AP BIOLOGY Review
AMINO ACIDS
\
- there are 20 amino acids * Our proteins are made of some of these combinations of these amino acids
- there are 4 levels of protein structure * primary - the order of the amino acids in that chain (string) * secondary - helix - alpha helix - beta pleated sheets (coils and sheets) * tertiary - an individual polypeptide * quaternary - made up of 4 polypeptides (Transthyretin) two alpha subunits and two beta subunits
- bonds between amino acids are peptide bonds - formed by a dehydration reaction - a water molecule is produced from that action
- hydrogen bond
- Di-sulfide bridge - firm covalent bond * Van Der Waals interactions (no need to explain it)
- ionic bond
- hydrophobic interaction is when things that are hydrophobic are pushed together (in oil in water)
- 4 atoms of iron for one Hemoglobin molecule
\
SUGAR-PHOSPHATE BACKBONE
\
- Sugars have carbons on unlabeled corners
- they can have a number of carbons but the most common are glucose and fructose (six carbons)
- phosphate ground has 4 oxygens one double bond and the central atom is a phosphate
- sugars have a phosphate group, a sugar, and a nitrogenous base
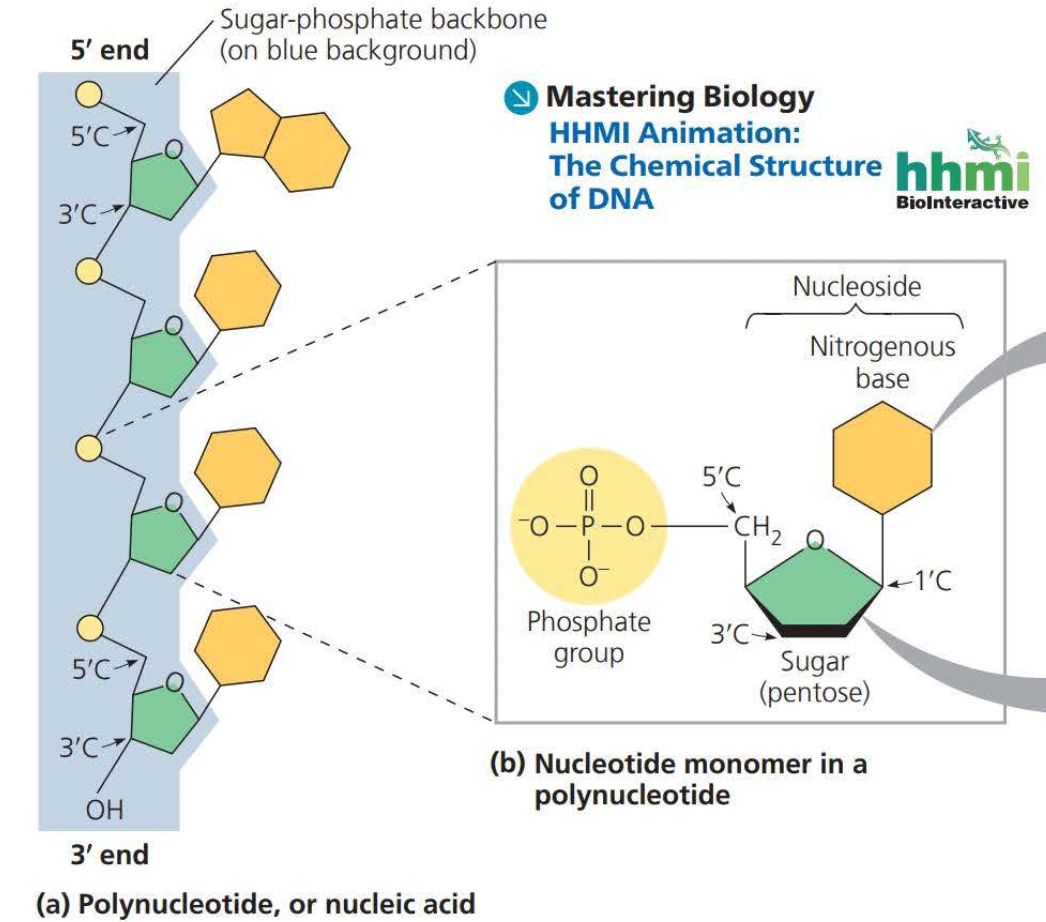
- The picture represents an RNA because its a singular strand and not a double strand 5’ and 3’ are opposites so in DNA one will be 5’ and the other will be 3’
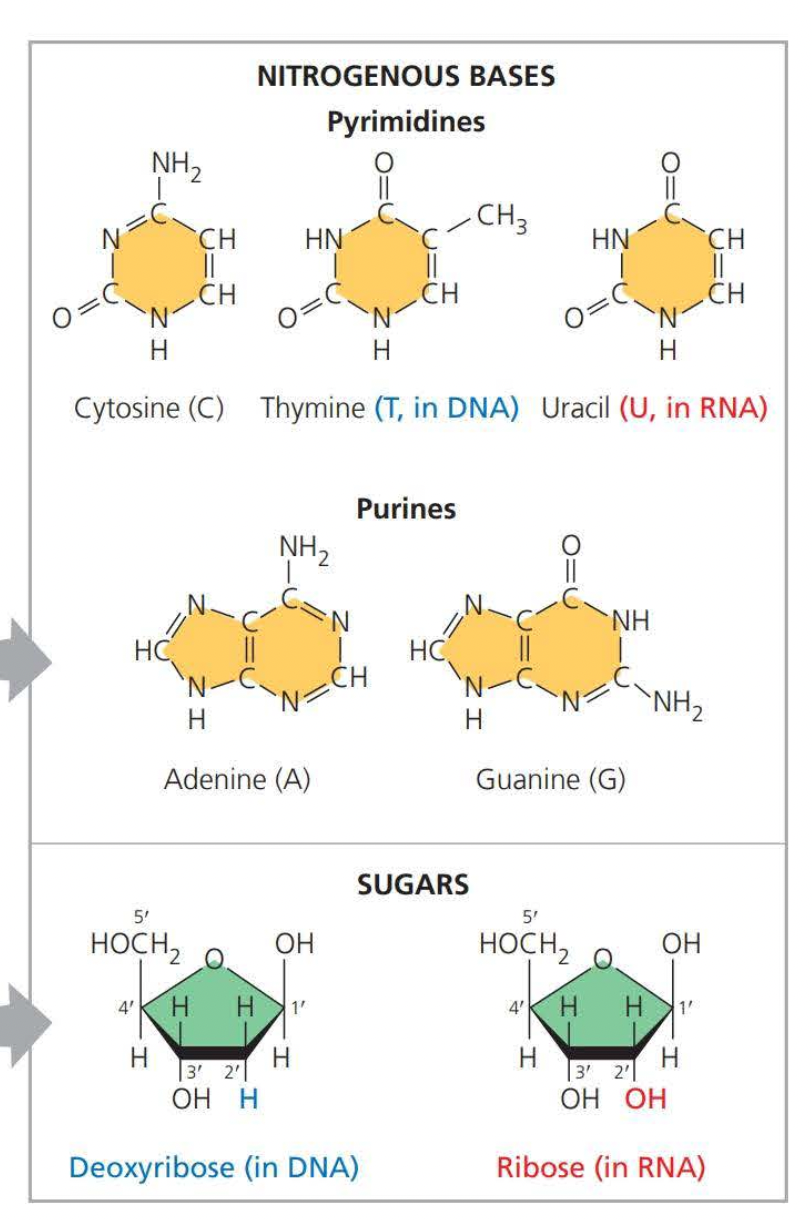
- Need to know that pyrimidines are singular rings
- And purines are double rings
- RNA may fold on itself but will still be a single strand
- RNA components * Adenine * Cytosine * Thymine * Uracil
- DNA components * Adenine * Cytosine * Thymine * Guanine
- Adenine - Thymine
- Cytosine - Guanine
or
- Cytosine - Uracil
\ \ Diagram of an RNA folding over on itself

- CHECK THE CHAPTER REVIEWS IN CAMPBELL’S BIOLOGY IN FOCUS
\