Physical and Chemical Change
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
25 Terms
What are the basic steps of the scientific method?
Observing a phenomenon and identifying a problem or question.
Collecting information about the phenomenon.
Forming a hypothesis, which is a tentative explanation for the observed phenomenon.
Designing and conducting an experiment with clear methods, objective measurements, and reproducible results.
Analyzing the collected data for trends or patterns.
Drawing conclusions to determine whether the data supports the hypothesis or leads to further observations and questions.
A__alters the form or appearance of a substance without changing its chemical composition.
Examples include melting ice, tearing paper, or dissolving sugar in water.
physical change
When a ___ occurs, the atomic structure of matter is not changed to form new products. a change in the size, shape, color, or state of matter of a substance without a change in its chemical composition.
Examples: Mixing, Dissolving, Freezing, Boiling, Evaporating, Condensing
physical change
A ___ results in the formation of a new substance with different properties. This occurs through chemical reactions, such as rusting iron, burning wood, or baking a cake. Signs of a chemical change include color change, gas production, temperature change, or the formation of a precipitate.
chemical change
One or more new substances with different properties are created. For example, hydrogen and oxygen combine to form water, which has different properties than its original elements.
Formation of a New Substance
Chemical reactions may release or absorb energy in the form of heat or light. Exothermic reactions release energy (e.g., burning wood), while endothermic reactions absorb energy.
Energy Change
A noticeable change in color, texture, or other physical properties may occur. For instance, iron reacts with oxygen to form rust, which has a reddish-brown color.
Change in Physical Properties
A chemical reaction may release gas bubbles (e.g., baking soda and vinegar reaction) or form a solid precipitate that does not dissolve in water.
Production of Gas or Precipitate
chemical changes are often ___, meaning the original substances cannot be easily recovered (e.g., burned wood cannot be restored to its original state).
irreversible
How can you determine if a change is physical or chemical?
Physical Change: A substance changes in form or state (solid, liquid, gas) without altering its chemical composition. Examples include water evaporating, ice melting, or carbonation escaping from a soda. These changes are usually reversible.
Chemical Change: A new substance forms with different properties. Clues include gas production (bubbles), formation of a precipitate (solid), color change, or energy change (heat or light). Unlike physical changes, chemical changes are often difficult to reverse.
A. Is there any evidence that a new substance has formed?
A. If yes, the change is chemical. If uncertain, ask the following questions.
B. Was a significant amount of energy gained or lost during the change?
B. A temperature change of more than a degree or two indicates a possible chemical change.
C. Did the properties of the original substances change?
C. There must be more than a change of state. Look for formation of a gas or solid.
D. Is the change difficult or impossible to reverse?
D. “Yes” answer to questions 2,3, and 4 indicates a chemical change.
The growth of grass is a __ because new substances are formed as a result of chemical reactions.
Grass absorbs nutrients and water from the soil and air, using energy from the sun to create new chemical compounds that make up the grass.
Since these reactions produce new matter and cannot be easily reversed, grass growth is classified as a chemical change. This applies to the growth of all living organisms.
chemical change
Physical Change
A physical change is usually being described if:
the change involves a physical process, such as crushing, grinding, or breaking.
there is a change of state, such as melting, freezing, boiling, evaporating, or condensing.
one substance is dissolved in another substance
Chemical Change
A chemical change is usually being described if:
the words “combined” or “reacted” appear.
a chemical equation appears. No chemical equation can be written for a physical change because all the substances keep their original formulas.
Remember that the most important clue to a chemical change is that one or more new substances are formed.
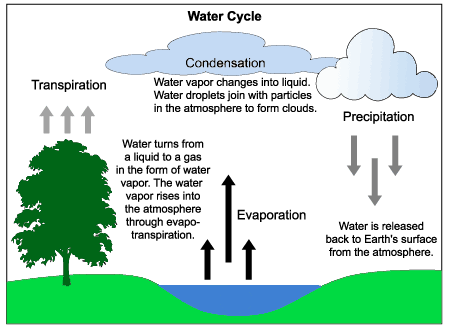
The Water Cycle involves ___, where water continuously changes state (solid, liquid, gas) through processes like evaporation, condensation, and freezing. No new substances are formed.
physical changes
The Rock Cycle includes both ___ Physical changes occur when rocks break apart due to weathering and erosion, while chemical changes happen when minerals react and form new types of rock over thousands of years.
physical and chemical changes.
The water cycle consists of continuous physical changes as water moves through different states:
Evaporation & Transpiration – Water changes from a liquid to a gas (water vapor) as it evaporates from bodies of water or is released by plants (transpiration).
Condensation – Water vapor cools and changes back into liquid droplets, forming clouds.
Precipitation – Water returns to Earth’s surface as rain, snow, sleet, or hail.
What are the key processes in the rock cycle?
The rock cycle describes the physical and chemical changes that form different types of rocks:
Igneous Rock Formation – Molten material (magma or lava) cools and solidifies into igneous rock.
Weathering & Erosion – Igneous rocks break down into small pieces, which are transported and deposited.
Sedimentary Rock Formation – Over time, layers of rock fragments compact and cement together to form sedimentary rock.
Metamorphic Rock Formation – Heat, pressure, and chemical reactions change existing rocks into metamorphic rock without melting them.
Melting & Restarting the Cycle – If metamorphic rock melts, it turns into magma, restarting the cycle as it cools into igneous rock again.