together
regulate signal transduction
localize signalling proteins to specific cellular areas
isolate specific signalling pathways
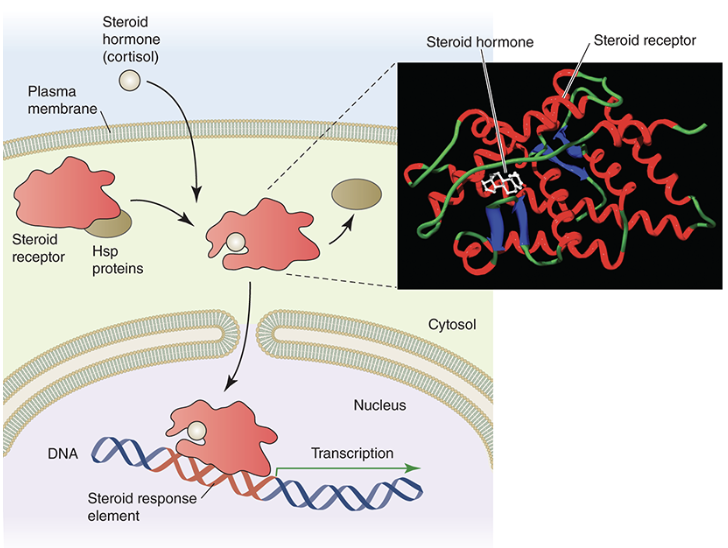
Nuclear Receptors
nuclear receptors are receptor proteins that are found in the cytosol of cells
certain ligands, such as steroids, can freely cross plasma membranes and bind to these intracellular receptors
once bound, these receptors move through nuclear pore complexes directly into the nucleus
once inside the nucleus, the activated receptor complex can bind to a specific DNA sequences called steroid response elements (SREs) to control the expression of genes
since they help to regulate gene expression, this class of receptors are also called transcription factors\these types of receptors also play a role in response to toxic substances
Signalling Proteins
the primary purpose of signalling proteins is to transmit and amplify signal information
signal proteins can also mobilize second messengers, which are non protein molecules that can ling signalling proteins together into further signalling pathways or have direct actions on their own
mobility → signalling proteins are highly mobile and can diffuse rapidly through the cytosol. if membrane associated, they move rapidly within the plasma membrane
catalysis → signalling proteins are either enzymes that can catalyze chemical reactions for signal amplification or they are capable of binding to enzymes
Signalling Proteins: G proteins
G proteins are proteins that bind to GTP and propagate signals
Monomeric G proteins
monomeric G proteins are single polypeptides that contain at least 2 different binding sites (one for GTP or GDP and one for the target protein) and a GTPase domain
they are NOT coupled to GPCRs
when GTP is bound, it is in an activated state and can bind to its target protein
the GTPase can then cleave the GTP to form GDP
eventually the GDP is released and GTP can then bind again to reactivate it
Heterotrimeric G proteins
similar in function to monomeric G proteins except they contain 3 different polypeptides
these G proteins are anchored to the plasma membrane and are activated by the G protein coupled receptors already mentioned
the alpha subunit is analogous to the monomeric G protein in that it binds GTP/GDP and a target protein
the beta/gamma subunits are attached together and their primary function is to stabilize the inactive (GDP bound) form of the alpha subunit
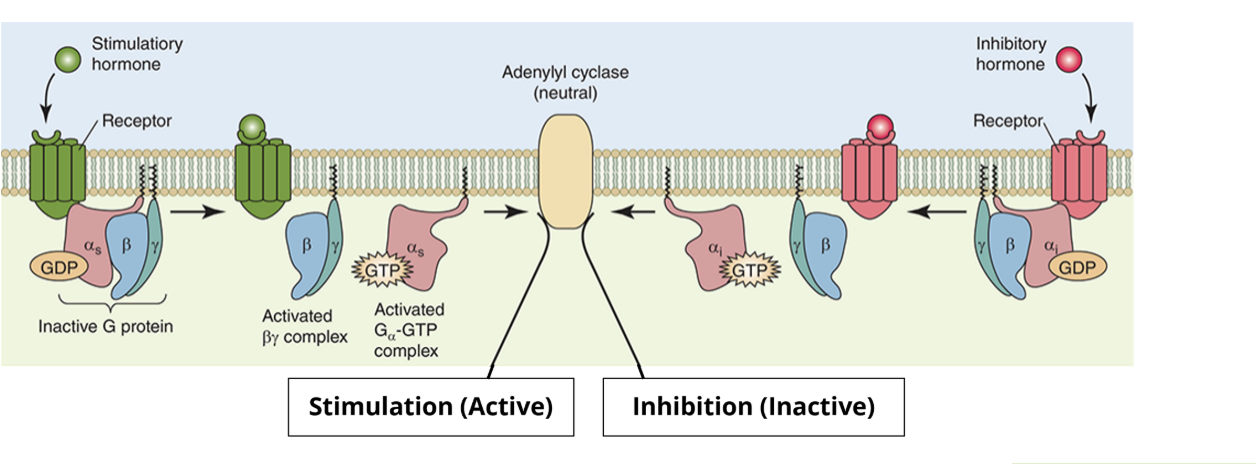
Activity of G proteins: Binding
the heterotrimer containing the alpha and beta/gamma subunits is bound to GTP. this is the inactive form
when a ligand binds to the receptor, it changes conformation to interact with the heterotrimeric G protein
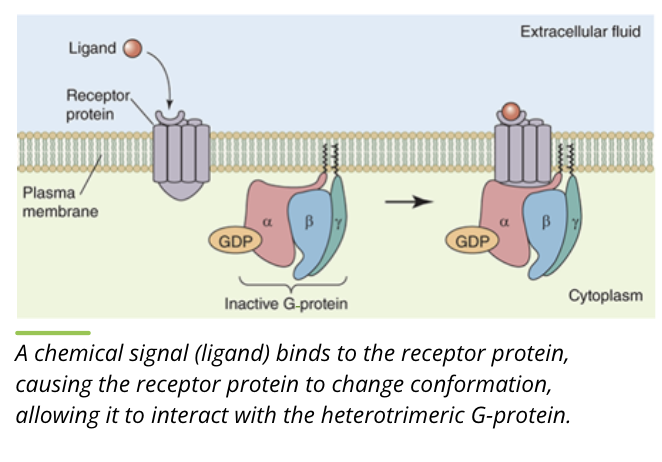
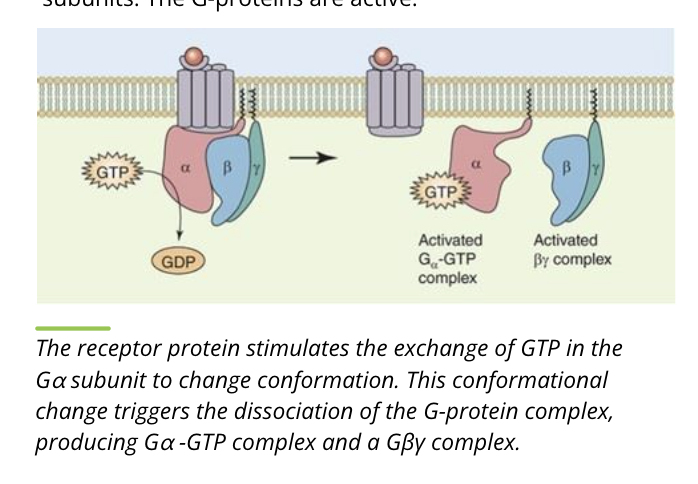
Activity of G proteins: Separation
the receptor protein causes exchange of GDP with GTP on the alpha subunit
the heterotrimer separates into separate alpha and beta/gamma subunits
the G proteins are active
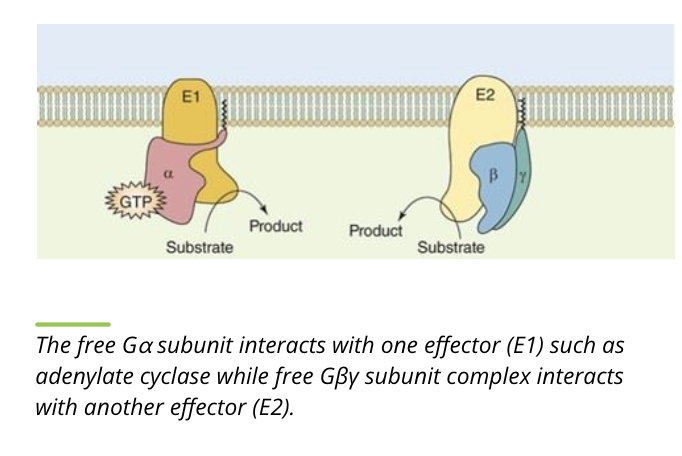
Activity of G proteins: Propagate
while separated, the alpha and beta/gamma subunits bind downstream targets, propagating the signal pathway
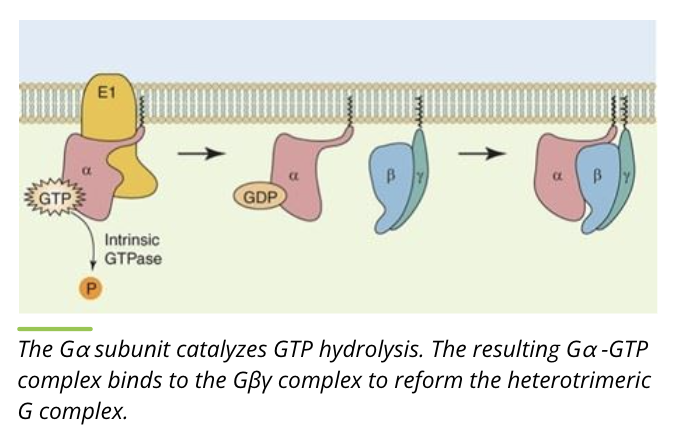
Activity of G proteins: cleave and reform
the alpha subunit cleaves GTP to form GDP, alpha and beta/gamma subunits bind to reform the heterotrimer
this returns the heterotrimeric G protein complex to the inactive form
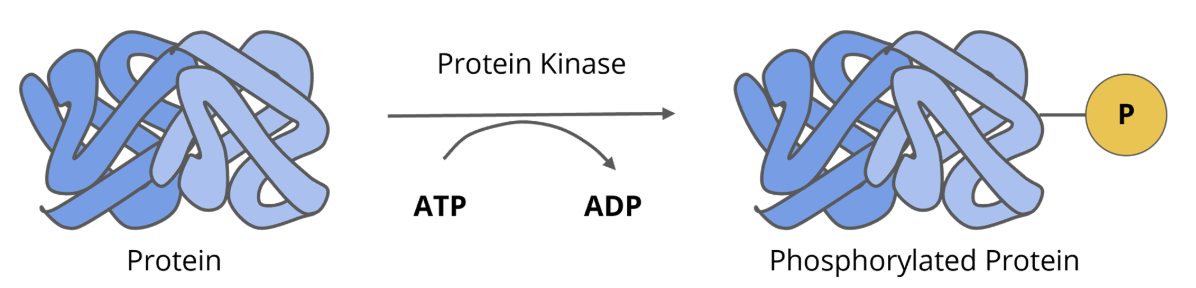
signalling proteins: protein kinases
protein kinases are enzymes that attach phosphate groups to tyrosine, serine, and theronine
in addition to receptor protein kinases, there are non receptor protein kinases
the majority of protein kinases are non receptor, cytosolic signalling proteins
cytosolic protein kinases can act as intermediates, in that once they are activated they can activate other protein kinases, other signalling proteins, or they can directly phosphorylate effector proteins like enzymes
in general, phosphorylation of target proteins leads to their activation byt some proteins are inactivated by phosphorylation
some protein kinases can enter the nucleus but they do not interact with DNA directly
instead, they can phosphorylate proteins that do interact directly with the DNA
Signalling proteins: calcium binding proteins
Ca2+ is an ion in the cell that has a number of function
typically, intracellular calcium is kept at low concentrations so when levels increase due to a signalling event, it can interact with certain proteins causing downstream effects
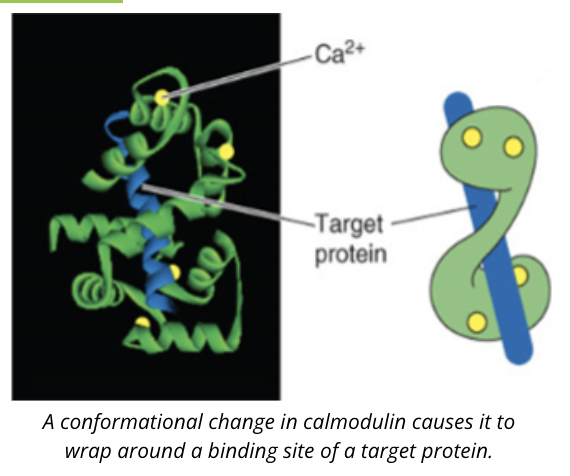
an example of calcium binding protein is calmodulin
when calcium concentrations rise, calcium binds to calmodulin inducing a conformational change that allows calmodulin to bind to its target protein
Adenylyl cyclase
another major class of intracellulr signalling proteins is adenylyl cyclase
they are related to guanylyl cyclase in that a nucleotide triphosphate is converted into another form
ATP is converted into cAMP, perpetuating the signal
in contrast, adenylyl cyclase is not linked to membrane receptors
instead, adenylyl cyclase binds to the alpha subunit of the heterotrimeric G proteins, which is why it is designated as a signalling protein instead of a receptor type
Signalling proteins: adenylyl cyclase subunits
there are two types of heterotrimeric G protein alpha subunits:
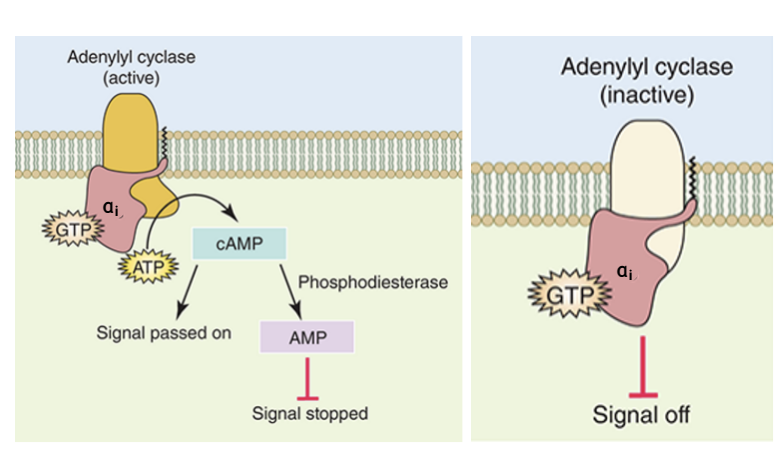
alpha s stimulates adenylyl cyclase
alpha i inhibits it
these two different forms of alpha subunits form parts of different heterotrimeric G proteins and are linked to different GPCRs which highlights a level of cellular decision making in which multiple pathways converge to allow a single response
Signalling proteins: lipid kinases
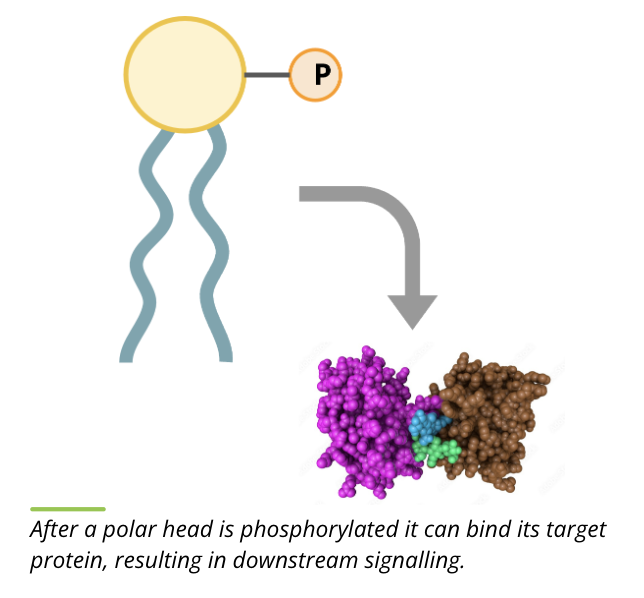
lipid kinases are the class of signalling proteins that phosphorylate phospholipids in the cytoplasmic leaflet of the membrane
in general, lipid kinases add a phosphate to the polar head group
phosphorylation of the polar head group results in a conformational change in the phospholipid and allows it to bind to its target protein in the membrane to pass the signal down the pathway
some phospholipids can be phosphorylated more than once to become an active signalling molecule
signalling proteins: adaptor proteins
nearly all signal transduction pathways have another class of proteins that are neither receptors or enzymes
these are known as adaptors
these proteins have different domains that recognize phosphorylated amino acids or other activated structures on signalling proteins
these domains along with others form the glue to hold elements of signalling networks together at the right time and place in a cell
the adaptor proteins are important to allow cascades to be associated in the right space and time to fulfill their tasks when and where they are needed in the cell
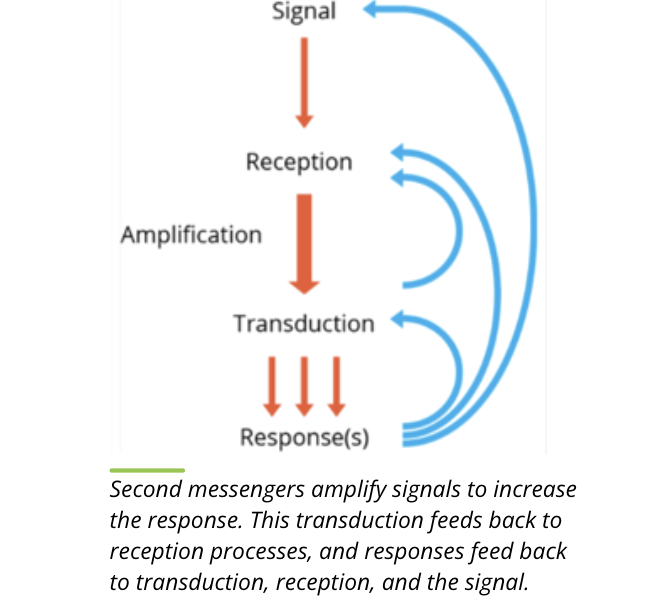
features of second messengers
key features
small in size
diffuse rapidly in the cytosol or membrane
can amplify signals so that the interaction of just a few ligands with their receptors can trigger a much larger response within a cell by mobilizing second messengers
they do not hang around in the cytosol for too long
second messengers such as cAMP and cGMP are degraded by specific enzymes called phosphodiesterases, while ionic messengers such as Ca2+ are sequestered into cellular organelles
other examples include hydrophobic molecules such as diacyglycerol (DAG) and inositol triphosphate (IP3) and some gasses like nitric oxide (NO)
summary of signalling pathways
heterotrimeric G protein signalling cascade
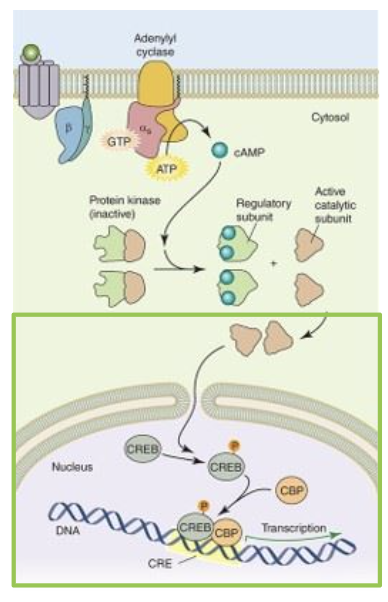
the signal transduction is initiated by the binding of a ligand to the GPCR. binding of the receptor allows the receptor protein to interact with the heterodimeric G protein
the ligand bound receptor stimulates the replacement of GDP for GTP in the alpha subunit. this causes the heterodimeric G protein to disassociate from the receptor and itself to leave a G (beta and gamma) subunit and an activated G alpha s GTP. the G apha s GTP then binds and activates the signalling protein adenylyl cyclase to convert ATP to cAMP, a second messenger
next, cAMP can bind to another signalling protein, protein kinase A (PKA). inactive PKA is a tetrameric protein with two regulatory subunits. the binding of cAMP to the regulatory subunits causes the protein to dissociate and release the active catalytic subunit. once active, the catalytic subunit can phosphorylate a number of cellular proteins
active PKA catalytic domains can enter the nucleus. a common nuclear target is the cyclic AMP response element binding protein (CREB). once phosphorylated by PKA, CREB binds CBP (CREB binding protein) and together, the two proteins can interact with DNA to initiate transcription
Summary
GPCRs
cAMP
PKA
CREB
Phospholipid Kinase Signalling Cascade
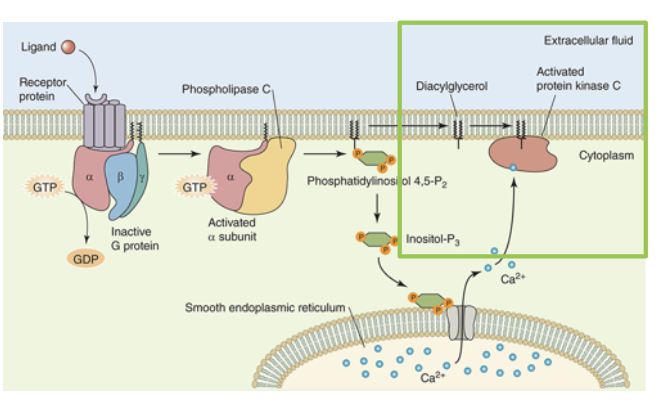
the signal transduction pathway is initiated by the binding of a ligand to the GPCR. binding of the receptor allows the receptor to interact with the heterotrimeric G protein. the ligand bound receptor stimulates the replacement of GDP for GTP in the G alpha subunit. this causes heterotrimeric G protein to to dissociate from the receptor and itself to leave a G beta gamma subunit and an activated G alpha GTP
the G alpha GTP binds the phospholipid kinase signalling protein phospholipase C (PLC)
an activated PLC breaks down the membrane phospholipid phosphatidylinositol 5,5-biphosphate (PIP2) to release two second messengers: DAG and IP3
IP3 diffuses freely in the cytosol and activates its receptor on the ER, which opens a ligand gated calcium channel. Ca2+ leaves the ER and, acting as a second messenger, can activate a number of calcium binding proteins
together, the membrane bound diacylglycerol and cytosolic Ca2+ bind to protein kinase C (PKC), resulting in its activation. activated PKC has numerous cellular targets it can phosphorylate to modulate the target’s activity
Summary
GPCR
PLC
PIP2/IP3
Ca2+
PKC
protein kinase signalling cascade
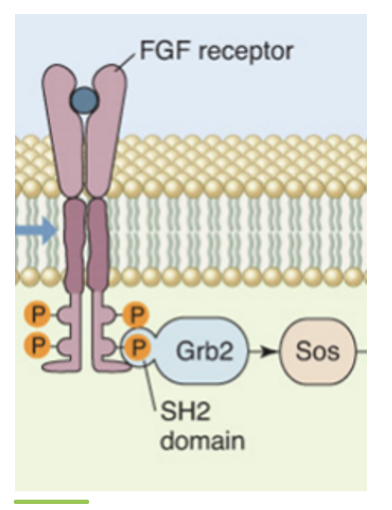
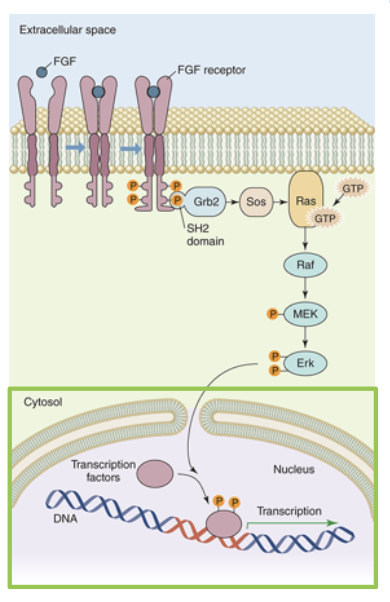
fibroblast growth facgtors (FGFs) are a class of proteins that stimulate the growth of most mammalian cells. FGFs bind to a family of receptor proteins called FGF receptors (FGFRs). FGFR is a homodimeric receptor kinase (tyrosine kinase). binding of FGF to FGFR causes the subunits to dimerize. once bound together, the FGFR undergose tyrosine transautophosphorylation to form phosphotyrosines on the cytoplasmic side. these phosphotyrosines can be bound by many different proteins
one such binding protein is the adaptor protein Grb2. binding to a phosphotyrosine causes Grb2 to undergo a conformation change to bind to Sos. Sos activation leads to its binding to a monomeric G protein Ras. Binding of Sos to Ras replaces the GDP with GTP and the now active Ras can bind to a serine/theronine kinase called Raf. Activated Raf can phosphorylate another protein kinase called MEK, which will in turn phosphorylate another serine/threonine kinase called Erk
phosphorylated Erk forms a dimer and can phosphorylate other signalling proteins in either the cytosol or the nucleus. Erk enters the nucleus to activate transcription factors, ultimately initiating transcription
Summary
FGFs
Grb2 & intermediates
Erk
lysosomes, proteosomes, peroxisomes
lysosomes → organelles that break down misfolded and damaged organelles, nucleic acids, lipids, and more
proteasomes → protein complexes that specifically break down damaged and misfolded proteins in the nucleus and cytosol
peroxisomes → peroxisomes handle dangerous free radicals including reactive oxygen species. thes are also problematic to the cell and needs a safe place to use these chemicals
getting cargo to the lysosome
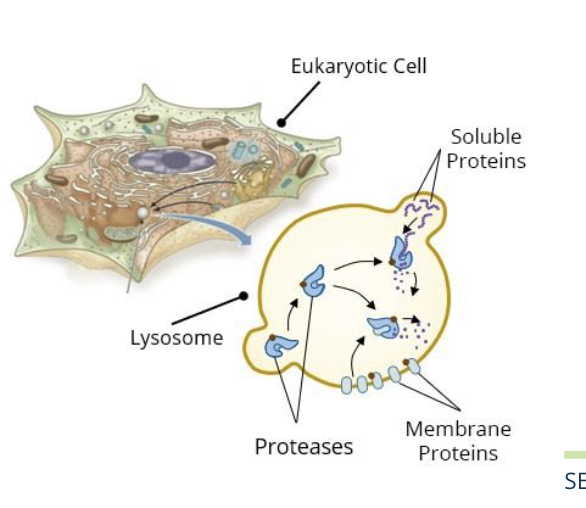
misfolded or non functional proteins ad other cellular contents are tagged for delivery to the lysosome in an endosome via the endomembrane system
cargo is targeted to the lysosome by a specific mannose 6 phosphate (M6P) sugar tag
the enzymes that degrade these damaged proteins are also directed to the lysosome with an M6P tag
vesicles
the engulfed proteins including the membrane proteins and suluble proteins are delivered by vesicles that empty their contents by fusing with the lysosome and are digested by the proteases
Proteases
the proteases are synthesized in the ER, tagged with M6P, and delivered to the lysosomes by vesicles
they digest both soluble proteins and membrane proteins in the lysosome
digestion in the lysosome
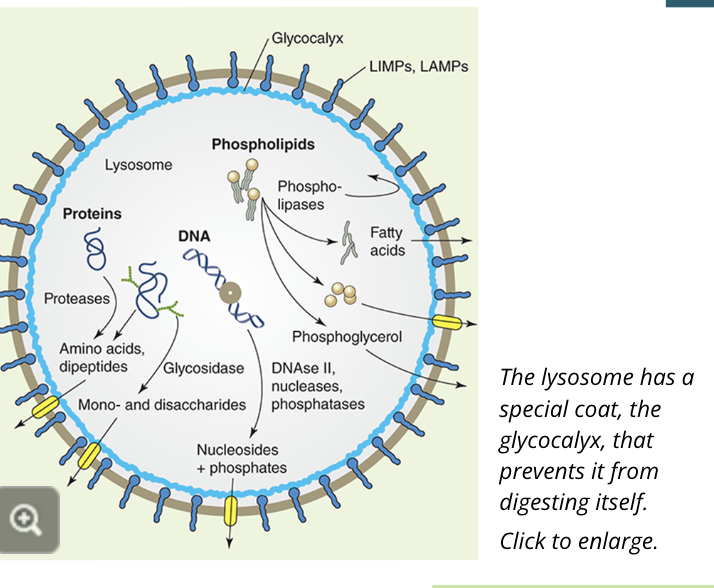
lysosomes are mainly responsible for the breakdown of proteins that are not endogenous to the cell or from other organelles
the lysosome contains high concentrations of proteases, which cleave both membrane proteins and proteins contained within the lysosome
the lysosome also contains enzymes that cleave and digest fats and sugars and can even engulf other organelles like damaged mitochondria or bacteria
once large molecules have been broken down into their basic parts like proteins into amino acids they are transported to the cytosol so the cell can reuse them
protein degradation by the proteasome
cytosolic proteins
cytosolic proteins that have been misfolded or damaged are tagged with a polyubiquitin chain, which is composed of multiple molecules of ubiquitin
multiple ubiquitins are required for the protein to be targeted and recognized by the proteasome and degraded
nuclear proteins
proteasomes are also located in the nucleus so the cell can degrade unwanted nuclear proteins without having to export them to the cytosol
damaged histones, for example, can by polyubiquitinated in the nucleus and degraded by nuclear proteasomes
function of peroxisomes
oxidizing agents like peroxides, ions, and free radicals are very hazardous to the cell
peroxisomes serve as a place to keep and use these reactive oxygen species safely using enzymes including catalase
peroxisomes are small, membrane enclosed organelles and contain enzymes that catalyze a variety of metabolic reactions
essential peroxisome proteins are called peroxins, they are synthesized in the cytosol and are targeted to the peroxisome by specific peroxisomal targeting signals (PTSs)
although they are hazardous, peroxisomes also carry out important decomposing functions for some cargo such as uric acid, amino acids, and long chain fatty acids
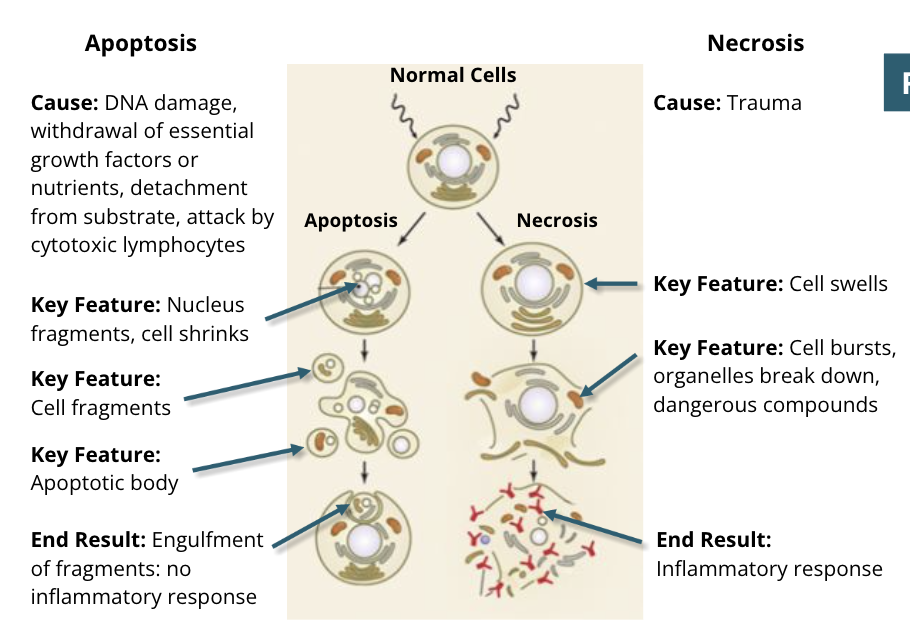
Apoptosis
programmed cell death
an energy consuming process
used to protect the body from damaged cells that no longer function properly
also used in development, ie. to remove the webbing from between fingers and toes in fetal development
kaboom! (but slowly, neatly. very demure, very mindful)
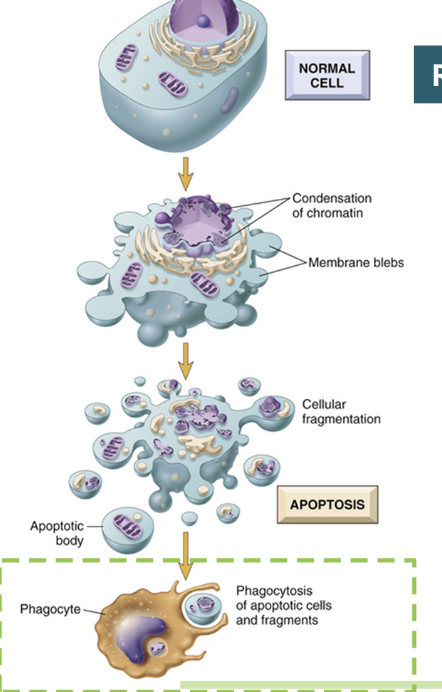
mechanisms of apoptosis: initiation
apoptosis is initiated by two different pathways: intrinsic and extrinsic
the cell initiates apoptosis itself
the intrinsic pathway originates in the outer membrane of the mitochondria
intracellular signals such as sever DNA damage, ROS, toxins, or other trauma will turn on the intrinsic pathway in the cell
extrinsic signals initiate apoptosis in the cell
the extrinsic pathway uses a plasma membrane receptor called the death receptor
neighbouring cells such as immune cells will release death ligands which bind to the death receptor on a damaged cell which activates additional signals that lead to apoptosis
mechanisms of apoptosis: membrane blebbing and enzyme activation
the cell begins to shrink and form blebs (small protrusions from the membrane)
this is the first visible signal that a cell is undergoing apoptosis
enzymes termed caspases are activated
the initiator caspases are activated by either the extrinsic pathway or intrinsic pathway
these caspases will cleave and activate other caspases called executioner caspases
mechanisms of apoptosis: cell structure changes
after the executioner caspases are activated the cell changes structure
DNA is fragmented, often between histones, and DNA repair halts
the nuclear membrane breaks down and the nucleus disappears
the cytoskeleton is disassembled and the plasma membrane phsopholipid content changes with scramblases, woth PS (phosphotidyl serine) being exposed on the exoplasmic leaflet of the plasma membrane
organelles persist, and are enclosed in apoptotic bodies
mechanisms of apoptosis: engulfment
phagocytes endocytose the apoptotic bodies to dispose of them
these are then safely digested by the phagocytes lysosomes
this causes a minimal amount of disturbance to the cells and surrounding tissues
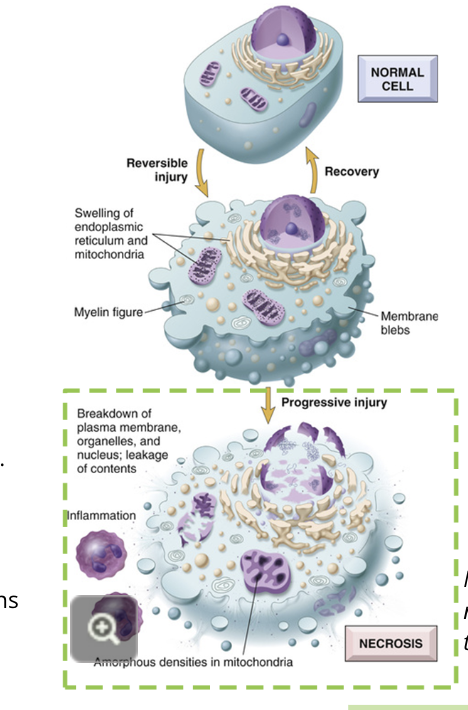
necrosis
resulting from cellular injury that cannot be repaired
the major pathway of cell death as a result of damage that cannot be repaired. the cell’s organelles are not able to function and it dies
mechanisms of necrosis: damage
the cell is damaged beyond repair. there can be many causes
toxins → ie. bacteria, drugs, chemicals
extreme heat or radiation → denatures proteins, damages DNA
freezing → ice crystals puncture the cell membranes and organelles
ischemia → blood flow is stopped to the tissue; lack of oxygen, glucose, etc, prevents the cell from receiving life essentials
pathogens → bacterial or fungal infections
mechanical trauma → physical injury to the cell
mechanisms of necrosis: swelling
the organelles begin to lose their structures and swell
vacuoles, or undefined bodies, form in the cell
depending on the type of damage, the DNA may be degraded
mechanisms of necrosis: destruction
the cell membrane and remaining organelles lose structural integrity
holes can be observed using microscopy
the cellular contents spill out of the cell, producing inflammatory signals
the mitochondria’s proteins are released and lysosomal contents are exposed
cells nearby are exposed to these remains of the cell, and are also damaged or have apoptosis signalling triggered
unlike apoptosis, it is difficult for the body to clean up the cellular remains after necrosis
Apoptosis vs Necrosis