Exam 3 Intermolecular Forces + Formal Charges
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
19 Terms
dispersion
all atoms and molecules
dispersion appearance
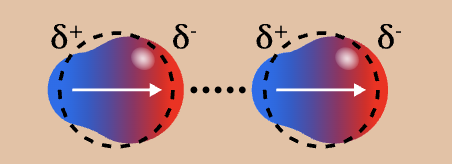
dispersion strength
¼ (weakest)
dipole-dipole
polar molecules
dipole-dipole appearance
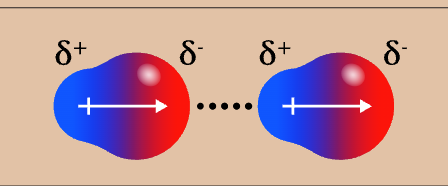
dipole-dipole strength
2/4 (weak)
hydrogen bond
molecules of H bonded to F, O, N
(highly electronegative elements)
hydrogen bond appearance
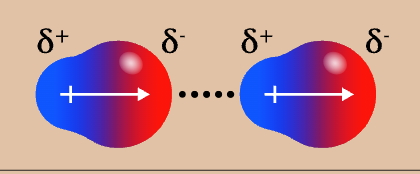
hydrogen bond strength
¾ (strong)
ion-dipole
mixtures of ionic compounds and polar compounds
ion-dipole appearance
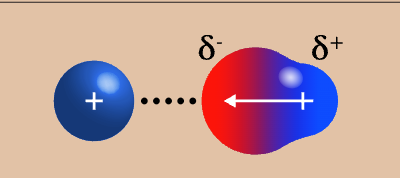
ion-dipole strength
4/4 (strongest)
ionic compounds
metal + nonmetal, transfer of electrons
covalent compound
nonmetal + nonmetal, sharing of electrons
polar compound
nonmetal + nonmetal, unequal sharing of electrons
non-polar compound
nonmetal + nonmetal, equal sharing of electrons
formal charge
valence electrons - (bonds+dots)
one bond
one formal charge number
pair of dots
two formal charge number