Local Anesthetics
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
What is a local anesthetic?
Agents used to block pain sensation by depression of excitation of the nerve potentials and inhibition of conduction process in the peripheral nerves.
define topical anesthesia
Aqueous solutions, sprays, ointments or gels intended for topical penetration
define ionophoresis
Inject though skin using an electrical current
define nerve block anesthesia
insert close to a nerve trunk but proximal to the intended area of anethesia
define infiltration anesthesia
Injecting directly into the area to be anesthetized
define field block anesthesia
Injected closer to a larger terminal nerve branch, subcutaneous or submucosal
What is the mechanism of local anesthesia?
reversible blockage of peripheral nerve conduction by inhibition of the movement of the nerve impulse
describe peripheral nerve conduction
myelin sheath prevents the local current from flowing across the membrane, and the current flows down the nodes of Ranvier. Upon stimulation, ion channels propagate the action potential to the next node
What are 2 things resting potential and the ability to generate an action potential depend on?
1 - concentration of electrolytes in the ECF
2 - Permeability of nerve membrane to Na+ and K+ channels
How do local anesthetics affect nerve conduction and action potentials?
Local anesthetics bind to voltage-gated Na⁺ channels and block the generation and conduction of action potentials.
How many alpha subunits are in a voltage-gated Na⁺ channel, which segment senses voltage changes, and what happens to that segment to open the channel?
There are 4 alpha subunits; segment 4 senses the voltage change; the segment becomes displaced.
what is the Na+ inactivation region located?
between the PKC site and inactivation trimer
how to determine rapid onset of an anesthetic
pKA of local anesthetic is close to the pH of the tissue
What are some properties of local anesthetics?
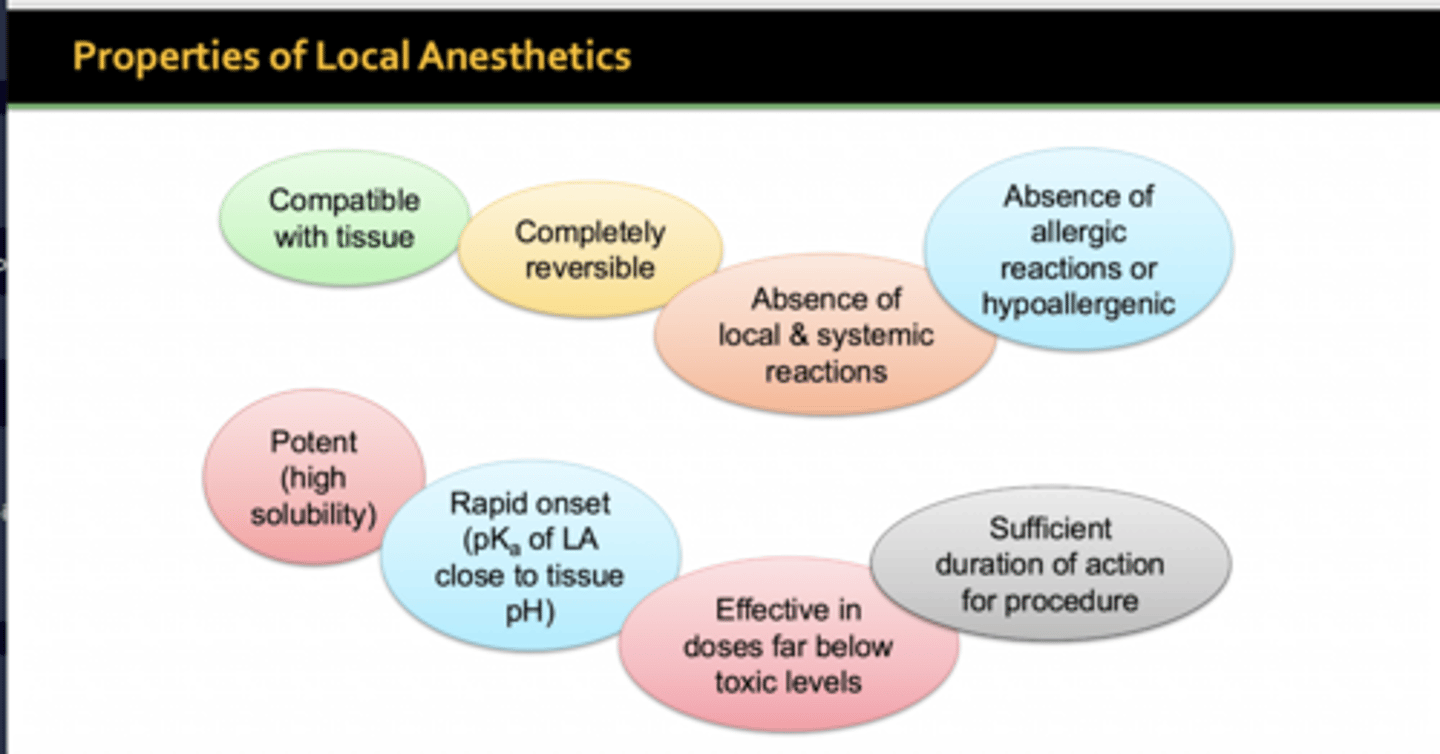
How are ester-type anesthetics commonly used, what are they associated with, and how are they metabolized?
Ester-type anesthetics are commonly used topically, are associated with allergic reactions, and are metabolized in tissue by pseudocholinesterase.
Where are amide-type local anesthetics metabolized, what properties do they have, and what do they bind to?
Amide-type local anesthetics are metabolized in the liver, have better lipid solubility, potency, and duration of action, and bind well to plasma proteins.
what structure of a LA contributes to lipophilicity and why is this important
aromatic residue; essential for drug to permeate the nerve sheath and nerve membrane
what structure of a LA contributes to hydrophobicity and why is this property important?
amino terminus, allows drugs to be dissolved in aqueous solution and prevents it from precipitating in interstitial fluids
pH and charge of LA lipophilic region
7.4; uncharged
pH and charge of LA hydrophilic region
4.5-6; cation
What type of compounds are local anesthetics that exist in equilibrium between nonionized and ionized forms?
weak bases
what is the pKa of an LA?
pH at which non-ionized and ionized forms of LA are equal
What is the ionized form of a local anesthetic?
salt form
What is the pka of LA affected by?
pH of the tissue
How does inflamed tissue affect local anesthetics and their absorption into nerves?
Inflamed tissue can be more acidic, which ionizes the local anesthetic and prevents absorption into the nerve.
2 vasoconstrictors used with local anesthetics in dental procedures
epinephrine or levonordefrin
What does the selection of vasoconstrictors depend on?
length of the dental procedure
requirement of postoperative pain control
medical status of the patient
what must require careful attention with addition of vasocontrictors
patients with uncontrolled cardiac history, hyperthyroidism, or using beta- blockers
some things that addition of epinephrine is contraindicated with
BP over 200 syst. or 115 diast
severe cardiovascular disease
daily episodes of angina
medication with a B blocker, MOAIs, TCAs, general anesthesia with halothanes
What determines the potency of local anesthetics, what does duration of action depend on, and how does protein binding affect toxicity risk?
Lipid solubility dictates potency, duration of action depends on protein binding, and increasing protein binding decreases the risk of toxicity.
2 things that affect elimination
metabolism and excretion
3 ester type LA
procaine (novocaine)
benzocaine
tetracaine
What is the major adverse reaction of procaine?
allergic reaction
major adverse reaction of benzocaine
methemoglobinemia, contraindicated in children under 2
What is a adverse reaction of tetracaine?
contact dermatitis, burning, stinging, angioderma
What is the pKa of lidocaine and why is this significant?
7.8 ; Lidocaine has a pKa close to physiologic pH (~7.4), so a larger fraction is in the nonionized form, allowing it to cross nerve membranes quickly and produce a rapid onset.
What is lidocaine with epinephrine commonly used for?
almost all dentistry
What form of mepivacaine should be used to avoid vasoconstrictors?
Plain mepivacaine
What are some adverse reactions of mepivacaine?
dizziness, tremors, blurred vision
What condition can ortho-toluidine induce? What LA causes this?
methemoglobinemia ; prilocaine
why does bupivacaine have a long duration of action? What is it? How could this potentially be a problem
it binds will to proteins; 340-440 minutes it could bind to Na+ channels necessary for other systems and lead to toxicity, try not to give in cardio conditions
Articaine is metabolized by what?
AChE
what part of Articaine structure conveys lipid solubility?
thiopene ring
What are some adverse reactions of ropivacaine?
hypotension, nausea, vomiting, bradycardia
How do maximum safe doses of local anesthetics differ between cardiovascular patients and healthy patients?
Maximum safe doses are lower in cardiovascular patients than in healthy patients.
some psychogenic reactions to LA administration
vasogagal
hyperventilation
anaphylactoid reaction
What are key features and causes of idiosyncratic reactions to local anesthetics?
Toxic reactions at small doses, often anxiety-induced, associated with vasoconstrictors, accidental IV injection, sulfites in solutions, and can occur with inhaled or ingested forms especially in asthma patients.
How common are allergic reactions to local anesthetics, what can they range from, and which group most commonly causes true allergic reactions and why?
Allergic reactions are rare, can range from rash to anaphylactic shock, and most true allergic reactions occur with ester local anesthetics due to production of PABA.
What factors is local anesthetic toxicity directly related to?
It is directly related to the rate of absorption and elimination.
two main systems affected by LA
CNS and cardiovascular system
What is more common to experience toxicity CNS or CVS?
CNS
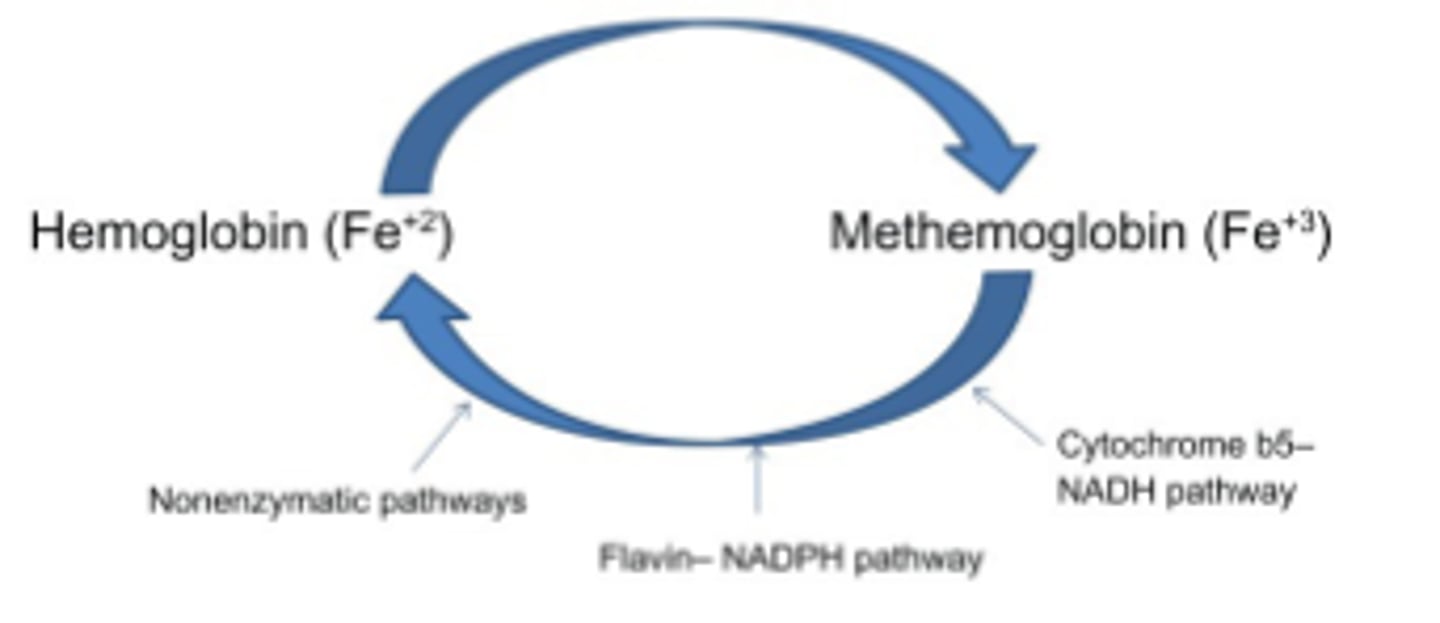
some symptoms of methemoglobinemia
shortness of breath, cyanosis, headache, fatigue, seizures
reversed by IV methylene blue
draw the cycle of methemoglobinemia
3 ways to prevent LA toxicity
1 - give the lowest dose required for anesthesia
2 - use proper injection techniques (always aspirate)
3 - use a vasoconstrictor containing agent if not contraindicated
mechanism of reversal of local anesthetics
administration of phentolamine to shorten the duration of action of LA containing a vasoconstrictor and produces alpha-adrenergic blockade of vascular smooth muscle causing vasodilation
things that can cause failure of local anesthetics
1 - inflamed tissue (lower pH, increased blood flow, alteration in Na+ channel numbers)
2 - incorrect needle selection
3 - starting procedure prior to maximum LA effect