Biochemistry Exam 2
1/128
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
129 Terms
A substrate with negative charges interact with what amino acids?
Positively Charged, basic
How is the affinity of an interaction determined?
Strength and stability of binding interactions
higher kd = less affinity
higher ka = greater affinity for substrate to protein = more binding
What is being measured in the affinity of an interaction?
The measurement assesses the strength and stability of binding interactions between a substrate and a protein.
How are the ka and kd defined?
ka is the association constant, and kd is the dissociation constant.
What is cooperativity? What kind of graphing curve does it have?
binding produces affinity for binding at another site
sigmoidal curve
What drives cooperative binding of O2 in hemoglobin? What are the T and R states of hemoglobin? How do they contribute to cooperativity? Why is cooperative binding of O2 important for Hemoglobin’s function in O2 transport from the lungs to peripheral tissues?
as O2 binds to the sites, the affinity grows in hte other sites
the sites move from T state (tense, low affinity) to R state (relaxed, high affinity) —> gradual relaxation
theres a lot of oxygen in hte lungs whil puts it in a more relaxed state moving through the curve; peripheral tissues get more relaxed as oxygen reaches them
What is allostery? What is the relationship between cooperative binding and allostery?
the binding of one site affects another
cooperative binding is a more specifc verson of allostery where it increases affinity as in binds
How can ATP hydrolysis be used to drive protein conformational changes?
Ex. muscle contraction : as ATP hydrolyzes, myosin and actin are aligned in the right place to contract then return to normal positioning
How can protein conformational changes be harnessed to provide information in biological processes such as growth factor signaling?
acting as molecular switches
ligand bindings triggers structural shits that alter enzyme activity (allosteric changes) facilitating signal transduction by altering protein affinity
would happen to contraction if ATP hydrolysis was blocked, but ATP binding was not?
Why are ADP and Pi release equally as important for muscle contraction as ATP hydrolysis?
What would happen if Pi release was blocked
failure to contract
drive “power stroke” or conformational change that align myosin and actin. and cross-brige attachment of myosin from actin which restarts the cycle
If Pi release blocked, no power stroke or conformational change
How do enzymes increase the rate of catalysis? What is their effect on ΔG?
Overcome energy barriers
Affects rates, not DG’0 or DG
Binding energy contribution to account for first law of thermo
Chymotrypsin mechanism****
How does affinity to substrate affect the reaction? What is the induced fit model?
faster when affinity is higher
induced fit is when an enzyme changes conformation to fit the substrate (transition state), pushing transformation forward
1. Why would an enzyme with high complementarity to the substrate be a poor catalyst?
What structure should an efficient enzyme prefer? What is the “transition state”?
What are transition state analogs?How does binding energy affect enzyme function? How does binding energy contribute to catalysis?
Higher complementarity would not push the substrate to the product (not fitting the transition state)
the transition state (in between the substrate and the product
they mimic the transition state of the substrate. Binding energy is released whebn the substrate binds and helps overcome activation energy. It stabilizes the transition state (pushed to product).
How are enzyme kinetics measured/determined—how is data collected to create a Vo/[S] graph? What parameter is held constant for these graphs? What is the Vo of a reaction? A Vmax? What is Km? How are they determined?
What would two enzyme reaction graphs look like for the same enzyme and substrate, but with twice the amount of enzyme added in one of the reactions?
What is the relationship between Km and affinity of enzymes for substrates? Does Km only describe affinity of the enzyme for the substrate?
What is kcat/Km? How is this related to enzyme efficiency? What does it mean that two enzymes can have the same Km for a substrate but different efficiencies? How can this occur?
Double-Reciporacal Plot
line
Lineweaver-Burk Plots for inhibition
competitive vs Uncompetitive vs. Noncompetitive
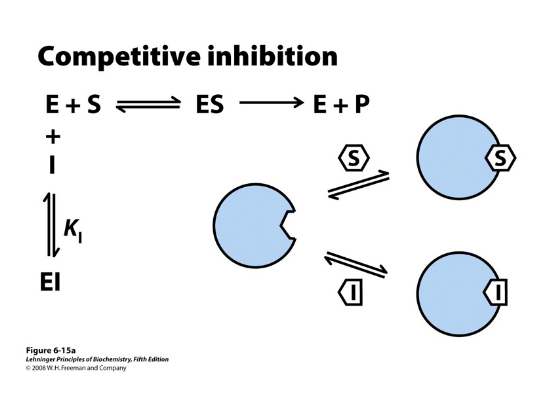
Competitive inhibition Kinetics
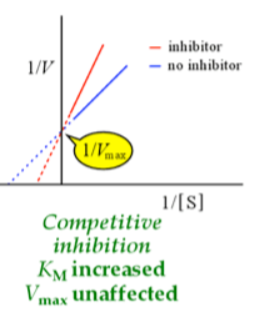
km increases (more substrate required to compete with the inhibitor) — x intercept chages, steeper slope
Vmax unaffected — y intercept does not change, once out compete, get to max velocity just need more substrate— turnover didn’t change, enzyme can still work at its max rate
competitor bind to the ENZYME, EI complex, not ES complex
doesn’t always bind to the active site it can be an allosteric
inhibitor: its binding to an
allosteric site could prevent the
enzyme from binding substrate.
If the substrate binds first, the
inhibitor can’t bind and catalysis
can happen. The “competition”
is for binding the enzyme first,
independent of where that
binding occurs
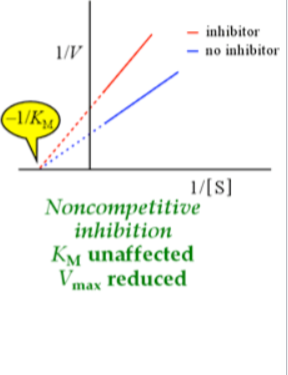
noncompetitive inhibition
bind at allosteric regulatory site (at E+S, beginning of reaction)— decreases activity of enzyme/change access to active site
Km unaffected (can’t increase substrate to overcome allosteric inhibition) — x-intercept stays the same
Vmax reduced — y intercept changes, Inhibitor can bind to enzyme with or without substrate, so apparent Km doesn’t
change but Vmax decreases
mixed inhibition: could bind to E+S or ES complex (2 options)
Both S and I can be bound to enzyme at same time or separately; i.e., Substrate binding is
unaffected, but catalysis is decreased
Inhibitor binds equally well to E or ES,
but it’s binding affects the conformation
of the catalytic site, decreasing the rate
of catalysis
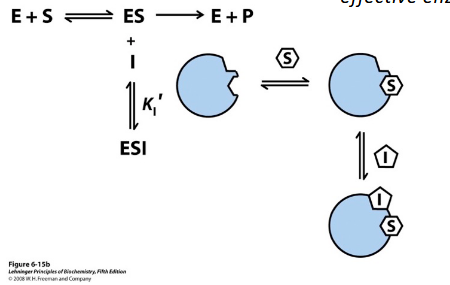
uncompetitive inhibitor
binds enzyme-substrate complex (after they come together), then controls and stops turnover/process
Km reduced - saturating protein earlier, can’t release substrate (temporarily locked → bad = taking enzyme out of solution
affinity is increasing because adding ESi complex→ favors backward reaction→ foming more ES (Le Chatlier’s)→ need lower Km for substrate to bind to
Vmax reduced
change proportionally to one another
Inhibitor will only bind ES complex; prevents or slows
ES→ EP and ES→E+S; Vmax is lower but apparent Km is
also lower as product release is slowed and ESI decreases
effective enzyme concentration,
Uncompetitive inhibition cannot be
reversed by increasing [S]
![<p>binds enzyme-substrate complex (after they come together), then controls and stops turnover/process</p><ul><li><p>Km reduced - saturating protein earlier, can’t release substrate (temporarily locked → bad = taking enzyme out of solution</p><ul><li><p>affinity is increasing because adding ESi complex→ favors backward reaction→ foming more ES (Le Chatlier’s)→ need lower Km for substrate to bind to</p></li></ul></li><li><p>Vmax reduced</p></li><li><p>change proportionally to one another</p></li><li><p>Inhibitor will only bind ES complex; prevents or slows</p><p>ES→ EP and ES→E+S; Vmax is lower but apparent Km is</p><p>also lower as product release is slowed and ESI decreases</p><p>effective enzyme concentration,</p></li><li><p>Uncompetitive inhibition cannot be</p><p>reversed by increasing [S]</p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/98d434de-8aea-415a-ab4d-acac0a4a9fbd.png)
What is Km? What would it mean if the Km is low?
affinity for substrate
if lower, has high affinity because not a lot of substrate is required to reach Vmax
half of Vmax
***Vmax = y intercept
What is the Vmax?
the max velocity of an enzyme reaction/ max turnover
What does the inhibitor bind to in uncompetitive inhibition?
ES complex at an
What does a high Km mean?
low affinity
you would need to throw higher concentration of substrate at the enzyme because the enzyme doesn’t want it as bad (it’s lazy)
In a hydropathy index, the higher or more postive the index, means the residue is_______
hydrophobic
Irreversible Inhibition
totally kills the enzyme, the reaction doesn’t return - ***typically because of a covalent bond between inhibitor and enzyme → others usually release making them reversible
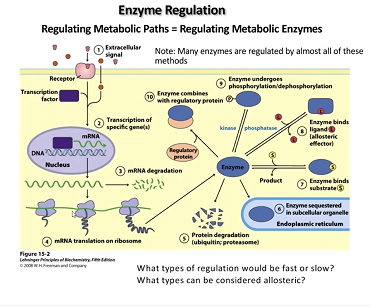
Enzyme Regulation
post-translational modification
phosphorylation - glucogenesis, kinase activity (opposite is phosphotase activity)
ubiquination - get rid of protein after doing its job
ex. acetylation, adenylation,
****pathways
in allosteric regulation, is it exactly like an on or off switch?
no, there is always leaky transcription, never truelyl one or off, just different levels of activity
to not run out of resources or burn out the system
***why drugs are not always effective — also apply to competitive inhibition
Regulatory enzymes
pathway control
often composed of regulatory subunits and catalystic subunits
exhibit non-Michaelis-menten kinetics
some exhibit cooperativity with small increases in the substrate
Where is the activity modulation binding site/ Allosteric Site?
Typically in regulatory subunit physically distant from the catalytic site in the quaternary strcucture
Homotropic Regulation
one of the substrates acts as an allosteric regulator
Importance of first phosphate in glycolysis?
to prevent reversibility of the process
added by PFK
phosphate adds to active site, ATP binds to active (high affinity) and allosteric site (low affinity, only binds when theres a lot of ATP→ negative regulation -→ homotropic regulation
phosphate→ glucose→ fructose→
Heterotropic Regulation
regulatory enzymes regulated by negative and positive modulators
homotropic and heterotropic working together to make a “dial” that turns the system up or down, not on or off
Aspartate Transcarbomoylase (ATCase) and heterotrophic modulation
important regulatory enzyme in the pathway of pyrimidine biosynthesis (make nitrogenous bases/nucleotides)
formation: carbamoyl Phosphate + aspartate
has 12 polypeptides assembled into 6 catalytic units plus 6 regulatory subunits
CTP binds to it in inactive T state → releases in active R state
Homotropcic Regulation: increases in aspartate and carbamoyl-P cause gradual T→ R state change
Heterotrophic regulation: moving enzyme in two different directions depending on the needs of the cell
Postive Heterotrpic modulation: ATP stimulates it → lots of excess energy → anabolic processes (replicate DNA, make more nucleotides)
Negative Modulation: CTP binding to allosteric sites
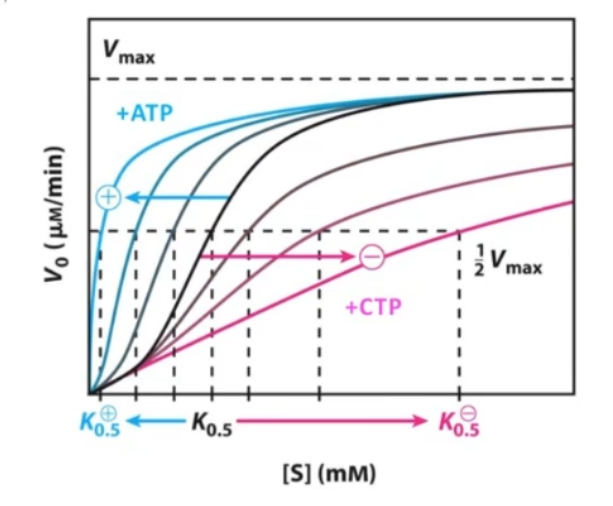
What is CTP?
pyrimidine nucleotide
inhibits ATCase in negative modulation
Why does it make sense for CTP to negatively modulate ATCase? (pyrimidine biosynthesis)
in the subunits there are multiple places for CTP to bind, and when it binds at these allosteric sites, it inhibits through negative modulation
Why would high levels of ATP be a logical activator of ATCase?
Postive Heterotrpic modulation (ligand binds to allosteric site increasing affinity): ATP stimulates it → lots of excess energy → anabolic processes (replicate DNA, make more nucleotides)
Homotropic activation in ATCase
an increase in either substrate promotes shift from T→ R state yielding sigmoidal kinetics
similar to cooperative binding, the sigmoidal kinetics is due to the combination of two distinct kinetics as a function of [S]
![<p>an increase in either substrate promotes shift from T→ R state yielding sigmoidal kinetics</p><ul><li><p>similar to cooperative binding, the sigmoidal kinetics is due to the combination of two distinct kinetics as a function of [S]</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/9caa3f24-9e14-484d-9a5b-05fa128cb746.png)
Heterotrophic Modulation
ATP positively modulates and promotes R state even at lower [S]
CTP negatively modulates and promotes T state even at high [S]
![<ul><li><p>ATP positively modulates and promotes R state even at lower [S]</p></li></ul><ul><li><p>CTP negatively modulates and promotes T state even at high [S]</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/31d5dcec-4795-4244-b920-019d498971c5.png)
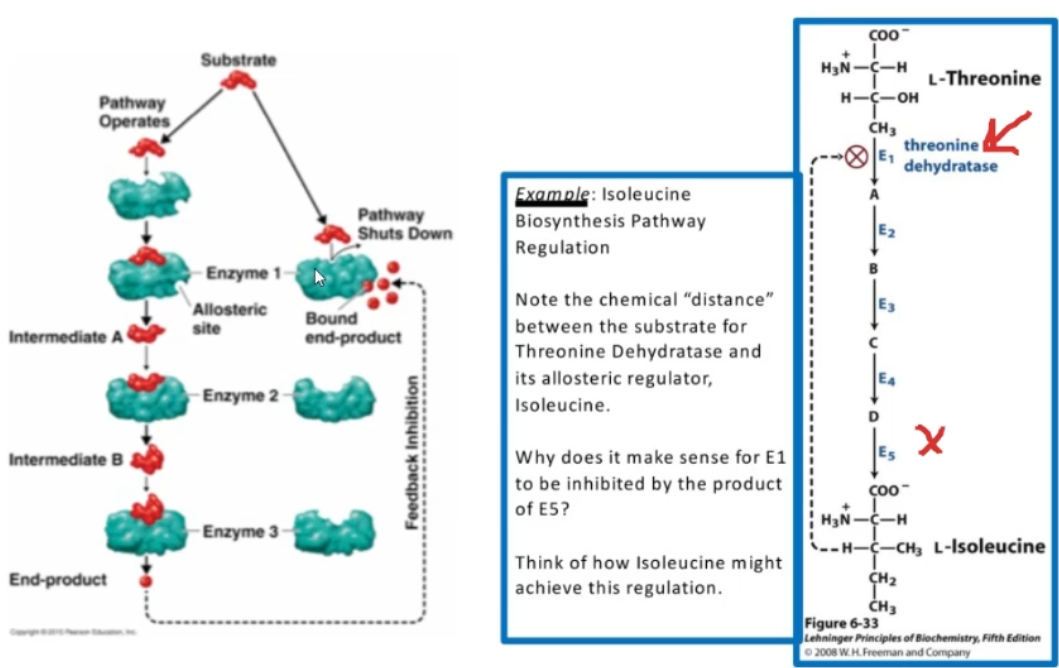
Regulatory enzymes provide pathway _______ ________ — a common theme in metabolic regulation
“feedback inhibition”
ex. Isoleucine biosynthesis pathway regulation
Threonine substrate→ isoleucine allosteric regulation
In a sigmoidal curve, shifting to the left means the enzyme what?
has more affinity, is more effective at binding substrate and creating product
Isoleucine Bisynthesis regulation pathway
Glycogen phosphorylase
Glycogen: Stored form of glucose kept largely in muscle and liver. Mobilized when here is an urgent need for glucose oxidation or during low nutrition.
Glucose is freed as Glc-1-P by a phosphorylysis reaction (addition of phosphate and breaking of glycogen)--- it is then converted to
Glc-6-P for entry into glycolysis when other sources of glucose are low
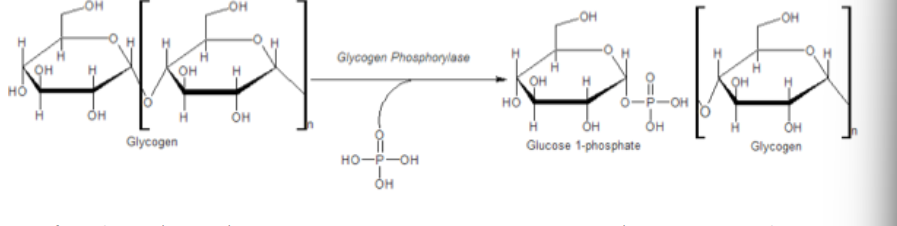
Explain how glycogen phosphorylase exists as an equilibrium of two states
(Relaxed) R-State: More active state
(Tense) T-State: Less active state (still active tho)
Phosphorylation of Serine 14 (A in diagram)by Phosphorylase Kinase shifts the equilibrium so the more active R state is favored (still possible to be inactive but the active state predominates)
Hormone signaling, such as adrenaline,
activates Phosphorylase Kinase
further modulation of sigmoidal curve: some heterotropic and homotropic regulation: in A more glucose-6-P → T state, in B more glucose or ATP → T state
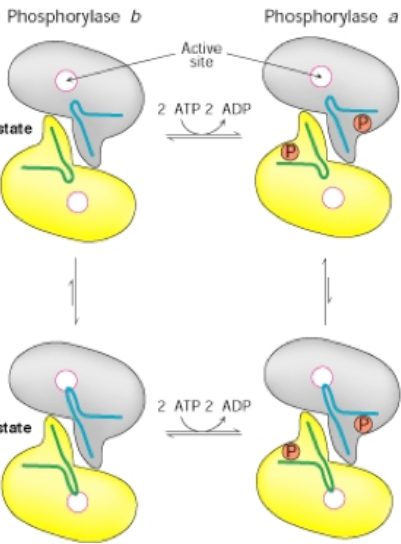
What is adrenaline, what physiological
response does it direct, and why would
adrenaline’s activation of phosphorylase
kinase make sense in muscle?
Covalent and/or Allosteric Regulation of Glycogen Phosphorylase
Conversion of T (less active) to R (more
active state) is also enhanced by AMP
enhanced by AMP + phosphoylation
What does increased [AMP] in the cell indicate
and why would increased Glycogen
phosphorylase activity be a good response in
muscle cells?
increased AMP → PKA activity→ phosphorylase kinase phosphorylated → activated phosphorylase A→ mobilized glycogen storage
activated when there isn’t enough glucose : starving
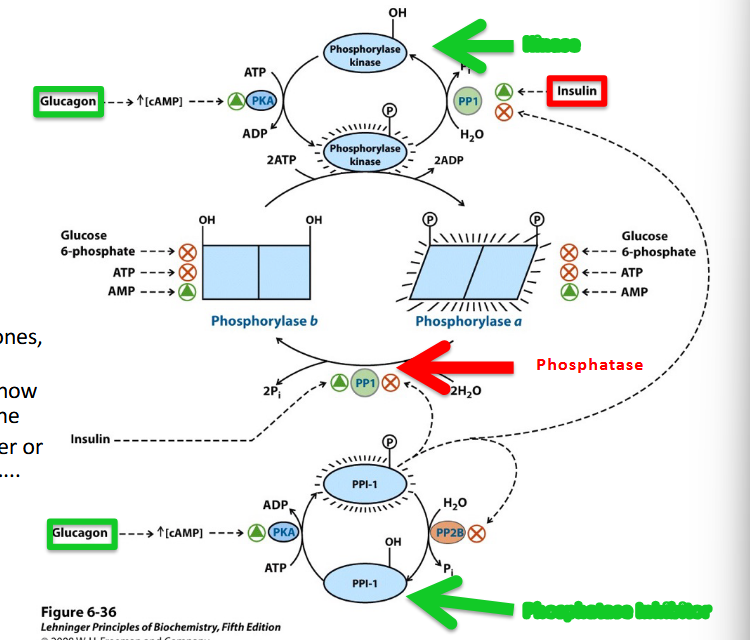
Glycogen Phosphorylase is highly regulated by Both Kinetic and Post-Translational modifications (PTM) Controls: ***pathway****
Phosphorylation on Serine
converts it from a lower
activity form
(Phosphorylase b) to the
higher activity form
(Phosphorylase a)
****Note roles of metabolic hormones,
Insulin (high blood Glc) and
Glucagon (low blood Glc) and how
they influence the activity of the
enzyme and determine whether or
not to release Glucose stores…...
insulin (removing phosphate groups): make more glycogen, absorbed sugar, anabolic activity, phosphorylase activity deactivated phosphorylase and brings it back to phosphorylase B
glucagon: increase phosphotase activity
when out of whack -→ diabetes
*****Think about the goals of insulin and glucagon signaling and why their opposing regulation of glycogen
phosphorylase makes sense….
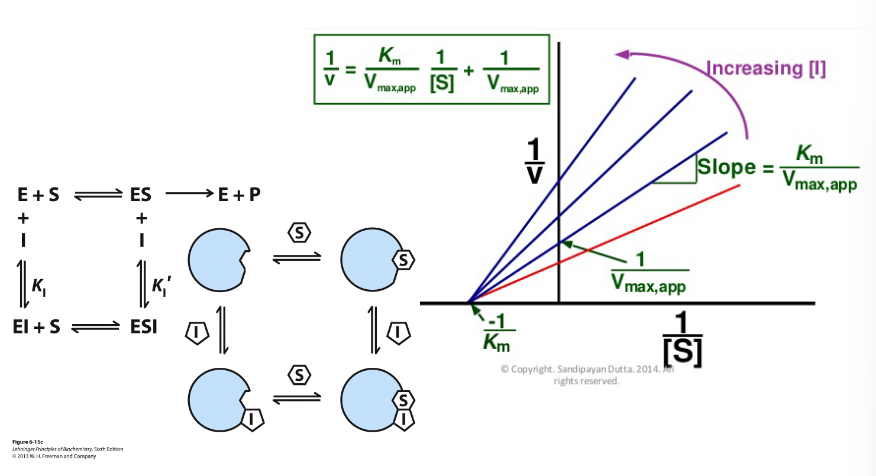
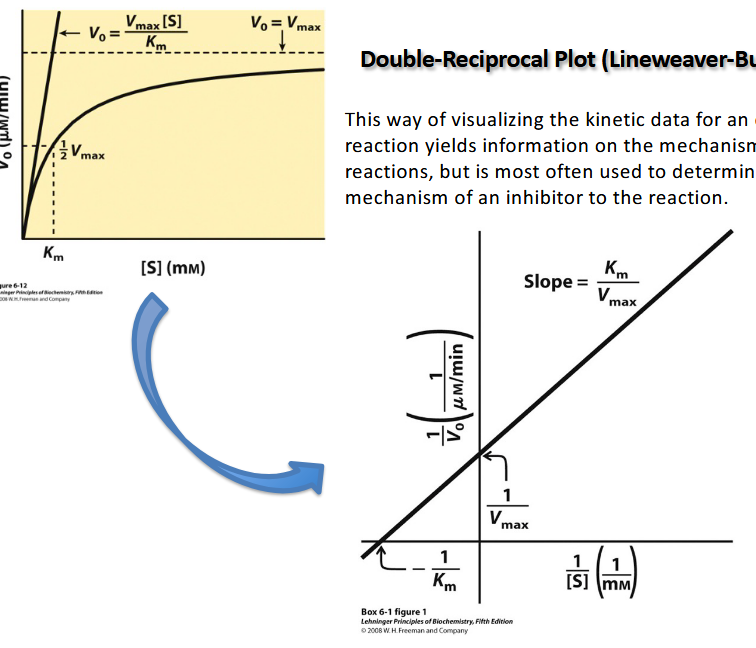
Double- Reciprocal Plot (Lineweaver-Burke Plot)
This way of visualizing the kinetic data for an enzyme
reaction yields information on the mechanisms of the
reactions, but is most often used to determine the
mechanism of an inhibitor to the reaction
aKm
aparrant Km of the substrate in hte presence of an inhibitor
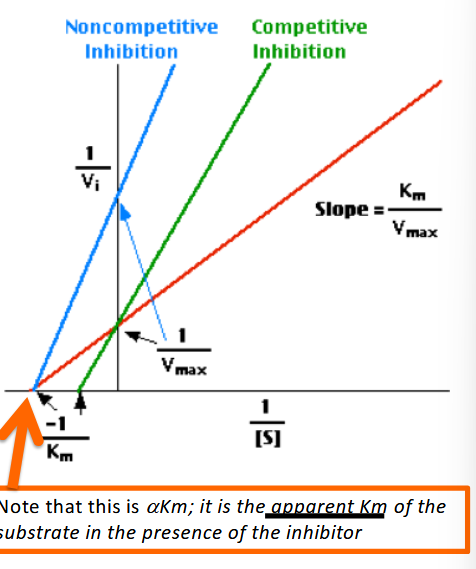
Does non-competitive have a sigmoidal curve?
yes
Does Uncompetative inhibition have a sigmoidal ccurve?
no, not allosterically regulated but binds to the ES complex
Regulatory enzymes exhibit reaction kinetics that are _______ due to changes in kinetics that occur with additional substrate binding (cooperativity)
sigmoidal
To survive cold environments, fish would need more _____ in their membranes
unsaturations
Flippases
send lipidsagainst concentration into cell using ATP
Floppases
send lipids against concentration gradient outside of cell using ATP
ER Scramblase
move lipids down gradient
restore equilibrium/maintain balance if there is asymmtry where there shouldn’t be
cell and organelle membrane advantages
structure, permeability keeping things in or out, doing work (electron transport, receptors for signaling, cellular communication and identification)- outweigh disadvantages
Which has more asymmetry Plasma Membrane or ER and why
more asymmetry in plasma membrane than ER
ER doesn’t need asymmetry so there is no flippases or floppases present
Types of Flippases
require ATP: flippases and floppases
Don’t: scrablases
**both are enzymes
Can you establish a concentration gradient without ATP?
no
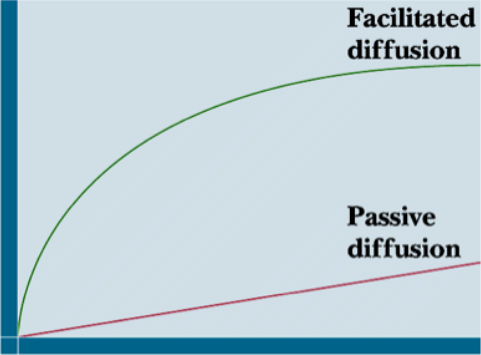
Passive vs. Facilitated diffusion kinetics
facilitated (similar to non-regulatory curve/ligand receptor binding) - increase in movement, but get saturated and level off
passive - totally dependent on concentration gradient
Integral membrane proteins embedded within lipid bilayer
alpha-helical structure and beta-barrel structure
eukaryotes have channels made of alphahelicies
beta-barral in prokaryotic organisms: ex.pathogens make channels
What goes easy through the membrane
small, nonpolar
What have trouble and what can’t go through membrane
big, polar - trouble
ions- can’t
Types of membrane transport
active (require energy/ATP, postive DG - moving hard to move molecules, unidirectional) and passive (diffusion and facilitated diffusion, negative DG, reversable)
difference between uniport, cotransport symport and antiport
*****can be facilitative or active
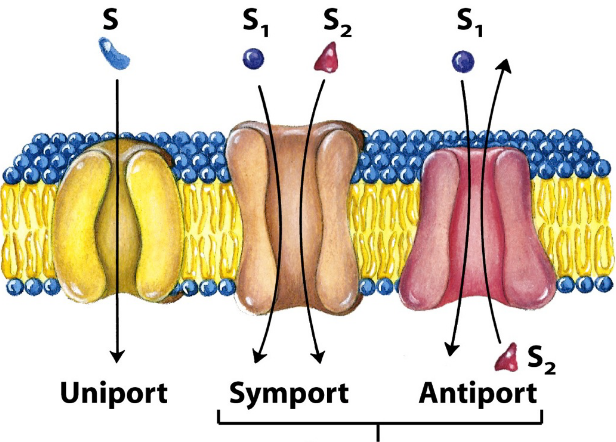
Facilitated transport
down concentration gradient/across energy barrier -→ eliminates need to remove hydrogen bonding/hydration shell (would be required with simple diffusion)
hydrophilic molecules require transporter
hydrophobic molecules don’t need it
transporters can have conformational change (even though don’t need ATP — stacked cylinders rotating in or out to be specific channels
alpha helicies - these can rotate: open or close → more specificity/regulated
beta-barrel structures have no control
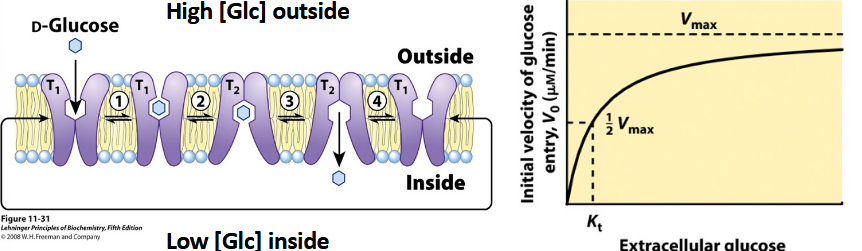
What types of transporter is glucose transporter?
facilitated transporter
induced fit → conformational change→ product release
T1 = ES
T2 = EP
not reversible
what would make it reversable?
Aquaporin
alphahelical structues
function: maintain fluid balance in many tissues, urine concentration, tears, edema etc.
Na+'/Ka+ Pump / Na+K+ ATPases
active transport: energy depended to move against gradient
antiport
makes cytosol more negative than outside: 2K+ in 3Na+ out, ECF or blood plasma: positve
simultaneously pump Sodium and Potassium
against their concentration/ electrochemical
gradients
Results in charge imbalance across a
membrane creating membrane potential for
current….action potentials in neurons
P-type ion pump: reversible autophosphorylation of the protein
Ca2+ ATPase
type of transport: active
regulate intracellular calcium levels
When there is no ATP, what can make ATP?
electron transport chain
Is glycolyis anerobic?
up to the production of pyruvate yes, but then it requries oxygen to the ETC
In glycolysis, lots of NADH does what?
push the reverse reaction (regulating metabolic “flux” overlaping with their roles as electron carriers for RedOx chemistry: high NADH drives reduction
chemistry; high NAD+ drives oxidation chemistry. High NADH also slows glycolysis–
reduction of pyruvate to lactate helps alleviate this “block” in exercising muscle
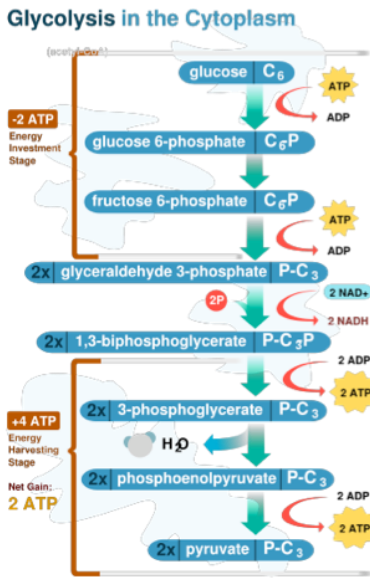
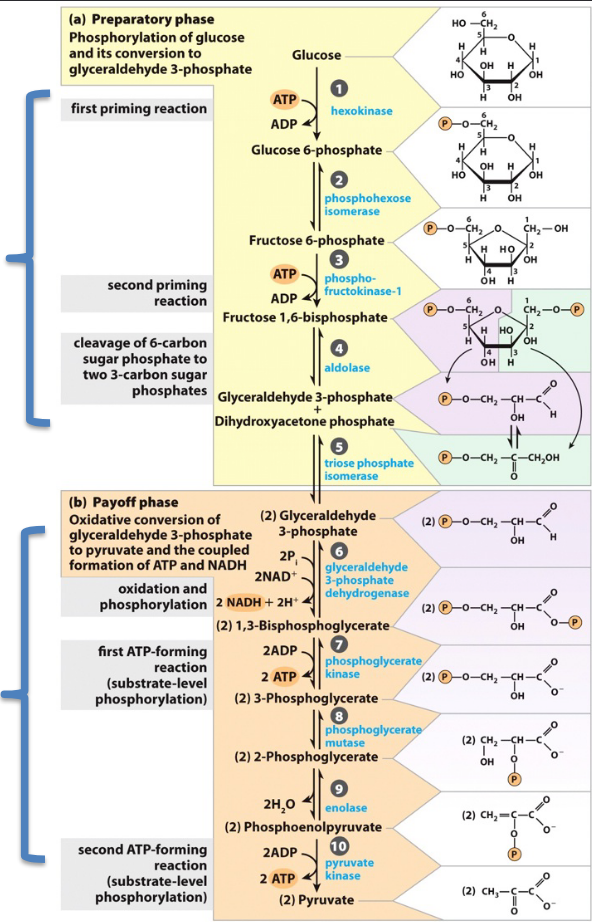
Glycolysis
glucose (6 carbons) → 2 pyruvate (3 carbons): oxidation
energetically favorable overall, but energy expenditure in Preparatory phase
is required to provide Payoff in the energy-producing phase
highly favorable reactions are sites of regulation– to control the pathway and
its direction, the process must be blocked before each plunge off the “cliffs”
more from summary of lecture 10….
afterwards, Citric acid cycle creates ATP with ETC
glucose into cell, add phosphate (anchored in cell), make into fructose with P, splits into two 3 carbond moelcules, 2 steps into PEP, into 2 pyruvate
uses ATP in steps 1 and 3, oxydized by NAD+ in step 4, creating ATP in steps 5 and 7
ATP needed to phoshporylate
sugars on the outside of cells are for
communication, structure, recognition and identification
glucose anabolism to cell wall and extracellular matrix to create structural polymers
Energetics of glycolysis
more endergonic in standard conditions than exergonic
in nature, bring down energy required to keep them going with enzymes, energy coupling, compartmentalization
Becoming more favorable inside the cell - key steps required big changes in free energy
hydrolyzing ATP has big energy changes (investing into phosphoylation with Hexokinase (keep glucose inside cell) and Phosphofructokinase (commit moleculeto glycolysis))
regulate the big drops by facilitating for blocking it —these places are difficult to reverse because going down the hill is easy, going up is hard
Highly favorable steps of glycolysis
a) sites of regulation
b) reactions that Gluconeogenesis (reverse
pathway) has to avoid or go around
catalyzed by hexokinase, phosphofructokinase-1 (PFK1), and pyruvate kinase.
2 phases of glycolysis
preparatory phase (glucose → G3P) - Sets up glucose as a higher energy substance prepped for -lysis and eventual extraction of energy: An up front investment of 2 ATP
payoff phase (G3P → Pyruvate)- Rearranges carbons to
form strong phosphate donors to
phosphorylate ADP; Reap a 100%
return on ATP investment, with some
NADH as a bonus
in ETC, what is NADH? what is made?
electron donor to make ATP
What can feed into glycolysis?
many sugars
lactose (made of galactose and glucose) - hard because the galactose need extra steps and enters glycolysis in a different part than glucose
sucrose - easy
In lactose, what is fed in where? Why is it not favored for glycolysis?
glucose → glycolysis
galactose → broken down into UDP-gal→UDP-glu→ glucose 1-phosphtate (not favored for glycolysis because of extra steps, extra time and energy investment
What is the regulatory enzyme in glycolysis?
PFK-1
When is energy required in glycolysis?
phosphorylate to keep in cell
destabilize to split into two molecules
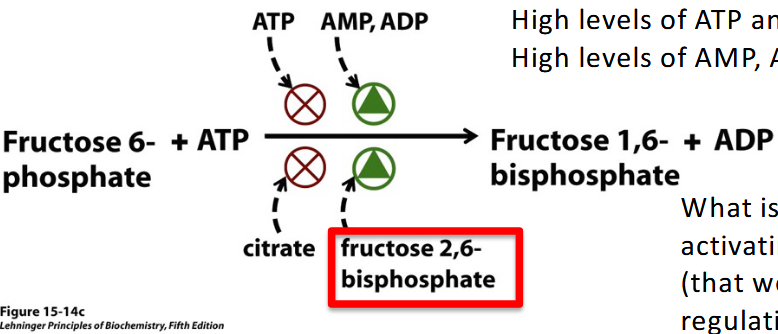
What is the difference between fructose 1,6-Bisphoshate and fructose 2,6-Bisphosphate?
fructose 1,6-Bisphoshate : component in glycolysis in the preparatory phase
fructose 2,6 - Bisphosphate - stimulates glycolysis by activating PFK 1 and inhibits gluconeogenesis
2 important enymes in step 1 of glycolysis
Hexokinase and Glucokinase
Hexokinase/Glucokinase Reaction
traps glucose in the cell → glucose-6-phosphate
raises glucose core energy level for lysis reactions
changes glucose to something different so that the glucose transporter reaction is no longer reversible
What can Glucose- 6- phosphate be turned into? ***metabolic cross road
Glucose-1-phosphate (on different carbon) → glycogen (energy storage)
6-phospoglucono-delta?-lactone → ribose-5-phosphate→ nucleotide biosynthesis
fructose-6-phosphate→ glycolysis (oxidation of fuel)
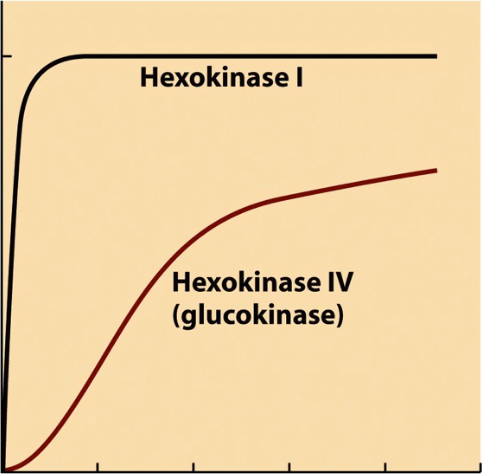
2 isoforms of hexokinase
Hexokinase I-III
low Km
where low levels of sugar are because of high affinity
Glucokinase (Hexokinase IV)
higher Km
sigmoidal shape - allostery (affinity changes)
primarily in liver, a tissue where glucose storage occurs
metabolic sensor
important for long-term homeostasis - required sustained insulin release
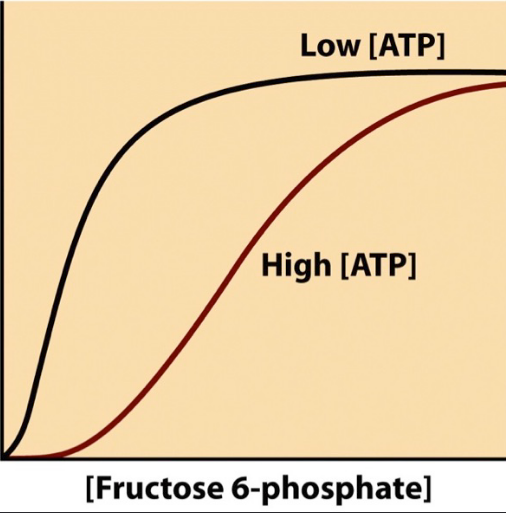
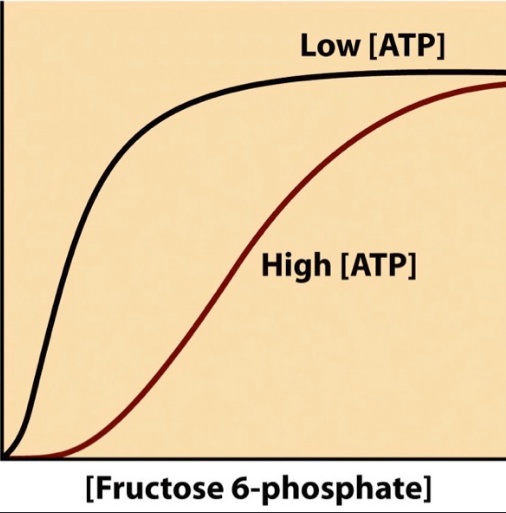
What is ATP to PFK I?
negative allosteric inhibitor
The key components in going from Fructose 6-phosphate-→ fructose 1,6-bisphosphate
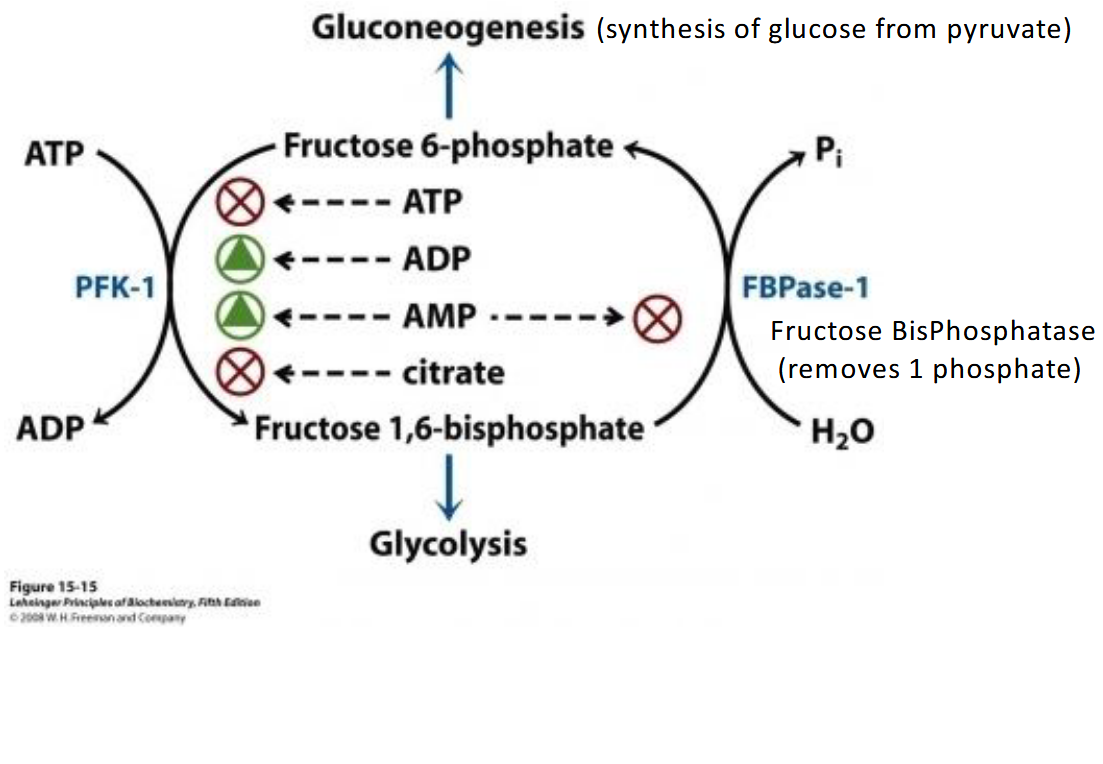
What acts as the opposite of PFK-1in glycolysis, for gluconeogenesis?
FBPase-1 (removes 1 phosphate with H2O)
What inhibits FBPase-1 in gluconeogenesis?
AMP
What is gluconeogenesis?
synthesis of glucose from pyruvate
Where does the first step of oxidization in glycolysis occur?
beginning of payoff phase to create 1,3-BPG
What does Adolase yield in glycolysis?
2 molecules of G3P
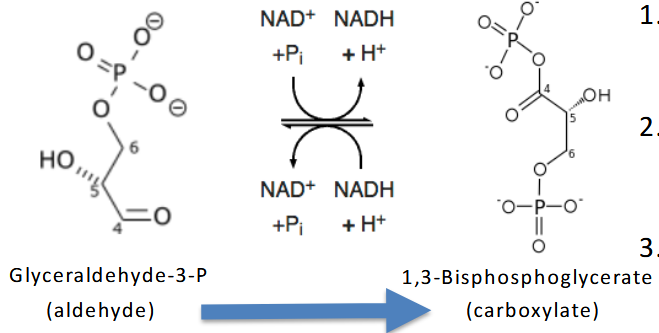
GAPDH– Glyceraldehyde-3-P Dehydrogenase
G3P → 1,3-bisphosphoglycerate
Two Step Reaction:
1. Highly exergonic oxidation of aldehyde
to carboxylic acid coupled to Reduction
of NAD+
2. Highly endergonic transfer of inorganic
phosphate to yield high energy
compound 1,3-BPG
3. NOTE: Reaction requires oxidized form
of NAD
Although the sum of the DG of
The reaction is somewhat
unfavorable, the next reaction
quickly removes 1,3-BPG and
thus decreases Q and lowers
overall DG
PEP is a high energy ______ donor
phosphate